-
PDF
- Split View
-
Views
-
Cite
Cite
Lea Timmermann, Moritz Schmelzle, Evgenij Potapov, Marcus Müller, Christian Lojewski, Johann Pratschke, Wenzel Schöning, Extended right posterior liver sectionectomy for HCC in a patient with left ventricular assist device—a case report, Journal of Surgical Case Reports, Volume 2020, Issue 9, September 2020, rjaa307, https://doi.org/10.1093/jscr/rjaa307
Close - Share Icon Share
Abstract
Successful implementation of left ventricular assist devices lead to a prolonged survival in patients with chronic terminal heart failure. Thus, patients with pre-existing left ventricular assist devices with abdominal comorbidities requiring abdominal surgery, e.g. for malignancy, are upcoming issues. We carried out a major liver resection for hepatocellular carcinoma in a patient with pre-existing left ventricular assist device. The importance of this case report is that it outlines the significance of oncologic resections in patients with left ventricular assist devices as an upcoming issue and provides an interdisciplinary approach.
INTRODUCTION
In chronic heart failure left ventricular assist devices (LVADs) are a well-established option [1]. In our centre 110–130 devices are implanted every year. Leading to a prolonged survival, comorbidities requiring abdominal surgery, e.g. oncologic resections, are upcoming issues.
CASE REPORT
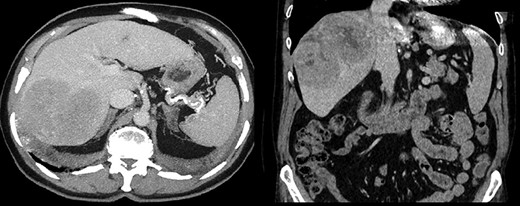
A 64-year-old male with severe ischemic cardiomyopathy underwent implantation of an LVAD (Heart mate II™, Abbott, Chicago, USA) in 2012. Decision of LVAD as destination therapy instead of heart transplantation was made due to history of hepatitis B infection, chronic kidney insufficiency and COPD. Patient remained in good condition with normal LVAD function. Further history included atrial fibrillation and pulmonary embolism. In 2019, hepatocellular carcinoma (HCC) was incidentally detected in the postero-lateral sector of the liver. Preoperative LiMax-Test [2] revealed good liver function (316 μ/kg/h), MELD XI Score was 15 without signs of portal hypertension. Staging revealed no distant metastases and confirmed local resectability (comp. Fig. 1).
Anticoagulation was performed with Vit K antagonists and one episode of LVAD partial thrombosis appeared in 2015. The patient was admitted 3 days prior to surgery allowing conversion to full heparinization with target aPTT prior to surgery, on postoperative day (POD) 1, 2 and 3 set as: 55–60 s; 45–50 s; 50–55 s and 55–65 s.
Because driveline was located in the way of planned laparotomy, surgery was scheduled as an interdisciplinary approach.
After right transverse and combined median laparotomy during which the driveline was identified, exploration of the abdominal cavity ruled out extrahepatic tumour spread. An intraoperative ultrasound confirmed local resectability.
LVAD-driveline was then dissected from scarring tissue, disconnected from its controller for 30 s and repositioned in the left upper quadrant. Circulation was maintained with the use of epinephrine and norepinephrine. Figure 2a and b show preoperative and postoperative driveline positions. Figure 2c shows relocated driveline position and former driveline exit.
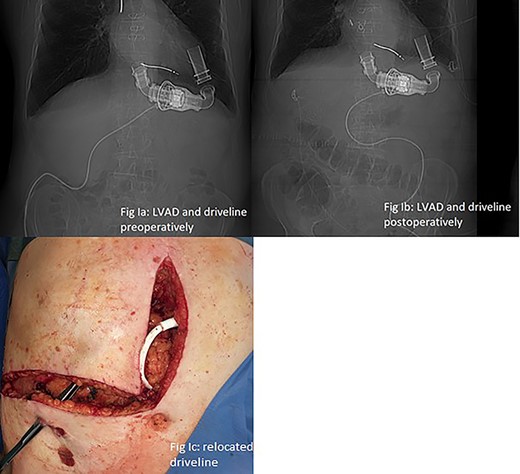
(a and b) LVAD and driveline in preoperative and postoperative CT scan scout. (c) Relocated driveline during laparotomy, forceps in former driveline exit.
Liver resection was performed via ‘pedicle approach’. After mobilization of the right hepatic lobe with posterior transection of Makuuchis ligament and preparation of the right hepatic vein, the vein was encircled with a loop and hilar preparation followed: after lowering the hilar plate, the right and left glissonean pedicle were identified. Right pedicle was followed until its branching into antero-medial and postero-lateral pedicles. Safety clamping of the posterolateral branch with sonography of the antero-medial branch was performed leading to a clear demarcation of the now ‘in-flow’ devascularized postero-lateral sector. Parenchymal transection was performed using CUSA™ (Cavitron Ultrasonic Surgical Aspirator, Valleylab Boulder, CO, USA) starting at the right hepatic vein and continuing alongside the border of segments VII/VI and VIII/V. Crossing structures were clipped or ligated with titan clips and 3–0 or 4–0 Vicryl ™. Pringle manoeuvre was used twice for a maximum of 15 min, interrupted by at least 5 min reperfusion. Finally, only the posterolateral pedicle and the right liver vein remained and were selectively clamped, dissected and oversewed (Fig. 3a).
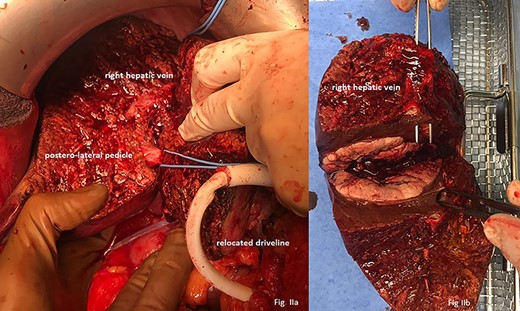
(a) Liver after parenchymal dissection showing the relocated driveline, the postero-lateral pedicle and the right hepatic vein. (b) Macroscopic view on tumour with forceps in right liver vein.
After rinsing and meticulous control for bleeding and bile leaks, drains were placed and an omental flap was prophylactically attached ibidem. The retrieved specimen is shown in Fig. 3b.
Postoperatively, the patient was admitted to the cardiosurgical intensive care unit. LVAD flow was over 5 l/min and no further use of vasopressors was necessary. After on POD 6 the former drainage exit started purulent secretion and a computed tomography (CT) scan revealed a small bilioma, an ERCP was performed. A bile leakage type B (ISGLS) was diagnosed and treated by stenting of the common bile duct. Concomitantly, a transient acute on chronic kidney insufficiency (AKIN °I) was diagnosed and conservatively managed. The patient was transferred to our surgical ward on POD 10 without signs of systemic infection. A superficial surgical site infection underwent conservative treatment. In postoperative quality management analysis the case was classified Clavien-Dindo °IIIa and SSI °I. Both liver and LVAD function remained satisfactory. Oral anticoagulation was reestablished and the patient was discharged on POD 19. Histopathology confirmed the diagnosis of HCC (pT4 G2 R0 L0 V0 Pn0 M0, 8th TNM classification).
Six weeks after surgery the bile duct stent was removed without complications.
The case was discussed in our interdisciplinary Hepato-Pancreato-Biliary (HPB) tumour conference and regular oncologic surveillance was recommended. After 6 months, the patient presented a good general condition and imaging showed no sign of local or distant recurrence.
DISCUSSION
Conditional on initial survival, long-term survival after LVAD implantation is possible [3]. This leads to more patients concomitantly requiring abdominal tumour surgery. HCC is a serious life threat and since the patient was in good general condition, an interdisciplinary approach was made to evaluate risk factors. To the best of our knowledge, there has no case of complex major liver surgery been reported in these patients previously. Elective abdominal surgery for patients with LVAD requires a specialized team and sufficient preoperative preparation. Relocation of the driveline may become necessary. Lower abdominal surgery requiring an infraumbilical median laparotomy often does not interfere with the driveline [4]. Exposure of the surgical site often requires retractors, which may interfere with the LVAD cannulas [5]. LVAD furthermore contributes a pulseless blood flow and microperfusion may be affected. Furthermore, continuous flow instead of pulsatile flow was described to harden the identification and palpation of vascular structures [5]. In our case, vascular structures were identified by subtle preparation (extrafascial pedicle and combined venous first approach) and confirmed by the use of intraoperative ultrasound.
At our centre the usual approach to postero-lateral sectionectomy is minimal invasive (laparoscopic or robotic-assisted). In the presented case we decided to perform an open approach, because, due to the driveline position, an emergency conversion might not have been possible. Regardless of choosing laparotomy or laparoscopy, awareness of the driveline-position is essential. Meticulous preoperative evaluation of comorbidities and treatment strategies is mandatory to tailor an individualized strategy.
CONCLUSION
In patients with LVAD and concomitant liver malignoma adequately prepared and scheduled surgery with interdisciplinary management may be a feasible treatment option. These patients should be presented to experienced centres’ HPB tumour boards rather than upfront indicating palliative therapy.
CONFLICT OF INTEREST STATEMENT
All authors declare no commercial associations that might represent conflicts of interest with this article and have nothing to disclose.
FUNDING
None.
AUTHOR CONTRIBUTIONS
All authors read and approved the submitted manuscript.



