-
PDF
- Split View
-
Views
-
Cite
Cite
Chen C Fang, Sanjeet Singh Avtaar Singh, Malcolm Will, Abdominal compartment syndrome after robotic-assisted thoracoscopic surgery (RATS) diaphragm plication, Journal of Surgical Case Reports, Volume 2026, Issue 2, February 2026, rjaf1050, https://doi.org/10.1093/jscr/rjaf1050
Close - Share Icon Share
Abstract
Diaphragm plication is an effective treatment for diaphragmatic paralysis, in which the affected diaphragm is sutured in place. Robotic-assisted thoracoscopic (RATS) diaphragm plication combines the advantages of standard open thoracotomy and conventional thoracoscopic technique, gaining popularity as a less invasive alternative to improve the overall lung function. We present a case, to our knowledge, of the first described instance of abdominal compartment syndrome in the literature following RATS diaphragmatic plication. Abdominal compartment syndrome potentially developed as a consequence of downward hepatic shift and reduced intra-abdominal volume. This was inferred by changes in the patient’s liver enzymes and was manifested by his oliguria and raised intra-abdominal pressure. The usual treatment of abdominal compartment syndrome is debatable as it usually involves conservative decompression including percutaneous options, but definitive surgical management includes decompression of the abdomen, often by emergent laparotomy, in those with moderately elevated intra-abdominal pressure. Conservative treatment by means of supportive therapy and abdominal decompression with nasogastric tube and passing of a flatus tube was performed in our case.
Introduction
Diaphragm plication is a surgical treatment for diaphragmatic paralysis, whereby the affected diaphragm is sutured in place, providing relief of symptoms with objective improvements in lung function and hypoxemia [1]. Robotic-assisted thoracoscopic (RATS) diaphragm plication combines the advantages of standard open thoracotomy and conventional thoracoscopic techniques, thus, gaining popularity as a less invasive alternative to improve the overall lung function. To our knowledge, we present a case of the first described instance of abdominal compartment syndrome (ACS) in the literature following RATS diaphragmatic plication.
Case presentation
A 53-year-old man presented with a 6-month history of shortness of breath and cough. His chest X-ray showed elevation of the right hemidiaphragm (Fig. 1) and the computed tomography (CT) scan noted no external compression or invasion to the right phrenic nerve alongside a significant loss of volume of the right lower and middle lobes due to compression from the liver and elevated diaphragm. An ultrasound sniff test demonstrated paradoxical movement of the right hemidiaphragm: while the left hemidiaphragm descended during inspiration, the right remained static—consistent with right phrenic nerve palsy.
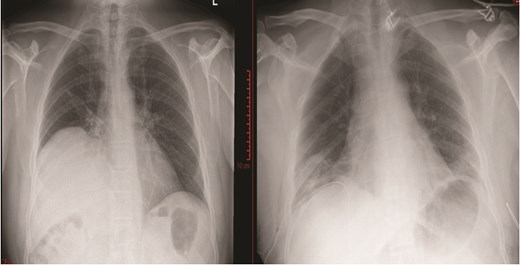
Chest X-ray prior to diaphragmatic plication (left); chest X-ray post diaphragmatic plication (right).
Despite being an active marathon runner beforehand, his recent pulmonary function tests showed significant airflow obstruction, and cardiopulmonary exercise test suggested deconditioning with little breathing reserve at the end of exercise, consistent with the right phrenic palsy and deconditioning. Both supine and standard pulmonary function tests were performed which showed an obstructive and restrictive component but supranormal diffusion capacity when corrected.
He underwent a RATS plication of the right hemidiaphragm after a year of watchful waiting to assess for spontaneous recovery. The diaphragm was plicated with multiple horizontal mattress 2/0 ethibond sutures reinforced with pledgets and secure with Coreknot device to plicate the diaphragm as it was quite tense and placing the knots with the robotic instruments was difficult. CO2 insufflation was performed up to 8 mmHg to assist in the procedure. Then multiple horizontal mattress 0 ethibond sutures reinforced with pledgets were used to provide additional support to the plication repair and were tied using the robotic instruments. Postoperative chest x-ray showed the right hemidiaphragm in an improved position (Fig. 1).
On the first post-operative day, he developed a mildly distended abdomen and became oliguric. Despite intravascular fluid replacement to an adequate mean arterial pressure (75 mmHg) and passage of a nasogastric tube to decompress the stomach, the oliguria persisted. Intra-abdominal pressure (IAP) was measured via a urinary catheter and noted to be 20 mmHg, which confirmed ACS. The serum lactate levels however remained within normal limits throughout. His renal function and liver function tests were as shown in Table 1.
| Post-operative Day . | −1 . | 1 . | 2 . | 3 . | 4 . | 5 . | 6 . |
|---|---|---|---|---|---|---|---|
| Urea (mmol/l) (6.6) | 6.2 | 7.4 | 4.9 | 2.7 | 2.1 | 2.9 | 2.6 |
| Creatinine (μmol/l) (111) | 94 | 169 | 97 | 78 | 82 | 71 | 93 |
| Alkaline Phosphatase (U/l) (125) | 63 | 64 | 56 | 58 | 89 | 133 | 135 |
| Alanine Transaminase (U/l) (50) | 36 | 966 | 823 | 679 | 514 | 369 | 300 |
| Bilirubin (μmol/l) (21) | 48 | 68 | 99 | 63 | 41 | 39 | 30 |
| Intra-abdominal pressure (mmHg) | 20 | 20 | 16 | 14 |
| Post-operative Day | −1 | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|---|
| Urea (mmol/l) (6.6) | 6.2 | 7.4 | 4.9 | 2.7 | 2.1 | 2.9 | 2.6 |
| Creatinine (μmol/l) (111) | 94 | 169 | 97 | 78 | 82 | 71 | 93 |
| Alkaline Phosphatase (U/l) (125) | 63 | 64 | 56 | 58 | 89 | 133 | 135 |
| Alanine Transaminase (U/l) (50) | 36 | 966 | 823 | 679 | 514 | 369 | 300 |
| Bilirubin (μmol/l) (21) | 48 | 68 | 99 | 63 | 41 | 39 | 30 |
| Intra-abdominal pressure (mmHg) | 20 | 20 | 16 | 14 |
His surgical options included release of the plication or a decompressive laparotomy should symptoms worsen. He was kept well hydrated throughout with his mean arterial pressure maintained above 70 mmHg. In the ensuing 24 h, his abdominal distension reduced and oliguria resolved. Subsequent IAP measurements were progressively lower, alongside normalization of renal and hepatic function Table 2. He was then discharged home on day 6.
| Pulmonary function test . | Actual percentage of predicted value (%) . | |
|---|---|---|
| Standing . | Supine . | |
| Forced Expiratory Volume in 1 second (FEV1) | 50 | 61 |
| Forced Vital Capacity (FVC) | 48 | 54 |
| Diffusion Capacity of Carbon Monoxide (DLCO) | 67 | N/A |
| DLCO corrected for Volume (KCO) | 132 | N/A |
| Cardiopulmonary exercise testing | ||
| Volume of oxygen consumed during peak exercise (VO2max) | 83 | N/A |
| Anaerobic Threshold (AT) | 39 | N/A |
| Pulmonary function test | Actual percentage of predicted value (%) | |
|---|---|---|
| Standing | Supine | |
| Forced Expiratory Volume in 1 second (FEV1) | 50 | 61 |
| Forced Vital Capacity (FVC) | 48 | 54 |
| Diffusion Capacity of Carbon Monoxide (DLCO) | 67 | N/A |
| DLCO corrected for Volume (KCO) | 132 | N/A |
| Cardiopulmonary exercise testing | ||
| Volume of oxygen consumed during peak exercise (VO2max) | 83 | N/A |
| Anaerobic Threshold (AT) | 39 | N/A |
Discussion
Elevations in liver enzyme, specifically in alkaline phosphatase (ALP) and alanine transaminase (ALT), and worsening of renal function test post-diaphragmatic plication are well described and other causes for transaminitis were ruled out. In addition, the patient's vitals had remained stable throughout his hospital stay with no intraoperative hypotension or vasopressor requirements, making ischemic hepatitis, and hemodynamic instability unlikely. Although the patient had a history of Gilbert's syndrome, causing a transient elevated bilirubin, it rarely causes sustained liver enzyme derangement. In considering the differentials, the plication procedure may have elicited an irritation of the hepatic capsule, which sits below the diaphragmatic fold [2]. Due to the lack of tactile feedback from placement of the sutures, care should be taken when placing the sutures.
ACS potentially developed as a consequence of a downward hepatic shift and reduced intra-abdominal volume. This was inferred by changes in the patient’s liver enzymes and was manifested by his oliguria and raised IAP [3]. Fortunately, our patient improved with symptomatic management after surgery, which is the treatment of choice for hepatic traumas in hemodynamically stable patients.
Our case represents the second report in the literature of ACS after a right-sided diaphragm plication with the difference being a RATS procedure as opposed to open thoracotomy [3]. RATS plication is believed to be associated with fewer complications, but our case shows that ACS may still occur. The modes of injury may mechanistically differ as with open thoracotomy plications, there may be a larger area of diaphragmatic displacement during retraction and a possibility of liver hematoma forming around the outgoing hepatic vessels [2]. By contrast, RATS plication is more likely to be localized and with controlled areas of hematoma formation, possibly only affecting vessels in close proximity.
The normal IAP ranges from 5 to 7 mmHg. Intra-abdominal hypertension (IAH) is described when IAP is consistently >12 mmHg. ACS is an increasingly recognized clinical entity. It is defined by an IAP of greater than 20 mmHg with signs of end-organ compromise.
Failure to recognize and manage ACS can lead to poor prognosis and is recognized as an independent predictor of mortality. The Incidence, Risk Factors, and Outcomes of Intra-Abdominal Hypertension in Critically Ill Patients (IROI Study) noted that 34% of patients in the critical care setting had IAH on the day of admission, rising to 48.9% within 14 days [4]. The development of IAH within the observed period was associated with mortality. IAH post thoracic surgery remains rare and probably under-reported within the literature. Complications of diaphragmatic plication including pneumonia and persistence of symptoms are reported in 0 to 30% of patients [4]. Mortality, when reported, tends to be due to progression of the pathology underlying the raised hemidiaphragm, rather than the plication itself.
The usual treatment of ACS is debatable as it usually involves conservative decompression including percutaneous options, but definitive surgical management includes decompression of the abdomen, often by emergent laparotomy, in those with moderately elevated IAP. In our case, we exercised conservative treatment using supportive therapy and abdominal decompression with nasogastric tube and passing of a flatus tube [2].
Conclusion
To our knowledge, this is the first report of ACS as a complication of RATS diaphragmatic plication. Principles of care include ensuring adequate hydration and perfusion of the abdominal organs by maintaining good perfusion pressure and careful monitoring of the intra-abdominal pressures.
Author contributions
CCF wrote the first draft of the manuscript, submitted the case report and was involved in the patient’s care. SS was involved in the patient’s care and reviewed and edited the manuscript. MW and SS performed the surgery. MW edited and reviewed the manuscript. CCF produced and edited figures. MW guided CCF through preparation of the manuscript draft. All authors read and approved the final manuscript.
Conflicts of interest statement
None declared.
Funding
The authors received no specific funding for this work.
Data availability
No new data were generated or analyzed in support of this research.



