-
PDF
- Split View
-
Views
-
Cite
Cite
James J Yahaya, Bartholin’s gland hyperplasia with dysplastic changes: a rare case report, Journal of Surgical Case Reports, Volume 2020, Issue 9, September 2020, rjaa312, https://doi.org/10.1093/jscr/rjaa312
Close - Share Icon Share
Abstract
The purpose of this paper is to report unusual, rarest and interesting case of a patient with nodular hyperplasia of Bartholin's gland with dysplastic changes. The case of a 30-year old female with right-sided Bartholin’s gland hyperplasia with dysplastic changes, which was confirmed histopathologically, is presented in this paper. The patient reported increased swelling of the right major labium when she became sexually aroused with intermittent dyspareunia during intercourse. Surgical excision of the swelling was done under general anaesthesia. Healing of the surgical scar was complete and she reported no any discomfort or dyspareunia during sexual intercourse. Bartholin’s gland swellings with firm consistency require complete excision due to possibility of being neoplastic rather than just inflammation, cyst or obstructed gland particularly in postmenopausal women. Diagnosis of nodular hyperplasia of Bartholin’s gland with areas of dysplastic changes in the present case is of great interest and requires further investigation.
INTRODUCTION
Tumours occurring in the site of Bartholin’s gland are rare and most of them are underreported. The mostly reported lesions in this site are cysts, inflammation and obstruction to the gland [1, 2]. Benign tumours affecting the Bartholin’s gland are adenoma (AD) and adenomyoma (AM), these usually occur less commonly than Bartholin’s glands nodular hyperplasia [3]. Occurrence of malignant tumours in the Bartholin’s gland is extremely rare and the most malignant tumours are adenocarcinoma and squamous cell carcinoma. These carcinomas have been thought to develop by the process of adenoma-carcinoma transformation from clonal expansion of transformed cells with malignant characteristics [3, 4]. Adenocarcinoma and squamous cell carcinoma account for 80–90% of primary cases and the remaining 10–20% of cases include transitional, adenoid-cystic or undifferentiated carcinomas [5]. This paper reports the case of unreported case of the patient with nodular hyperplasia of the Bartholin’s gland consisting of a focus with dysplastic changes.
CASE REPORT
A 30-year-old female presented with a 6-month history of a progressive painless swelling on the right vaginal wall. She reported that the swelling would increase in size when she was sexually aroused with intermittent dyspareunia during intercourse. On physical examination, she had a nodular swelling on the right wall of the vagina involving the upper part, which measured 2.1 × 9.0 cm with intact overlying mucosal surface. The swelling was painless on palpation, firm in consistency and mobile with regular margins. Bartholin’s cyst was considered as a clinical diagnosis. She underwent excisional removal of the swelling under general anaesthesia. Her postoperative period was uneventful.
Microscopic examination of the excisional biopsy materials showed a lesion composed of proliferating acinar (Fig. 1) with lining epithelium composed of columnar to cuboidal mucin-secreting cells showing no atypia (Fig. 2). The lesion was divided by fibrous septae to form lobules (Fig. 3). There was a focal area that was showing element of moderate dysplastic changes with element of obliterating tubules, increased hyperchromasia and moderate pleomorphism (Fig. 4). The ratio of duct-to-acini was maintained. Sparse chronic inflammatory infiltrate composed of mainly lymphocytes was present. Neither mitoses nor necrosis were present. The surgical margins were free of the lesion. This confirmed the histopathological diagnosis of Bartholin’s gland hyperplasia with dysplastic changes. After a period of 1 month she was called for clinical evaluation. Healing of the surgical scar was complete and she reported no discomfort or dyspareunia during sexual intercourse.
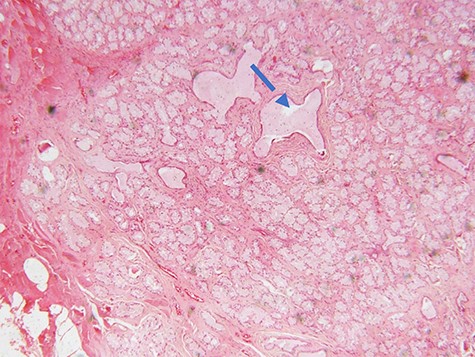
photomicrograph of haematoxylin and eosin (H and E) stained tissue section showing a lobulated mass composed of proliferating acini and some cystically dilated tubules with inspissation of pinkish proteinaceous secretions (arrow) (×40 magnification)
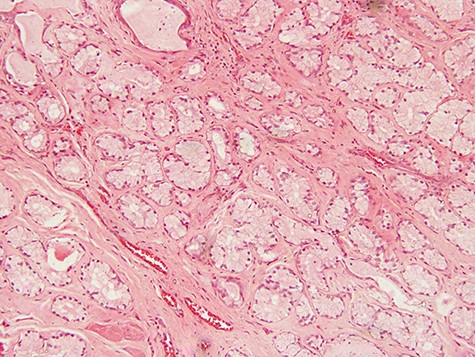
photomicrograph of haematoxylin and eosin (H and E) stained tissue section showing a lesion divided into lobules by fibrous septae in which proliferating and congested blood vessels are seen. The typical columnar to cuboidal mucin-producing epithelial cells are basally located with abundant pale cytoplasm (arrow) (×100 magnification)
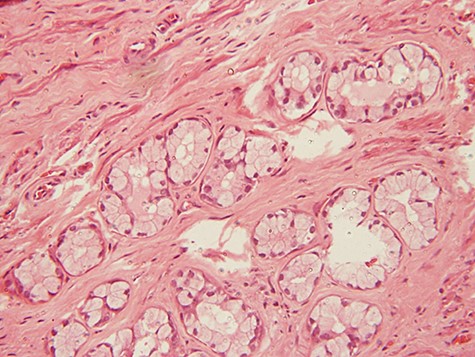
photomicrograph of haematoxylin and eosin (H and E) stained tissue section showing a lesion divided into lobules by fibrous septae. The typical columnar to cuboidal mucin-producing epithelial cells are basally located with abundant pale cytoplasm (arrow) (×200 magnification)
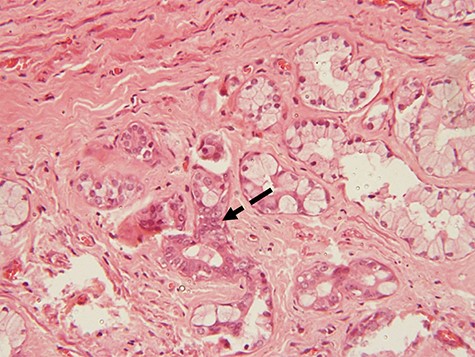
photomicrograph of haematoxylin and eosin (H and E) stained tissue section showing a focus of dysplastic changes. The areas with dysplastic changes show proliferating hyperchromatic and moderately pleomorphic cells causing obliteration of the lumen of the tubules (dotted arrow) (×200 magnification)
DISCUSSION
Clinically patients with nodular hyperplasia (NH) of the Bartholin’s gland have been reported to have no specific presentations. Nonetheless, most of case reports and case series reported in the literature have shown that, patients with NH present with painless swellings; sometimes the swellings may be painful when involved with friction during sexual intercourse (dyspareunia), vulvar ulcer and abnormal inspissation of mucin into vaginal vestibulum during sexual arousal [1–3, 6]. The swellings are usually unilateral; however, bilateral cases are also quite common [2–4].
Studies have shown that the cause of NH is not clear; however, most of studies suggest that inflammation and obstruction to the Bartholin’s gland duct may be responsible for inducing the proliferation of acini, which is the hallmark microscopic picture of the disease entity [7]. There has been a debate on how adenomas can be histologically distinguished from NH. In 1998, Koenig and Tavassalo included 17 cases in their study in trying to distinguish adenoma from NH of the Bartholin’s gland and they concluded that, in NH, the distribution of duct-to-acinar ratio is normal and the acini have a single layer of mucin-secreting cells composed of basally located columnar or cuboidal epithelial lining with sparse lymphocytic infiltrate in the fibrous stroma. On the other hand, in adenomas the ducts are not seen and the acini tend to have two cell layers (basal and columnar or cuboidal cells) without traces of inflammatory cells in the stroma [3]. Additionally, the lesion in NH is lobulated and well delineated from the adjacent normal stroma whereas in adenomas the lesion is not lobulated and has irregular borders [3].
Adenomas of the Bartholin’s gland are neoplastic and are thought to transform into carcinomas as a result of clonal expansion following mutation. This can be evidenced by P53 expression in adenomatous lesions and not in non-adenomatous lesions [4, 8]. The risk of adenomas to transform into cancer is reported in postmenopausal women unlike in premenopausal and adolescent women [2, 9].
Management of Bartholin’s gland cystic lesions requires marsupialization and fistulization with Word catheterization [1, 4]. NH and other neoplastic lesions (adenomas and adenomyomas) are managed surgically by excision. There are two major challenges related with management of such lesions as it was reported by Burger et al [4]. Incomplete excision is a frequent experience. Partly the problem may be due to the fact that, the Bartholin’s gland has no capsule, rendering it difficult to find the surgical dissection plane. For example, Santos et al. [10] reported the highest rate of relapse of 80% in patients with NH. Another reason may be that, sometimes the enlarged gland is deeply situated in the stroma and laterally of the labiocrural fold making vestibular incision of the gland approach not possible to achieve complete excision of the lesion; therefore, a transcutaneous incision alongside the labiocrural fold would help to ensure complete excision of the enlarged gland [4].
When managing patients with NH, the enlarged gland may be missed because either the swelling is located deep in the perineum for example, 2–3 cm lateral and inferior to the fourchette or because the clinician attending the patient is unfamiliar with enlarged glands and it may be misinterpreted as a ‘deeply situated’ Bartholin's cyst. [4]. Therefore, to overcome such challenges, Matthe et al. advised that, during palpation of the swelling, both the intravaginal index finger and the opposite external thumb must make contact with the bony pelvis before being moved medially and posteriorly [4].
In conclusion, the presence of dysplastic changes in the case of NH of Bartholin’s gland as it was in the present case is an intriguing subject due to the reason that, NH is non-neoplastic; instead it is merely a proliferative process of the acini.
ETHICAL APPROVAL
Not needed.
INFORMED CONSENT
Written informed consent was obtained from the patient for the publication of the details related to the patient and a copy has been kept for review by the Editor-in-Chief of this journal when needed to do so.
CONFLICT OF INTEREST STATEMENT
The author of this work declares that he has no conflict of interest related to this work to be disclosed.
FUNDING
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
ACKNOWLEDGEMENTS
J.J.Y. thanks the team of gynaecologists at Mulago National Hospital for helping him to obtain the details of the case reported in this paper.



