-
PDF
- Split View
-
Views
-
Cite
Cite
Ivan Adamovic, Hans-Dieter Mäder, Alexander Golowan, Kostas Michael, Thibika Sivakumar, Jörg Lucian, Lukas Krähenbühl, The use of intact fish skin grafts in the treatment of acute complicated bite wounds: a case report, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag197, https://doi.org/10.1093/jscr/rjag197
Close - Share Icon Share
Abstract
The management of complex bite wounds, particularly in patients with comorbidities, presents a significant clinical challenge due to the high risk of infection and prolonged healing times. This case report highlights the successful use of intact fish skin grafts as a novel treatment modality for a primary contaminated bite wound at the patient with comorbidities such as diabetes mellitus and rheumatoid artritis with cortisone use. A 54-year-old European female patient with a medical history of rheumatoid arthritis and type 2 diabetes mellitus presented with a large, full-thickness dog bite wound on her right lower leg. Surgical debridement and broad-spectrum antibiotic administration, an intact Kerecis fish skin graft was applied and secured. The application of an intact fish skin xenograft, combined with surgical debridement and appropriate antibiotic therapy, proved to be a highly effective treatment for a complex bite wound in an immunocompromised patient.
Introduction
Existing evidence shows that acellular fish skin grafts AFSGs accelerate wound healing, reduce pain, prevent antibiotic administration, and cause no autoimmune reactions [1, 2].
A search of major medical databases to our knowledge does not reveal any dedicated case reports, case series, observational studies, or clinical trials on the use of Kerecis for human bite wounds. However, some research on primarily contaminated wounds suggests that Kerecis fish skin xenograft products, which have been introduced to treat diabetic wounds, traumatic wounds, partial-thickness burns, and necrotic wounds, may be applicable [3, 4].
Microorganisms found in bite wounds from cats and dogs are often a mix of bacteria from the animal's oral flora and the human's skin flora. One microorganism stands out as the most common and clinically significant pathogen, especially in cat and dog bites: Pasteurella multocida. It causes a rapidly progressing infection with intense pain, redness, and swelling, often appearing within 24 hours of the bite [5]. While P. multocida typically causes an acute infection, it can lead to a prolonged, chronic wound in patients with underlying conditions, especially by immunocompromised patients [6].
Early identification and surgical debridement and antibiotics application are critical for managing this kind of injury and its associated complications. Following the removal of ischemic and necrotic tissue, reconstructing the substantial soft-tissue defects is a necessity. Such defects may require the use of skin flaps or split-thickness grafts, while others may necessitate a free-tissue transfer [7].
The Kerecis™ fish skin xenograft is a product derived from minimally processed North Atlantic Cod, which allows it to act similarly to human skin. The porous structure of Kerecis prevents fluids from collecting in potential pockets of the bite wound, which avoids an additional risk of abscess formation or the spread of infection [8].
Case report
A 54-year-old European female patient presented immediately after sustaining a dog bite injury from own dog on 28 September 2024. She presented with an open wound and a large skin and subcutaneous defect (6 cm wide × 6 cm long and 0.6 cm deep) on the distal-lateral aspect of her right lower leg. Her medical history was significant for rheumatoid arthritis (using corticosteroids) and type 2 diabetes mellitus, which are both known to cause delayed wound healing (Fig. 1).
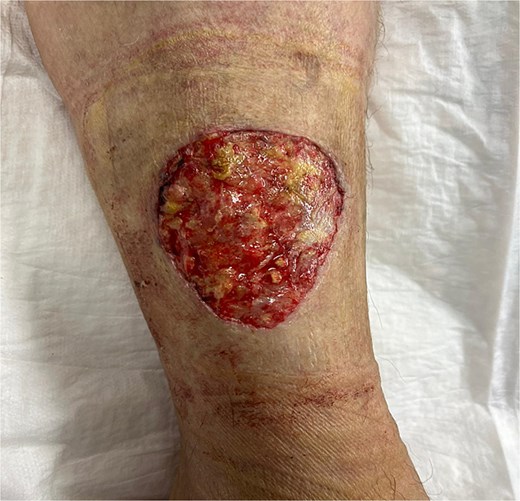
Wound-large skin and subcutaneous defect on the distal-lateral aspect of her right lower leg.
Clinical assessment showed absence of clinical signs of infection. The patient's history included a tendency for prolonged wound healing after injuries and the formation of scar tissue.
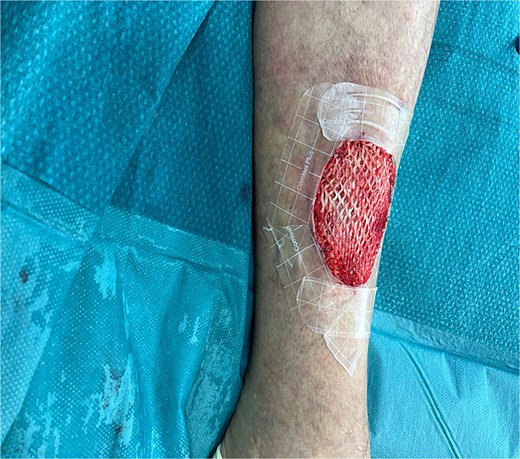
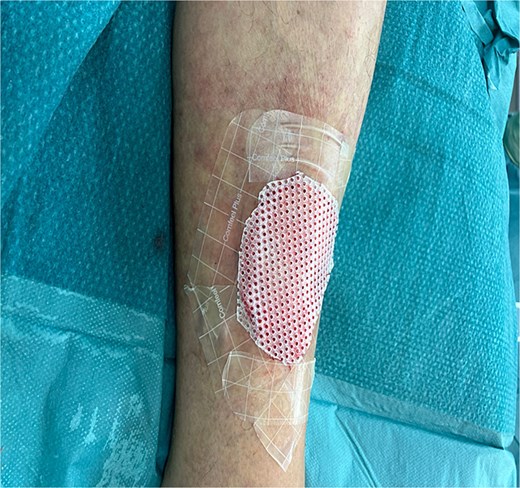
Given her history of prolonged wound healing our treatment plan included surgical debridement in the operating theatre to remove all ischemic and non-viable tissue. This was followed by the application of a Kerecis fish skin graft to cover the defect (Fig. 2). The graft was fixated to the intact skin edges with Monocryl 4–0 sutures to prevent migration. Application of a Mepitel spacer grid protected the graft after dressing change (Fig. 3).
Microbiology results isolated only P. multocida, which was found to be sensitive to amoxicillin-clavulanic acid. The studies showed that the best treatment for animal bites with suspected Pasteurella spp usually entails a combination of amoxicillin and the β-lactamase inhibitor clavulanic acid [9]. For most superficial bite wound infections without deep tissue involvement, a 7- to 10-day course is generally sufficient. Drug resistance in Pasteurella spp human infections has rarely been reported in the literature [10].
The wound was then managed with negative pressure wound therapy (NPWT) for 3 days to optimize the wound bed and promote healing (Fig. 4). Continuous negative pressure of 125 mmHg was applied.
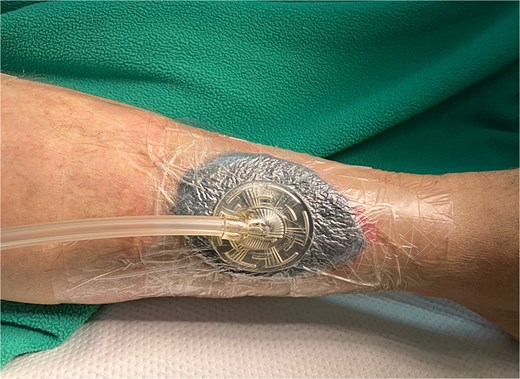
After VAC removal, we discharged the patient on the third postoperative day (Fig. 5).
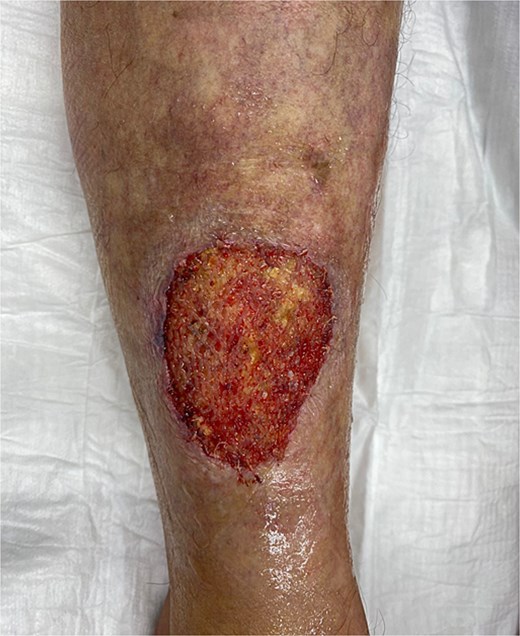
Wound assessment three days postoperatively, following removal of the VAC dressing: Showing a partially integrated Kerecis fish skin graft with beginning granulation and without any signs of infection.
Follow-up inspections have been done twice a week (Monday and Thursday). Following the removal of the previous dressing, the wound was cleaned and disinfected with hypochlorous acid. A non-adherent spacer grid–Mepitel, was then applied to the wound bed to protect the new tissue. This was followed by the application of a superabsorbent pad to manage any exudate. Figures 6 through 9 illustrate the progression of the wound until complete healing:
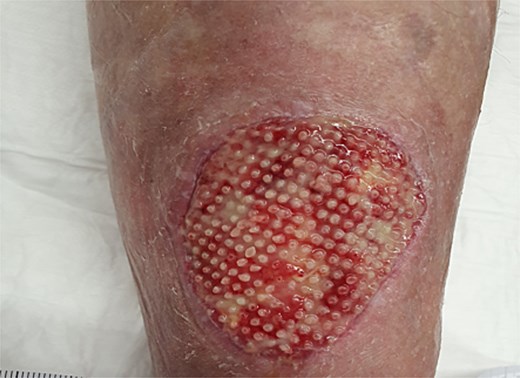
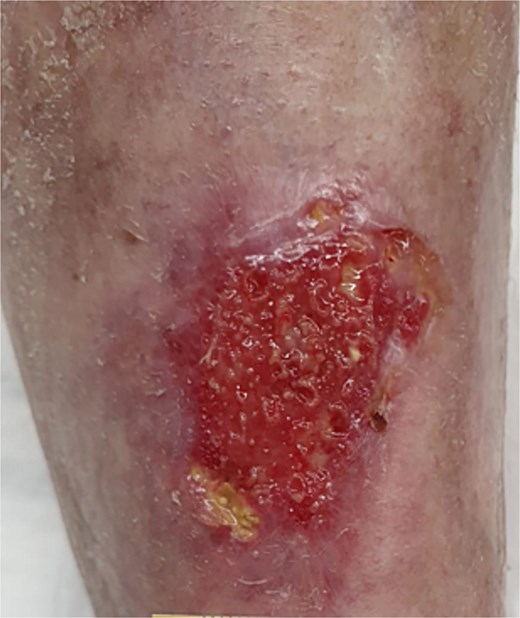
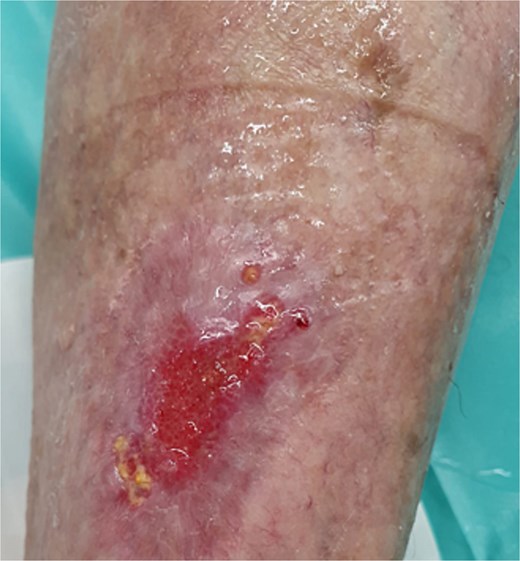
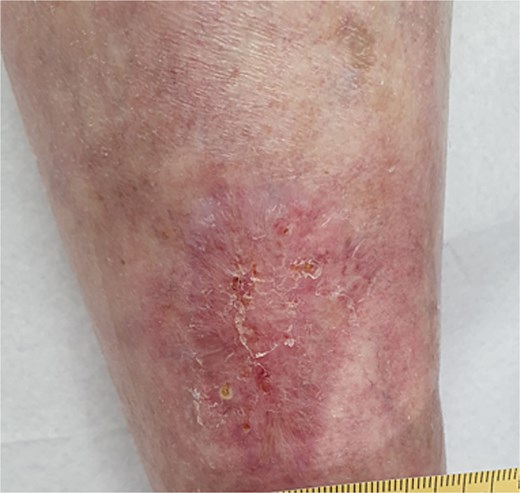
We achieved wound closure in 10 weeks. After a 6-month follow-up, there was no new re-opening of the wound, and the skin and scar quality was much better and more elastic than the rest of the parchment skin. Important dates and times in this case are presented in Table 1 as a timeline.
| Date/Timeframe . | Event . |
|---|---|
| 28 September 2024 (Admission) | Patient presented immediately after dog bite |
| Day 1 (Surgery) | Surgical debridement performed in the operating theatre and Kerecis fish skin graft applied. |
| Day 3 (Post-Op) | NPWT removed and patient discharged. Wound check performed |
| Follow-up inspections twice a week (Monday and Thursday) from Day 4 till Day 28. | Routine dressing changes and wound care by the same team. |
| 4 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 6 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 8 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 10 Weeks Post-Op | Final assessment. Wound closure achieved. The patient was completely mobile without pain. |
| 6-Month Follow-up | Final evaluation. No new re-opening of the wound; scar quality was noted to be much better and more elastic than the surrounding parchment skin |
| Date/Timeframe | Event |
|---|---|
| 28 September 2024 | Patient presented immediately after dog bite |
| Day 1 (Surgery) | Surgical debridement performed in the operating theatre and Kerecis fish skin graft applied. |
| Day 3 (Post-Op) | NPWT removed and patient discharged. Wound check performed |
| Follow-up inspections twice a week (Monday and Thursday) | Routine dressing changes and wound care by the same team. |
| 4 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 6 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 8 Weeks Post-Op | Wound check with illustration shows continued healing and granulation. |
| 10 Weeks Post-Op | Final assessment. Wound closure achieved. The patient was completely mobile without pain. |
| 6-Month Follow-up | Final evaluation. No new re-opening of the wound; scar quality was noted to be much better and more elastic than the surrounding parchment skin |
Discussion
Full-thickness wounds in patients with comorbidities and infection can take up to 24 months to heal. An established infection, as is often caused by P. multocida, significantly delays healing. Pre-existing conditions such as diabetes mellitus or rheumatoid arthritis are known to impair wound healing as well [11, 12]. According to these findings, we observed that the application of AFSGs-Kerecis accelerated wound healing to only 10 weeks. The healing progressed steadily without any deterioration.
Scar tissue tensile strength will only reach 80%, compared to the original tissue [13]. In our case, the quality of the scar tissue was clinically very good and more elastic than the existing parchment skin. The tissue's tensile strength was 100%, which is 20% greater than the average outcome.
In our experience the consistent application of follow-up and wound care by the same team of experts is of great importance. This continuity ensures a unified treatment strategy and accurate assessment of the wound's progress, which are critical for optimal healing outcomes.
In the existing literature to our knowledge, we did not find any supportive paper on the use of fish skin grafts for human bite wounds, with the exception of one case study on animals [14].
Using the Kerecis graft as a proactive measure, even in primary bite wounds without immediate signs of infection, is a key takeaway. This approach aims to prevent complications from developing, rather than treating them after they occur.
The findings of this case should encourage further research. A controlled study comparing outcomes of traditional wound care with Kerecis grafts in similar patient populations would provide more robust evidence and help establish it as a standard of care.
Acknowledgement
Many thanks to our dedicated wound nurse—Bettina Maeder and department manager—Marion Notter, for meticulous documentation, administrative support, and photographs.
Author contributions
I.A. wrote the manuscript and was operating surgeon, H-D.M., A.G., K.M., T.S., J.L. have done literature search and helped by discussion, L.K. helped draft and corrected end version of the manuscript.
Conflicts of interest
The all authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
No specific funding was disclosed.
Data availability
The dataset supporting the conclusions of this article is available from the corresponding author upon reasonable request.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Written informed consent was obtained from the patient for the publication of this case report and any accompanying images. A copy of the written consent is available upon request.



