-
PDF
- Split View
-
Views
-
Cite
Cite
Yuta Endo, Shu Soeda, Yuki Yoshimoto, Yukitoshi Todate, Tatsuru Hashimoto, Shigenori Furukawa, Yasushi Teranishi, Keiya Fujimori, Successful management of locally advanced cervical cancer via total pelvic exenteration following neoadjuvant chemotherapy, Journal of Surgical Case Reports, Volume 2024, Issue 7, July 2024, rjae452, https://doi.org/10.1093/jscr/rjae452
Close - Share Icon Share
Abstract
Stage IVA cervical cancer is a tumor that invades the mucosa of the bladder or rectum without distant metastasis and is difficult to treat, and concurrent chemoradiotherapy is recommended. Although radical surgery following neoadjuvant chemotherapy is a treatment option for stage IVA cervical cancer, the evidence is limited. A 51-year-old woman with bulky cervical cancer and rectal invasion was referred to our hospital. Paclitaxel and cisplatin were administered as neoadjuvant chemotherapies. After two cycles of chemotherapy, the tumor size decreased markedly. Total pelvic exenteration was performed, and a complete resection was achieved. Four cycles of paclitaxel and cisplatin were administered postoperatively. Thirty-three months after the completion of adjuvant chemotherapy, the patient was alive and free of disease. Radical surgery after neoadjuvant chemotherapy may be a treatment option for stage IVA cervical cancer with bulky tumors.
Introduction
According to the International Federation of Gynecology and Obstetrics (FIGO), stage IVA cervical cancer (CC) is a tumor that invades the mucosa of the bladder or rectum without distant metastasis, comprising 3.1% of cases; concurrent chemoradiotherapy (CCRT), including platinum-containing chemotherapy and brachytherapy, is recommended [1, 2]. Treatment complications can be severe, including fistula formation presented at the time of diagnosis and development during or after treatment, and the prognosis of stage IVA disease is poor; the 5-year overall survival rate ranges from 5% to 50% [1, 3, 4].
Neoadjuvant chemotherapy followed by radical surgery (NAC-RS) is an option for the treatment of local advanced CC. NAC aims to reduce tumor size, facilitate surgical resection, reduce complications through surgical management, and improve local control (LC) [5]. However, data on NAC-RS for stage IVA CC are limited [6, 7]. No ongoing clinical trials have compared NAC-RS and CCRT for stage IVA CC [1]. Herein, we present a case of a bulky CC directly invading the rectum that was successfully managed via total pelvic exenteration (TPE) following NAC.
Case report
A 51-year-old woman with CC with rectum invasion was referred to our hospital. Vaginal examination revealed a continuous neoplastic lesion extending from the cervix to the vaginal wall, directly invading the rectum. The histopathological diagnosis was squamous cell carcinoma. Her serum squamous cell carcinoma antigen (SCC-Ag) was elevated to 19.2 ng/ml. Magnetic resonance imaging (MRI) and computed tomography (CT) revealed a 91 × 73 mm mass that replaced the cervix and posterior vaginal wall and directly invaded the rectum without hydronephrosis (Fig. 1a and b). Positron emission tomography (PET)-CT revealed no distant metastases. Stage IVA CC was diagnosed according to FIGO 2018.
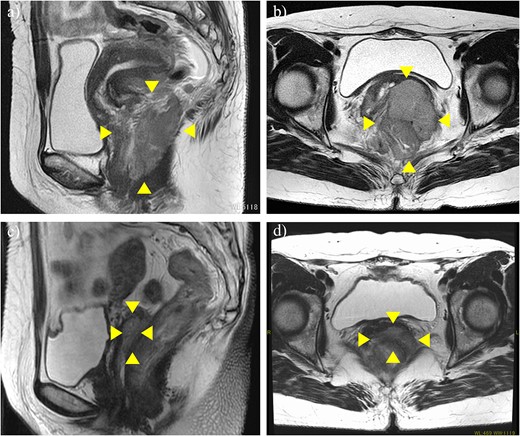
MRI before neoadjuvant chemotherapy (a, b) and after two cycles of paclitaxel and cisplatin (c, d). Arrowheads show the cervical tumor. The tumor shrank markedly after neoadjuvant chemotherapy.
Although CCRT following colostomy was suggested, the patient refused and strongly preferred surgical cytoreduction. According to a radiotherapist, LC may have been difficult with CCRT because the tumor was too bulky. Therefore, we decided to perform NAC-RS. TP therapy (paclitaxel 175 mg/m2 and cisplatin 50 mg/m2) was initiated as NAC. After two courses of chemotherapy, her serum SCC-Ag was reduced to 1.8 ng/ml. CT and MRI revealed a marked reduction in tumor size (Fig. 1c and d) and the absence of the novel lesion. TPE, including radical hysterectomy bilateral salpingo-oophorectomy, radical cystectomy, Hartmann procedure, pelvic lymphadenectomy, colostomy, and ileal conduit placement, was performed (Fig. 2). The duration of surgery and blood loss were 617 min and 1670 ml, respectively. Blood transfusion was required. Macroscopically, no lesions were observed in the cervix. Pathological examination of the surgical specimen revealed squamous cell carcinoma with left internal iliac node metastasis (ypT4N1M0). The surgical margin was negative for cancer. Recovery was uneventful, and the patient was discharged 21 days postoperatively. Forty-nine days after the surgery, TP therapy was restarted, and another four cycles were administered. Thirty-three months after the completion of adjuvant chemotherapy, the patient was alive without recurrence (Fig. 3).
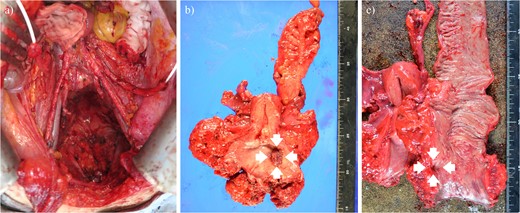
Operative finding after tumor resection (a) and surgical specimen (b, c). Arrows show the tumor that directly invaded the rectum.
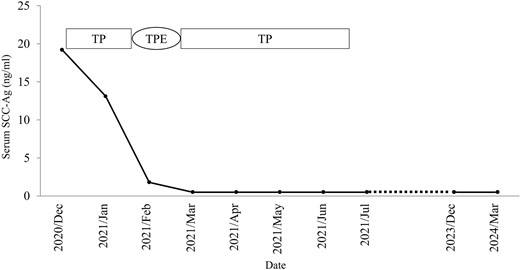
Clinical flowchart of the patient. SCC-Ag, squamous cell carcinoma antigen; TP, paclitaxel and cisplatin; TPE, total pelvic exenteration.
Discussion
Here, we presented a case of bulky CC with rectal involvement that was treated with TPE following NAC and showed no evidence of disease for 33 months after treatment.
The EMBRACE-I study evaluated the efficacy and morbidity of MRI-guided adaptive brachytherapy in patients with locally advanced CC and showed excellent 5-year LC (91%). In contrast, the 5-year disease-free survival (DFS) and overall survival (OS) rates were 47% and 52%, respectively. Fistulae and ureteric strictures were more common in stage IVA CC (18.6% and 21.3%, respectively) than in stage IB1-IIB cancer (1.3% and 0.7%, respectively) [4], suggesting that in stage IVA CC, even if LC is successful, the recurrence rate is high and the prognosis is poor. Several reports have demonstrated that a large tumor before RT or CCRT was a significant unfavorable factor [8, 9]. Wakatsuki et al. [10] demonstrated that rectal invasion is a poor prognostic factor in stage IVA CC treated with radiotherapy. Therefore, a combination of systemic chemotherapy and surgical cytoreduction may be necessary to improve the prognosis of bulky-stage IVA CC, as observed in the present case.
Clinical trials have compared NAC-RS with CCRT. Gupta et al. [11] conducted a randomized controlled trial in the Tata Memorial study. In this trial, the 5-year DFS was 69.3% in the NAC-RS group and 76.7% in the CCRT group (hazard ratio, 1.38; 95% confidence interval (CI), 1.02–1.87; P = .038). NAC-RS resulted in inferior DFS than CCRT, although OS was equivalent. In the EORTC 55994 trial, the NAC-RS group was comparable in terms of 5-year OS (71.7% vs. 75.5%; P = .297), but inferior in terms of 5-year progression-free survival (56.9% vs. 65.6%; P = .021). However, these clinical trials included only stage IB to IIB CC, and to our knowledge, no ongoing clinical trials have compared NAC-RS and CCRT for stage IVA CC. One retrospective study demonstrated the efficacy of NAC-RS against stage IVA CC [6]. In this study, NAC was feasible in half the patients with stage IVA CC. A multicenter retrospective study recently demonstrated that NAC-RS improved the survival outcomes of locally advanced CC, including stages IB2 to IVA, compared with that via CCRT [12]. 81.8% of the patients showed a pathological response (complete and partial response) after NAC, and it was concluded that patients who achieved NAC-RS may benefit from long-term survival. NAC-RS may be a treatment option for bulky stage IVA CC, as in the present case. However, in these reports, the regimens were not uniform, and the results were controversial.
Recently, the efficacy of immune checkpoint inhibitors for CC treatment has been demonstrated. The KEYNOTE-A18 demonstrated that pembrolizumab combined with CCRT significantly improved PFS in patients with newly diagnosed, high-risk locally advanced CC [13]. Given the results of this clinical trial, the opportunity for NAC-RS may decrease. However, we believe that it is important to preserve the NAC + RS option.
In conclusion, NAC-RS may be a treatment option for stage IVA CC with bulky tumors. This type of management can reduce the tumor size and complications associated with surgical management, improve the outcome of local resection, and avoid complications associated with fistula formation.
Acknowledgements
The authors would like to thank Editage (www.editage.com) for English language editing.
Conflict of interest statement
None declared.
Funding
None declared.



