-
PDF
- Split View
-
Views
-
Cite
Cite
Budoor Mohamed Al-Musaifer, Veena Nagaraj, Latifa Al-Buainain, Abdulla Darwish, Glycogen rich clear cell carcinoma of the breast: a rare subtype with good prognosis, Journal of Surgical Case Reports, Volume 2019, Issue 5, May 2019, rjz150, https://doi.org/10.1093/jscr/rjz150
Close - Share Icon Share
Abstract
Glycogen rich cell carcinoma (GRCC) is a rare subtype of primary malignant neoplasm of the breast. Less than 150 cases have been reported since its first description, thus, making its prognosis unclear and vary from one literature to another. Here we present a 5-year cancer free after the completion of chemotherapy, radiotherapy and targeted-therapy in a 55-year-old female patient with GRCC of the breast. The purpose of reporting this case is to increase the knowledge about this rare subtype of breast cancer and chance of better survival.
INTRODUCTION
Glycogen rich clear cell (GRCC) carcinoma is a rare subtype of breast cancer in which more than 90% of the neoplastic cells have abundant clear cytoplasm containing glycogen [1]. It presents most commonly in patients age from 41 to 78 years with median age of 57. GRCC of the breast make up 1.4–3% of all breast malignancies [1, 2]. Fewer cases have been described with GRCC since the first case was reported in 1981 by Hull et al. [3]
The clinical presentation usually like other invasive breast cancers. The tumor may range from 1 to 10 cm in size and can have either well circumscribed, irregular, intraductal papillary or intracystic appearance [4]. Despite few reported cases the prognosis for patients with GRCC is relatively unclear. However, tumor staging with hormones markers [progesterone receptor (PR) and estrogen receptor (ER)] and human epidermal growth factor receptor 2 (Her2) status play an important role in prognosis [5]. The present work reports a case of GRCC with axillary lymph node involvement with 5-year successful outcome.
THE CASE
A 55-year-old mother presented to the breast clinic with left breast lump for the past two weeks. Physical examination revealed a 3×2 cm solitary hard mass located at the left inner upper quadrant, with no palpable axillary lymph nodes. There was no skin changes or nipple discharge. Mammogram showed a 3.2 cm round high density mass in the inner upper quadrant of the left breast. No micro calcifications noted (Fig. 1). Ultrasound guided fine needle aspiration (FNAC) demonstrated features consistent with breast carcinoma.
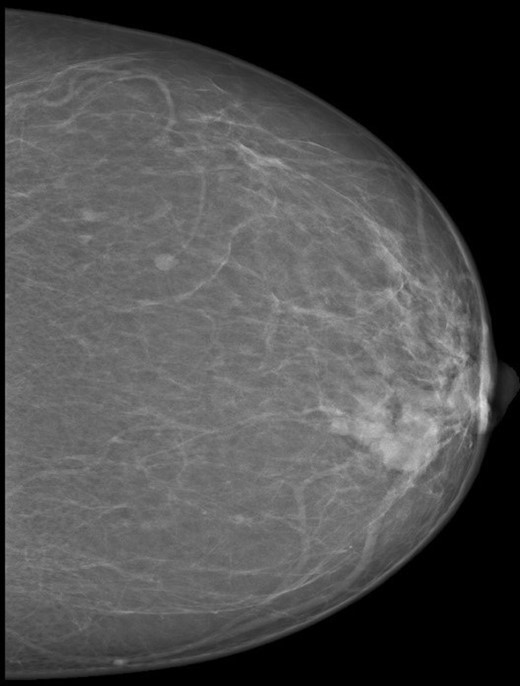
Mammogram picture of left breast showing a lobulated poorly define mass m 3 × 2 × 2.2 cm.
The breast lump was fully excised via a wide local excision along with axillary clearance. The pathological examination of the specimen showed a 3×2.2 cm firmly well circumscribed lesion with unremarkable surrounding fibrofatty tissue. The histological examination showed a Grade 3 invasive clear cell (glycogen rich) carcinoma composed of clear cells with pleomorphic nuclei (Fig. 2). The tumor cells showed intense positivity for Periodic Acid Schiff (PAS), diastase sensitive (Fig. 3A and B), confirming glycogen content in tumor cells. The immunohistochemistry profile showed many cells were positive for Her2 score 3 (Fig. 4), but negative for ER and PR. The only single sentinel lymph node showed tumor metastasis while the other eight retrieved lymph nodes were normal. Neither vascular invasion nor microcalcification noted. The possibility of metastasis was excluded by clinical and radiological investigations. The final pathological stage was pT2 (3 cm in maximum dimension) pN1a (1/9) pM0. The Nottingham prognostic index (NPI) score was 5.6. Post-operative recovery was uneventful.
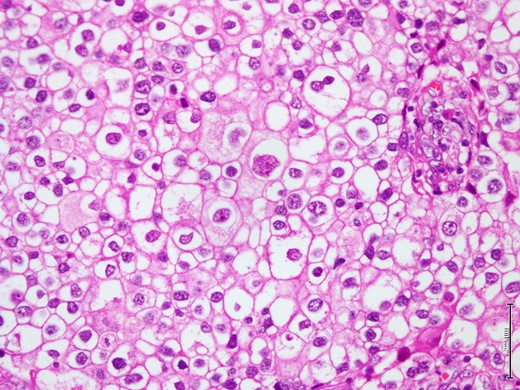
Shows microscopic appearance of the tumor composed of large polygonal cells with pleomorphic nuclei and well define nuclear membrane (H&E stain, 40X).
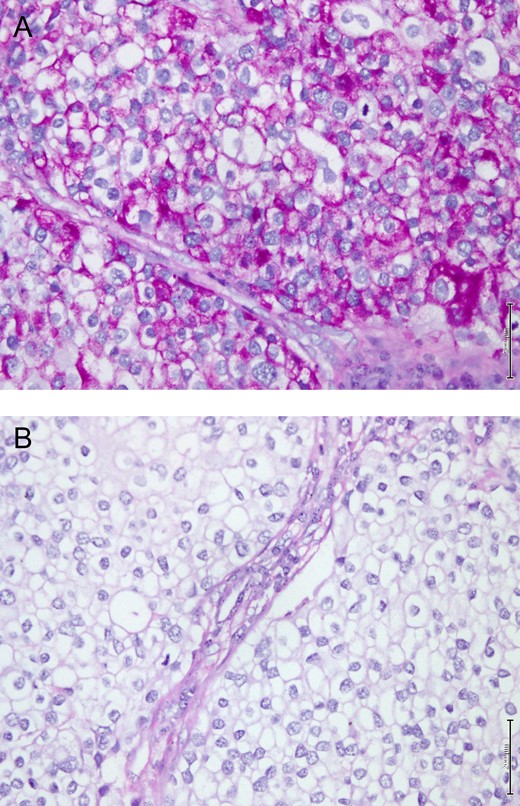
(A) Shows microscopic appearance of the tumor cells in GRCC of breast strongly positive for PAS stain. (B) sensitive for diastase digestion (PASD stain, 20X).
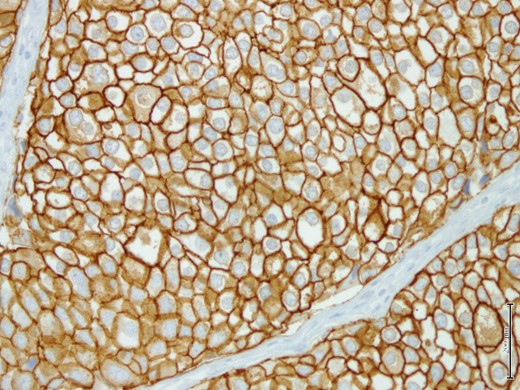
Tumor cells show strong positivity for Her2/neu immunohistochemistry marker.
The patient completed four cycles of chemotherapy (Doxorubicin 60 mg/m2 and Cyclophosphamide 600 mg/m2) followed by targeted-therapy (Trastuzumab /Herceptin) and radiotherapy (60 Gy). Regular five-year follow up showed no evidence of recurrence.
DISCUSSION
Clear cell carcinomas arise in many organs including lung, endometrium and kidney. The tumor cells may have either cytoplasmic glycogen or mucin that have an optically clear vacuolated appearances [6].
GRCC of the breast is a rare subtype of breast cancer in which the tumor cells contain abundant glycogen. These tumors in most of the patients usually present either as ductal carcinomas in situ (DCIS) or DCIS with invasive component [7]. Invasive lobular, tubular and medullary carcinomas have been also reported in GRCC in small number of patients [2]. The GRCC is a primary malignant neoplasm should be differentiated from other tumors which have similar appearances. Firstly, primary malignant tumors of the breast such as lipid rich carcinoma, signet ring cell carcinoma, and others. Secondly, from benign breast tumors with clear cell morphology such as clear cell hidradenoma and benign myoepithelioma. Finally, metastatic tumors from other organs like kidney, adrenal gland, ovary and lung with clear cell morphology should be excluded clinically, immunohistochemistry (IHC) markers and radiological investigations [4, 6, 8].
The present patient was 55 years old with paternal family history of breast cancer of unknown subtypes. The absence of primary neoplasm in other organs excluded metastasis from other sites and malignant histological features of this tumor exclude the list of primary benign tumors of the breast. The final histological diagnosis was concluded as primary breast carcinoma of clear cell (glycogen rich) type which was supported by the invasive growth behavior and tumor metastasis in the sentinel lymph node. Furthermore, the IHC study confirmed the diagnosis by excluding the possibility of metastasis from other organs malignancies.
In this case, both ER and PR were negative, but the tumor cells were strongly positive for Her2/neu (scored 3+). Kuroda et al., found no significant difference in the incidence of Her2/neu overexpression between GRCC and invasive carcinomas subtypes while the incidence of ER and PR positivity appear to be quit less than the other breast cancer subtypes [8]. However, other researchers opined no significant difference in hormone receptor status [4].
The majority of reported GRCC cases are considered to be highly aggressive associated with late detection and propensity for lymph node metastasis [4]. The number of positive lymph nodes are significantly related to the prognosis of GRCC. Patients with more than ten nodal micro metastases were at high risk, and usually present with recurrences and distant metastasis [9]. Toikkanen et al., suggest that GRCC cases are more aggressive than other common breast cancers [10]. However, Baslaim MM et al., concluded that in term of prognosis, GRCC are similar to other breast carcinomas [11]. The calculation score of NPI in this case equal to 5.6 indicating poor score. The predicted five and ten-year breast cancer-specific survival is 86% and 73% respectively [12]. In our opinion the prognosis of GRCC of breast depend on the stage matched basis if it compared with another invasive ductal carcinoma [5].
CONCLUSION
We describe a relatively rare subtype of breast carcinoma in clinical practice with a single positive axillary lymph node involvement and a good clinical outcome five-year after the completion of therapy. However, the prognosis of GRCC of the breast remains unclear due to is rarity and limited research. Therefore, further research is required to understand its prognosis and effective management.
ACKNOWLEDGMENTS
None.
FUNDING
No funding provided for this study.
CONFLICTS OF INTEREST STATEMENT
The authors have received no internal or external funding for this report. There are no financial disclosures. No potential conflicts of interest are declared. The patient gave her written consent for this report.



