-
PDF
- Split View
-
Views
-
Cite
Cite
Luis Osvaldo Suárez Carreón, José Maria Zepeda Torres, Danya Cecilia Muñoz Balderas, Jose Abraham Flores Cardoza, José Víctor Pérez Navarro, Martin Islas Torres, Isac Ismael Ramírez Preciado, Rodrigo Hernández Ramírez, Carlos Francisco Gallegos De Luna, Brian Alejandro Díaz Sandoval, Kevin Kleiman Andrade, Nahomi Sharon Siordia Cruz, Alondra De la Cruz Rodríguez, Aida Estefanía Pelayo Ruiz, Leslie Elizabeth Leiva Rodríguez, Primary gastric squamous cell carcinoma of indeterminate origin managed by laparoscopic multivisceral resection: a case report, Journal of Surgical Case Reports, Volume 2025, Issue 12, December 2025, rjaf993, https://doi.org/10.1093/jscr/rjaf993
Close - Share Icon Share
Abstract
Primary squamous cell carcinoma (SCC) of the stomach is an exceptionally rare tumor, comprising <0.5% of gastric malignancies. Diagnosis requires strict histopathologic and immunohistochemical confirmation to distinguish it from adenosquamous carcinoma or esophageal extension. We report a 30-year-old woman with chronic anemia and recurrent upper gastrointestinal bleeding. Endoscopy revealed a large gastric polypoid lesion, and computed tomography showed an exophytic mass invading the pancreatic tail and spleen. The patient underwent total gastrectomy with distal pancreatectomy, splenectomy, and colectomy, initiated laparoscopically and converted to open surgery for en bloc resection. Histopathology confirmed a moderately differentiated pure squamous carcinoma with lymphovascular and perineural invasion. Despite postoperative complications, the patient recovered after multiple re-explorations. This case underscores the diagnostic challenge and clinical significance of primary gastric SCC, highlighting the importance of accurate identification to guide appropriate surgical management.
Introduction
Primary gastric squamous cell carcinoma (GSCC) is an extremely uncommon histological variant, accounting for 0.04%–0.4% of gastric malignancies. Since the first case described by Röring in 1895, fewer than 100 have been documented worldwide. It predominates in men and typically appears in the sixth decade, most often in the upper stomach. Its pathogenesis remains elusive; proposed mechanisms include squamous metaplasia from chronic inflammation, differentiation of multipotent stem cells, or transformation of ectopic squamous epithelium [1–3].
Clinical manifestations are nonspecific, ranging from epigastric pain and weight loss to upper gastrointestinal bleeding. We report a rare GSCC managed initially by laparoscopy, later converted to open surgery due to multivisceral invasion [1, 2].
Case report
A 30-year-old woman with chronic iron-deficiency anemia and recurrent upper gastrointestinal bleeding was admitted for evaluation. On presentation, she had grade II anemia and severe malnutrition (CONUT [4] score 12).
Endoscopy demonstrated multiple gastric polyps. Biopsy revealed hyperplastic polyps with mild dysplasia and chronic active inflammation. A subsequent endoscopy identified a tubular-type polyp with severe edema, ulceration, and mixed inflammatory infiltrate. Persistent bleeding prompted a third endoscopy, which showed a friable, ulcerated, polypoid mass >10 mm along the greater curvature. Partial endoscopic resection revealed a moderately differentiated, non-keratinizing invasive squamous carcinoma, with tumor cells positive for CK 5/6, CK 7, and focal CK 20, a Ki-67 index of 50%, and negative for chromogranin. The neoplasm was therefore classified as a moderately differentiated squamous carcinoma of indeterminate origin (gastric vs pancreatic).
Triple-phase contrast-enhanced computed tomography (CT) of the upper abdomen demonstrated an exophytic, well-defined, lobulated lesion of the gastric fundus measuring 83 × 72 × 79 mm (Fig. 1).
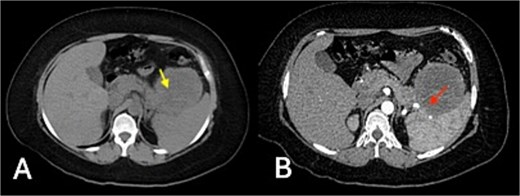
Triple-phase contrast-enhanced CT of the upper abdomen: (A) non-contrast phase showing an exophytic, well-defined, lobulated lesion of the gastric fundus (83 × 72 × 79 mm) with isodense attenuation (20 HU) (yellow arrow); (B) arterial phase revealing mild heterogeneous enhancement (up to 35 HU) and central hypodensity, with loss of interface between the mass, pancreatic tail, and spleen (red arrow indicates area of interface loss).
Given progressive anemia and recurrent bleeding, the patient underwent total gastrectomy with Roux-en-Y esophagojejunostomy, distal pancreatectomy, splenectomy, and segmental resection of the transverse colon with mechanical end-to-end anastomosis. The procedure began laparoscopically and revealed a 10 cm tumor between the spleen and pancreatic tail, eroding the serosa of the greater curvature and invading the transverse colon. Dissection proceeded with mobilization of the greater curvature, the hepatoduodenal ligament, and the pancreatic head (Fig. 2). Due to colonic invasion, conversion to open surgery was required to complete the colectomy and reconstruction. Estimated blood loss was 800 ml, and intraoperative peritoneal lavage cytology was negative for malignant cells.
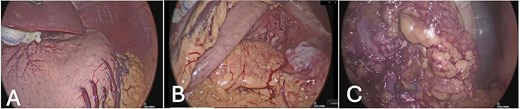
Intraoperative laparoscopic views: (A) stomach in situ before dissection; (B) exposure after mobilization of the greater curvature; (C) dissection of the hepatoduodenal ligament and pancreatic head.
Gross examination of the resected specimen revealed an exophytic, ulcerated mass (6 × 6 × 3.5 cm) in the distal stomach and a solid, pale lesion (6 × 3.5 × 2 cm) within the pancreatic tail, both contiguous (Fig. 3). Microscopy confirmed a keratinizing, moderately differentiated squamous carcinoma infiltrating the gastric wall and pancreas, with lymphovascular and perineural invasion. One of the 14 perigastric lymph nodes was positive for metastasis; surgical margins were negative, and the spleen and colon were free of tumor.
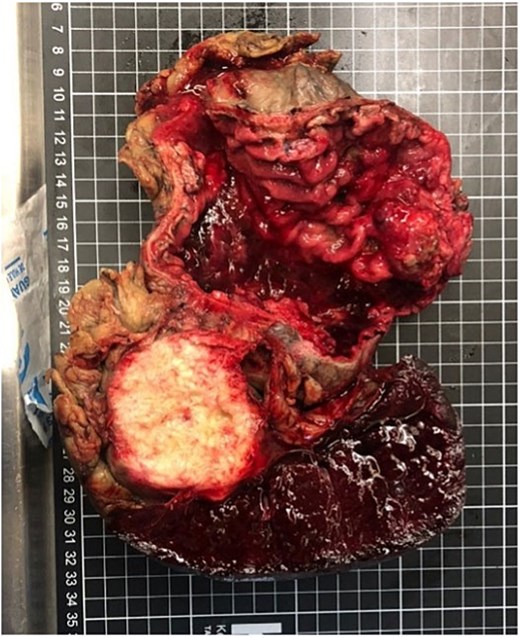
Gross specimen showing an exophytic, ulcerated distal gastric tumor (6 × 6 × 3.5 cm) and a solid lesion in the pancreatic tail (6 × 3.5 × 2 cm), both in continuity.
During early postoperative period, the patient developed an enterocutaneous fistula secondary to anastomotic leakage and intra-abdominal sepsis. She required four sequential re-operations over ~3 weeks:
First re-exploration (postoperative Day 6): abdominal lavage (~700 ml intestinal fluid drained), creation of a transverse colostomy, and placement of a vacuum-assisted closure (VAC) system.
Second intervention (5 days later): repeat abdominal lavage, ostomy remodeling, and VAC replacement.
Third procedure (4 days after the second): further cavity debridement and new VAC system placement.
Final operation (5 days later, postoperative Day ~20): definitive wound closure and removal of negative-pressure therapy following adequate granulation and infection control.
Cultures from drainage fluid grew Klebsiella pneumoniae (ESBL-negative), Acinetobacter baumannii complex, and Enterococcus faecium. Directed antibiotic therapy with carbapenems and fluconazole led to clinical and biochemical improvement. Nutritional support was maintained via jejunostomy and parenteral supplementation until contrast imaging confirmed the integrity of the esophagojejunal anastomosis. Oral feeding was then resumed and gradually advanced to a soft diet with good tolerance.
The patient was discharged in stable condition after an extended hospitalization. Final pathology confirmed a moderately differentiated squamous carcinoma of uncertain origin (gastric vs pancreatic), with hepatic metastatic foci detected radiologically but not surgically resected. She remains under oncologic and nutritional follow-up for systemic therapy and colostomy care (Fig. 4).
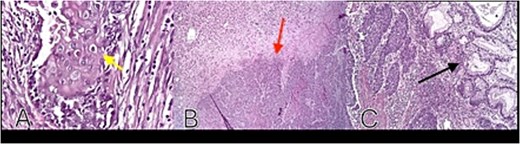
Histopathology: (A) neoplastic squamous cells with eosinophilic cytoplasm, well-defined borders, oval nuclei, prominent nucleoli, and keratin pearl formation (H&E, ×40) (yellow arrow); (B) tumor invasion into mucosa and submucosa (red arrow); (C) adjacent gastric glands without atypia (black arrow).
Discussion
Primary GSCC is an exceptional diagnostic entity, as most cases initially labeled “squamous” are later reclassified as adenosquamous carcinomas or direct extensions of esophageal tumors. The diagnostic criteria proposed by Parks exclude lesions involving the gastric cardia or those with esophageal extension [1]. Boswell and Helwig [2] described the classic histologic hallmarks of keratin pearls, mosaic arrangement, and intercellular bridges. The Japanese Gastric Cancer Association defines GSCC as a carcinoma composed entirely of squamous cells without glandular differentiation and originating from the gastric mucosa [5, 6]—criteria all fulfilled in this case.
Immunohistochemistry confirms squamous differentiation (CK5/6, p53) and excludes adenocarcinoma or neuroendocrine components. The pathogenesis remains uncertain, with hypotheses including chronic irritation, ulcer scarring, or stem-cell metaplasia [3, 5, 6]. A hereditary predisposition has not been proven, although our patient had a strong familial oncologic history.
Clinically, GSCC resembles gastric adenocarcinoma and is usually detected at advanced stages, explaining its poor prognosis. Five-year survival is reported at <10%, rising to about 35% in Japanese series [6]. Surgery remains the mainstay of treatment; the benefit of adjuvant or neoadjuvant therapy is unclear, although cisplatin-based combinations have been reported [7].
Our patient underwent total gastrectomy with distal pancreatectomy and splenectomy due to multivisceral invasion. The initial laparoscopic approach was converted to open surgery for en bloc resection. The relevance of this report lies not in the surgical technique but in the rarity of the histologic subtype and its diagnostic challenge. The postoperative course was complicated by anastomotic leakage and sepsis, underscoring morbidity in malnourished patients, reflected by a CONUT score of 12 [4].
Misdiagnosis as adenocarcinoma or esophageal squamous carcinoma could result in inappropriate staging or therapy. Accurate histopathologic and immunohistochemical evaluation is therefore crucial. Follow-up with clinical review and CT or magnetic resonance imaging every 6–12 months for at least 3 years is recommended.
Conflict of interest statement
The authors declare no conflict of interest.
Funding
No external funding was received.



