-
PDF
- Split View
-
Views
-
Cite
Cite
Vincent Demesmaker, Faouzi Abou-Messaoud, Muriel Parent, Bernard Vanhoute, Fadi Maassarani, Konstantinos Kothonidis, Pancreatic solid serous cystadenoma: a rare entity that can lead to a futile surgery, Journal of Surgical Case Reports, Volume 2019, Issue 12, December 2019, rjz360, https://doi.org/10.1093/jscr/rjz360
Close - Share Icon Share
Abstract
Although many types of pancreatic tumors exist, pancreatic solid serous cystadenoma stand as the most rare. Despite advances in medical imaging, definitive diagnosis remains complex. Here, we report a case of a 63-year-old man with a suspicious lesion of the pancreas discovered during a positron emission tomography–computed tomography. Despite an echo-endoscopy being performed, no biopsies were contributive. The magnetic resonance imaging did highlight another lesion in the liver. Due to the suspicion of a neuroendocrine tumor of the pancreas with liver metastases, a cephalic duodenopancreatectomy with partial hepatectomy was performed. During the procedure, another hepatic lesion was resected. The final histological diagnosis was a serous solid adenoma of the pancreas associated with a cholangiocarcinoma. During the follow-up, the patient presented a massive metastatic hepatic relapse even after the administration of a palliative chemotherapy.
INTRODUCTION
Cystic tumors of the pancreas are very rare entities, accounting for 1–2% of exocrine pancreatic tumors [1].
These cystic tumors are divided into two categories (serous and mucinous), and within the serous forms there are five subcategories [2]. The serous solid adenoma is the rarest entity of these five subcategories. The first case was described in 1996 by Perez-Ordonez et al. [3].
Cytological, immunological and histological characteristics are the same as those found in other cystic tumors of the pancreas [4].
Its rarity, as well as the difficulty of differential diagnosis with pancreatic solid tumors, makes the preoperative diagnosis complex. The absence of malignant behavior imposes an accurate diagnosis. Therefore, a monitoring or a simple excision, when feasible, is often adequate [5].
CASE REPORT
A 63-year-old man with a medical history of dysplastic colic polyps consults his gastroenterologist for weight loss. An assessment and a positron emission tomography–computed tomography (PET-CT) are performed.
In the patient’s history, we note a seminal tumor treated by radiotherapy, a thyroidectomy, a resection of sigmoid polyps, and Crohn’s disease.
The PET-CT carried out 2 days later reveals a suspicious hypervascular cephalo-isthmic lesion of the pancreas, without 18F-fluorodeoxyglucose fixation.
In order to specify the nature of this tumor, an echo-endoscopy is performed. This exam confirms the presence of a 2.5 mm suspicious isthmic tumor, hypoechoïc and heterogeneous, well delineated. We also note the compressive nature on the main pancreatic duct. No vascular invasion and no suspicious lymphadenopathy are observed. All of the biopsies performed are negative.
A CT scan later highlights the 25 mm lesion and the dilation of the Wirsung canal (Fig. 1). The contrast enhancement does not support the diagnosis of adenocarcinoma. Considering these features, the differential diagnosis with a neuroendocrine tumor (NET) must be made. No pathological somatostatin receptor fixation is observed during octreoscan.
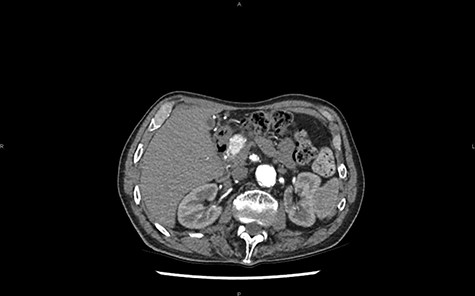
CT in axial section, arterial phase: visualization of a hypervascular structure at the level of the pancreas head.
Magnetic resonance imaging (MRI) confirms the hypervascularized and atypical character of this pancreatic tumor (Figs 2 and 3). Moreover, one hepatic lesion with the same characteristics is highlighted.
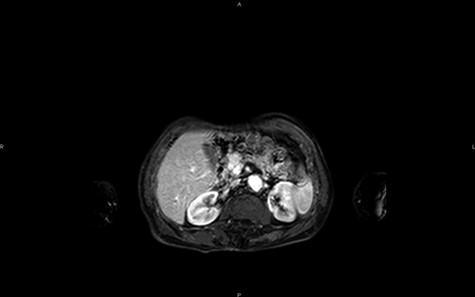
MRI in axial section, phase T1 + gadolinium: visualization of a structure with hypersignal at the level of the head of the pancreas.
The final diagnostic assumption is an NET of the pancreas with a single metastatic hepatic lesion (segment II). During surgical exploration another hepatic lesion is found (segment V). As a result, a cephalic duodenopancreatectomy is performed with a resection of the two hepatic lesions.
The histopathological analysis of the two hepatic tumors shows a moderately differentiated cholangiocarcinoma without vascular or perineural infiltration. For the pancreatic lesion, the diagnosis of serous solid adenoma is suggested. The immunostaining of this tumor is CK7, CK18+, KI67 < 3%, chromogranin, synaptophysin, PS 100, CK20.
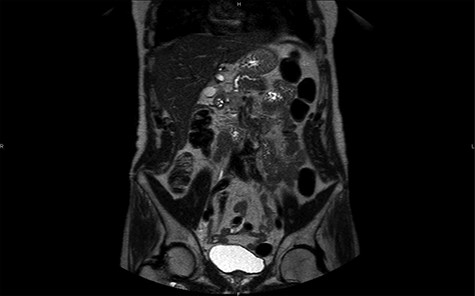
MRI in coronal section, T2 phase: visualization of a compression of the main pancreatic duct.
The conclusive stage of bilobar cholangiocarcinoma is TNM stage II (pT2BN0M0). In multidisciplinary oncologic council, no adjuvant treatment is proposed. Unfortunately, a massive relapse of multifocal cholangiocarcinoma occurs 3 months later. A palliative chemotherapy is decided upon, and the patient dies one and a half years later.
DISCUSSION
As discussed earlier, the cystic tumors of the pancreas can be subdivided into serous and mucinous forms. Mucinous forms often have a higher malignancy potential than serous forms [6].
Table 1 contains the clinical characteristics of the 21 cases previously described, in addition of this case. According to this analysis, this tumor occurs most frequently in women (60%) with an average age of 61 years. Different locations are possible without any real predominance: body (36%), head (27%), or tail of the pancreas (18%). The diagnosis is most often made accidentally (45%) or during examinations performed as a result of abdominal pain (40%).
| N° . | Author . | Year . | Age . | Sex . | Location . | Symptoms . | Tumor size (cm) . | Preoperative diagnosis . | Operative procedure . | Outcome/follow . | Capsule . | C-MPD . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Perez-Ordonez | 1996 | 70 | F | Tail | Abdominal plain | 4 | NET | DPS | Alive 5 years | − | − |
| 2 | Hayashi | 2001 | 57 | F | head | Incidental | 2,1 | / | / | / | + | − |
| 3 | Kosmahl | 2004 | 50 | M | Head | incidental | 2,5 | PDA | / | / | / | / |
| 4 | Yamamoto | 2004 | 60 | M | Uncus | Epigastric pain | 2 | NET | Enucleation | / | + | / |
| 5 | Gabata | 2005 | 59 | F | Body | Abdominal pain | 2 | NET-SSCA | DP | / | / | / |
| 6 | Matsumoto | 2006 | 39 | F | Body | Incidental | 3,4 | NET | Enucleation | Alive 1,5 year | + | − |
| 7 | Yamaguchi | 2006 | 58 | F | Body | Incidental | 2 | Malignant NET | DP | Alive 1 year | + | + |
| 8 | Reese | 2006 | 66 | M | Head | Incidental | 3,5 | NET | PPPD | / | / | / |
| 9 | Stern | 2007 | 62 | M | Head/body | Abdominal pain | 4,2 | NET, PDA, SPPT and metastasis | DPS | / | − | / |
| 10 | Sanaka | 2007 | 74 | M | Body | Incidental | 1,6 | NET | Enucleation + YPJ | Alive 2 months | + | / |
| 11 | Casadei | 2008 | 59 | F | Tail | Abdominal pain | 4 | NET, SPPT and metastasis | DPS | / | + | / |
| 12 | Hayashi | 2009 | 58 | F | / | / | 3,2 | / | / | / | + | / |
| 13 | Yasuda | 2011 | 72 | F | Head | Incidental | 1,7 | NET | PPPD | / | + | / |
| 14 | Hayashi | 2012 | 74 | F | body | / | 4.2 | / | / | / | + | / |
| 15 | Lee | 2013 | 56 | M | Tail | Incidental | 2,5 | NET | Laparoscopic DP | Alive 1 year | + | / |
| 16 | Kishida | 2014 | 58 | M | Body | Incidental | 2,2 | NET | DP | Alive 2 years | + | / |
| 17 | Wu | 2015 | 48 | M | Head | abdominal pain | 2,2 | NET | PPPD | Alive 2 years | + | / |
| 18 | Wu | 2015 | 65 | F | Body | Incidental | 1,8 | NET | Laparoscopic DP | / | + | / |
| 19 | Geramizadeh | 2015 | 68 | F | Head | Abdominal pain | 3 | NET | CDP | Alive 3 months | + | − |
| 20 | Katsourakis | 2016 | 72 | F | Tail | Abdominal pain | 3 | NET | DP | / | + | − |
| 21 | Hamid | 2017 | 53 | F | Body | Abdominal pain | 3,5 | NET, SPPT | Laparoscopic DPS | Alive 9 months | + | / |
| 22 | Current case | 2019 | 63 | M | Isthmus | Weight loss | 2,8 | NET | CDP | Death 1,5 year (cholangiocarcinoma) | − | + |
| N° | Author | Year | Age | Sex | Location | Symptoms | Tumor size (cm) | Preoperative diagnosis | Operative procedure | Outcome/follow | Capsule | C-MPD |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Perez-Ordonez | 1996 | 70 | F | Tail | Abdominal plain | 4 | NET | DPS | Alive 5 years | − | − |
| 2 | Hayashi | 2001 | 57 | F | head | Incidental | 2,1 | / | / | / | + | − |
| 3 | Kosmahl | 2004 | 50 | M | Head | incidental | 2,5 | PDA | / | / | / | / |
| 4 | Yamamoto | 2004 | 60 | M | Uncus | Epigastric pain | 2 | NET | Enucleation | / | + | / |
| 5 | Gabata | 2005 | 59 | F | Body | Abdominal pain | 2 | NET-SSCA | DP | / | / | / |
| 6 | Matsumoto | 2006 | 39 | F | Body | Incidental | 3,4 | NET | Enucleation | Alive 1,5 year | + | − |
| 7 | Yamaguchi | 2006 | 58 | F | Body | Incidental | 2 | Malignant NET | DP | Alive 1 year | + | + |
| 8 | Reese | 2006 | 66 | M | Head | Incidental | 3,5 | NET | PPPD | / | / | / |
| 9 | Stern | 2007 | 62 | M | Head/body | Abdominal pain | 4,2 | NET, PDA, SPPT and metastasis | DPS | / | − | / |
| 10 | Sanaka | 2007 | 74 | M | Body | Incidental | 1,6 | NET | Enucleation + YPJ | Alive 2 months | + | / |
| 11 | Casadei | 2008 | 59 | F | Tail | Abdominal pain | 4 | NET, SPPT and metastasis | DPS | / | + | / |
| 12 | Hayashi | 2009 | 58 | F | / | / | 3,2 | / | / | / | + | / |
| 13 | Yasuda | 2011 | 72 | F | Head | Incidental | 1,7 | NET | PPPD | / | + | / |
| 14 | Hayashi | 2012 | 74 | F | body | / | 4.2 | / | / | / | + | / |
| 15 | Lee | 2013 | 56 | M | Tail | Incidental | 2,5 | NET | Laparoscopic DP | Alive 1 year | + | / |
| 16 | Kishida | 2014 | 58 | M | Body | Incidental | 2,2 | NET | DP | Alive 2 years | + | / |
| 17 | Wu | 2015 | 48 | M | Head | abdominal pain | 2,2 | NET | PPPD | Alive 2 years | + | / |
| 18 | Wu | 2015 | 65 | F | Body | Incidental | 1,8 | NET | Laparoscopic DP | / | + | / |
| 19 | Geramizadeh | 2015 | 68 | F | Head | Abdominal pain | 3 | NET | CDP | Alive 3 months | + | − |
| 20 | Katsourakis | 2016 | 72 | F | Tail | Abdominal pain | 3 | NET | DP | / | + | − |
| 21 | Hamid | 2017 | 53 | F | Body | Abdominal pain | 3,5 | NET, SPPT | Laparoscopic DPS | Alive 9 months | + | / |
| 22 | Current case | 2019 | 63 | M | Isthmus | Weight loss | 2,8 | NET | CDP | Death 1,5 year (cholangiocarcinoma) | − | + |
PDA: pancreatic ductal adenocarcinoma, SSCA: solid serous cystadenoma of the pancreas, SPPT: solid pseudopapillary tumors, DPS: distal pancreatectomy with splenectomy, DP: distal pancreatectomy (without splenectomy), PPPD: pylorus-preserving pancreatoduodectomy, YPJ: Roux-en-Y pancreaticojejunostomy, CDP: Cephalic duodenopancreatectomy.
Microscopically, the serous solid adenoma has the same architectural and cytological characteristics as the serous microcystic cystadenoma, namely, a round or ovoid structure formed of cuboid cells derived from the ductular epithelium. The cytoplasm is rich in glycogen with a small central nucleus [7]. The prevalent difference being that the solid adenoma has no secretory activity, giving a more compact structure the main difference is the absence of secretory activity, which confers a more compact structure to this lesion compared to cystic forms. According to the analysis of the different cases, the average size is 2.7 cm among the 22 cases previously described. We also note that a capsule is found in 73% of cases.
Concerning immunostaining, there are similar characteristics with the cystic forms, such as the presence of markers: cytokeratins, α-inhibin, mucin-1 (MUC-1), and mucin-6 (MUC-6) [8]. Table 2 shows the varying markers identified in the 21 cases described previously as well as in the case report before.
Different immunomarkers found in the 22 described cases of pancreatic solid serous cystadenoma
| . | 1 . | 2 . | 3 . | 4 . | 5 . | 6 . | 7 . | 8 . | 9 . | 10 . | 11 . | 12 . | 13 . | 14 . | 15 . | 16 . | 17 . | 18 . | 19 . | 20 . | 21 . | 22 . | % + . | % − . |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CK7 | / | / | / | / | / | 1 | 1 | 1 | 1 | / | / | / | / | / | 1 | / | 1 | 1 | 1 | 1 | / | 1 | 45% | 0% |
| CK19 | / | / | / | / | / | 1 | / | / | 1 | / | / | / | / | / | 1 | / | 1 | 1 | 1 | 0 | / | / | 27% | 4,5% |
| NSE | 1 | / | 1 | / | / | / | / | 1 | 1 | / | / | / | / | / | / | / | / | / | / | 1 | / | / | 23% | 0% |
| AE1/AE3 | / | / | / | / | / | 1 | 1 | / | / | / | / | / | / | / | / | / | 1 | 1 | 1 | / | / | / | 23% | 0% |
| CAM 5.2 | 1 | / | / | / | / | 1 | 1 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | 18% | 0% |
| MUC1 | 1 | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 18% | 0% |
| CK18 | / | / | 1 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | / | 1 | 18% | 0% |
| α1-Antitrypsin | 1 | / | 0 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 14% | 9% |
| Alpha-inhibin | / | / | 1 | / | / | / | / | / | 1 | / | / | / | / | / | / | / | 1 | 0 | / | / | / | / | 14% | 4.5% |
| MUC6 | / | / | 1 | / | / | / | / | 1 | / | / | / | / | / | / | 1 | / | / | / | / | / | / | / | 14% | 0% |
| CK8 | / | / | 1 | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | 1 | / | / | 14% | 0% |
| Vimentin | / | / | 0 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 1 | 1 | / | 0 | / | / | 9% | 14% |
| a-1-Antichymotrypsin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 9% | 0% |
| B-Catenin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 9% | 0% |
| Calponin | / | / | / | / | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | 1 | / | 9% | 0% |
| LU5 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 4.5% | 0 |
| MA902 | / | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 4.5% | 0% |
| Synaptophysin | / | / | 0 | / | / | / | / | / | 0 | 0 | / | / | / | / | 0 | / | 0 | 0 | 0 | / | / | 0 | 0% | 36% |
| Chromogranin | / | / | 0 | / | / | / | 0 | / | / | 0 | / | / | / | / | / | / | 0 | 0 | 0 | / | / | 0 | 0% | 31% |
| CEA | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Insulin | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Glucagon | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Serotonin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| Gastrin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| Calcitonin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| CD56 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| CD10 | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | 0 | 0 | / | / | / | 0% | 14% |
| Progesterone R | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | / | / | / | / | 0% | 14% |
| CK20 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0% | 9% |
| MUC2 | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| MUC5 | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK5/6 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK10 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK14 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK17 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| PS-100 | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0% | 9% |
| RCC | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% | |
| SMA | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| MA903 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| Estrogen R | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| Calretinin | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| TH | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 | 22 | % + | % − | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CK7 | / | / | / | / | / | 1 | 1 | 1 | 1 | / | / | / | / | / | 1 | / | 1 | 1 | 1 | 1 | / | 1 | 45% | 0% |
| CK19 | / | / | / | / | / | 1 | / | / | 1 | / | / | / | / | / | 1 | / | 1 | 1 | 1 | 0 | / | / | 27% | 4,5% |
| NSE | 1 | / | 1 | / | / | / | / | 1 | 1 | / | / | / | / | / | / | / | / | / | / | 1 | / | / | 23% | 0% |
| AE1/AE3 | / | / | / | / | / | 1 | 1 | / | / | / | / | / | / | / | / | / | 1 | 1 | 1 | / | / | / | 23% | 0% |
| CAM 5.2 | 1 | / | / | / | / | 1 | 1 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | 18% | 0% |
| MUC1 | 1 | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 18% | 0% |
| CK18 | / | / | 1 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | / | 1 | 18% | 0% |
| α1-Antitrypsin | 1 | / | 0 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 14% | 9% |
| Alpha-inhibin | / | / | 1 | / | / | / | / | / | 1 | / | / | / | / | / | / | / | 1 | 0 | / | / | / | / | 14% | 4.5% |
| MUC6 | / | / | 1 | / | / | / | / | 1 | / | / | / | / | / | / | 1 | / | / | / | / | / | / | / | 14% | 0% |
| CK8 | / | / | 1 | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | 1 | / | / | 14% | 0% |
| Vimentin | / | / | 0 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 1 | 1 | / | 0 | / | / | 9% | 14% |
| a-1-Antichymotrypsin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 9% | 0% |
| B-Catenin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 1 | 1 | / | / | / | / | 9% | 0% |
| Calponin | / | / | / | / | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | 1 | / | 9% | 0% |
| LU5 | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 4.5% | 0 |
| MA902 | / | / | / | / | / | 1 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 4.5% | 0% |
| Synaptophysin | / | / | 0 | / | / | / | / | / | 0 | 0 | / | / | / | / | 0 | / | 0 | 0 | 0 | / | / | 0 | 0% | 36% |
| Chromogranin | / | / | 0 | / | / | / | 0 | / | / | 0 | / | / | / | / | / | / | 0 | 0 | 0 | / | / | 0 | 0% | 31% |
| CEA | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Insulin | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Glucagon | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 18% |
| Serotonin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| Gastrin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| Calcitonin | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| CD56 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | 0 | / | / | / | 0% | 14% |
| CD10 | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | 0 | 0 | / | / | / | 0% | 14% |
| Progesterone R | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0 | / | / | / | / | 0% | 14% |
| CK20 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0% | 9% |
| MUC2 | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| MUC5 | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK5/6 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK10 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK14 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| CK17 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| PS-100 | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | 0 | 0% | 9% |
| RCC | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% | |
| SMA | / | / | / | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| MA903 | / | / | / | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| Estrogen R | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| Calretinin | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
| TH | / | / | 0 | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | / | 0% | 4.5% |
CK: cytokeratin, NSE: neuron-specific enolase, AE1/AE2: human anion exchanger 1 and 2, CAM 5.2: anti-cytokeratin, MUC: mucin, LU5: pan cytokeratin, MA: antibodies specific for cytokeratin, CEA: carcinoembryonic antigen, PS 100: 100 protein, RCC: renal cell carcinoma marker, SMA: smooth muscle actin, Estrogen R: estrogen receptor, TH: thyrosine hydroxylase, CD: cluster of differentiation, Progesterone R: progesterone receptor.
The diagnosis of solid serous cystadenoma is complex because of its solid macroscopic appearance. This aspect requires a differential diagnosis between this benign entity and other solid tumors with poorer prognosis (NET, ductal adenocarcinoma, etc.). Unlike malignant tumors of the head, which tend to cause repression or invasion of adjacent structures, compression of the main pancreatic pathway is found in only 10% of cases.
The most often described aspect on the CT is a tumor that enhances rapidly in the early phase with rapid washout. Unfortunately, this characteristic is often found in NET [9], meaning that in most cases CT or MRI is not useful for differential diagnosis. From the analysis of the 22 cases described, examination of CT images advanced the diagnosis of NET in 63% of the cases.
More invasive tests such as endoscopic ultrasound (EUS) are not always contributive. Nevertheless, if sampling is feasible, negative immunostaining for chromogranin and synaptophysin can help to rule out the diagnosis of a NET [8]. In our case, despite two fine needle biopsies and one core biopsy, the EUS failed to give the diagnosis.
The absence of a diagnosis leads to an aggressive surgical management of these tumors, whereas minimal treatment can be performed [10]. If we refer to the procedures performed on the 22 patients suffering from this pathology, we find distal pancreatectomy with (27.3%) or without (18.2%) splenectomy, pylorus-preserving pancreatoduodenectomy (13.6%), cephalic duodenopancreatectomy (9.1%), and enucleation (13.6%). In our case, the duodenopancreatectomy was performed not only for oncologic reasons (lymphadenectomy) but also due to tumor placement (pancreatic head with main pancreatic duct compression.
Conflict of interest statement
None declared.
REFERENCES
- magnetic resonance imaging
- biopsy
- cholangiocarcinoma
- adenoma
- cystadenoma, serous
- neuroendocrine tumors
- pancreatic neoplasms
- pancreaticoduodenectomy
- surgical procedures, operative
- diagnosis
- neoplasms
- pancreas
- computed tomography/positron emission tomography imaging
- endoscopic ultrasound
- hepatic lesions
- palliative chemotherapy
- cephalic



