-
PDF
- Split View
-
Views
-
Cite
Cite
Quoc Dung Nguyen, Jason Diab, David Khaicy, Vanessa Diab, Zachias Hopkins, Lai Heng Foong, Christophe R Berney, The impact of COVID-19 on delayed presentations of necrotising fasciitis, Journal of Surgical Case Reports, Volume 2022, Issue 2, February 2022, rjac015, https://doi.org/10.1093/jscr/rjac015
Close - Share Icon Share
Abstract
The purpose of this study was to determine the impact of coronavirus disease 2019 (COVID-19) on the delayed presentation of necrotising fasciitis (NF). A retrospective study was conducted of adult patients (≥16 years old) diagnosed with NF at a hospital from 2017 to 2020. A quantitative comparative analysis for the COVID-19 group and control group between 2017 and 2019. Structured interviews were conducted to examine the impact of COVID-19 on patients. There were 6 patients in the COVID-19 group and 10 patients in the control group. The COVID-19 group had a longer mean onset of symptoms till hospital presentation of 4.1 days and a longer mean operative time. The COVID-19 group was more likely to be admitted to intensive care unit. Three patients in the COVID-19 group did not survive compared to survival in the counterparts. Participant responses indicated the COVID-19 pandemic did not prevent them from presenting to ED.
INTRODUCTION
Coronavirus disease 2019 (COVID-19), a contagious disease, known as the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), was declared a pandemic by the World Health Organisation on 11th March 2020. The Australian government introduced ‘social distancing’ on 16th March 2020 and significant changes in the public health system were implemented. In emergency departments (ED), international communities recognized a reduction in presentations during the pandemic where reported decreases ranged from 13.0 to 46.3% in ED cases between March and April 2020, compared with the previous year [1, 2]. Anecdotally, we noted an unusual increase and pattern at a hospital with delayed presentations of necrotising fasciitis (NF). Two case studies have reported delayed presentations of NF due to fear of contracting SARS-CoV-2 [3, 4]. NF is a rare disease, but life-threatening soft tissue infection characterized by rapid inflammation and necrosis involving the epidermis, dermis, subcutaneous tissue, fascia and muscle [4]. The incidence of NF has been reported as 0.3–15 per 100 000 in the general population [5, 6]. It is considered a surgical emergency with a mortality of 20.6% commonly afflicting the perineum, lower extremities, abdomen and post-operative wounds [7]. This project aimed to study the impact of presentation and clinical outcomes for NF during the COVID-19 pandemic, compared with previous years at a hospital.
CASES SERIES
A retrospective review of all recorded case of NF was undertaken at a hospital from April to August, 2017–2020. Adult patients (≥16 years old) with a confirmed clinical diagnosis of NF were included. Patients were divided into two cohorts: the COVID-19 cohort included patients who presented to hospital from April to August 2020 and the control cohort for patients who presented from April to August 2017–2019.
All patient data were collected through electronic medical records for NF. The data collected included demographic information, clinical presentation, symptomology, medical comorbidities (including Charlson comorbidity index [11]), biochemical parameters (including Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC) score [8]), computed tomography (CT) scan, American society of anaesthesiology (ASA) score, time to theatre (hours), operative time (hours), surgical technique (debridement, flap), number of operations and Post-operative outcomes (complications, intensive care unit [ICU] admission and length of stay [LoS], total LoS and in-hospital mortality).
For the qualitative component, structured interviews examined the impact of COVID-19 on patients, their perception and feelings during the current outbreak by using bimodal questions (yes/no), grading questions (1 = very hard/most unlikely to 5 = very easy/most likely) and open-ended questions (Table 1).
Twelve questions for the structured interviews for patients in the COVID-19 cohort included [27]
|
Did you know someone who died from COVID-19? How easy or difficult was it for you to find information related to COVID-19? How easy or difficult was it for you to understand what to do if you had COVID-19? How easy or difficult was it for you to understand the restrictions and recommendations if you had COVID-19? How easy or difficult was it for you to follow the recommendations if you had COVID-19? How easy or difficult was it for you to follow the recommendations about staying at home from work? What did you consider the probability of getting COVID-19? How susceptible did you consider getting COVID-19? Did you know how to protect yourself from COVID-19? During the last 7 days before admission, which of the following measures did you take to prevent infection of COVID-19? Regarding the patient, how did you feel about COVID-19? Before presenting to the emergency department, were there any reasons in not coming due to COVID-19 or otherwise? |
Statistical analyses were performed using IBM SPSS Statistics version 26.0. Continuous variables were expressed as mean, range and standard deviation (SD). Continuous variables assessed the relationship between linear data and correlation based on a level of significance set at P value of 0.05. The differences between proportions between the COVID-19 and control cohort derived from categorical data were analysed using Fisher’s exact test and a Mann Whitney U test for continuous variables.
There were 6 patients in the COVID-19 cohort and 10 patients in the control cohort (Table 2). The COVID-19 cohort was 30.3 years older than their counterparts (73.8 vs 53.4, P = 0.008) with a male predominance (3:1). Caucasian ethnicity accounted for 62.5% of the total patients. Seven patients (43.8%) were from CALD communities. The average BMI was 30.5 kg/m2 with 37.5% obese. Four patients in the COVID-19 group (40.0%) and one patient in the control group (16.7%) were smokers. The COVID-19 group had greater proportions of the highest Charlson comorbidity index (≥3) compared with the control (6 vs 2, P = 0.007, Table 3).
| . | n (%) . |
|---|---|
| Demographic information | |
| Age [years], mean (SD) | 54.9 (± 23.3) |
| Range | 16–90 |
| Gender | |
| Male | 12 (75.0%) |
| Female | 4 (25.0%) |
| Weight [kg], mean (SD) | 94.2 (± 36.8) |
| Range | 59–200 |
| Body mass index (BMI) [kg/m2], mean (SD) | 30.5 (± 10.5) |
| Range | 20.2–57.8 |
| Obesity | 5 (31.3%) |
| Ethnicity | |
| Caucasian | 10 (62.5%) |
| Middle Eastern | 3 (18.8%) |
| Indigenous | 1 (6.3%) |
| Asian | 2 (12.5%) |
| CALD | 7 (43.8%) |
| Smoker | 5 (31.3%) |
| Clinical presentation and symptomology | |
| Onset of symptoms till presentation to the emergency department [days], mean (SD) | 3.9 (± 2.9) |
| Range | 1–10 |
| Clinical signs | |
| Disproportionate pain at the site of injury | 12 (75.0%) |
| Swelling of the skin | 14 (87.5%) |
| Erythema of the skin | 14 (87.5%) |
| Skin crepitus | 1 (6.3%) |
| Haemorrhagic bullae or blisters | 1 (6.3%) |
| Symptoms | |
| Presence of abdominal pain | 1 (6.3%) |
| Site of Injury | |
| Peripheral | 9 (56.3%) |
| Central | 7 (43.8%) |
| Previous trauma to the site | 2 (12.5%) |
| Influenza like symptoms | 3 (18.8%) |
| Decreased urine output | 1 (6.3%) |
| SIRS | 8 (50.0%) |
| Sepsis | 2 (12.5%) |
| Medical comorbidities | |
| Pre-existing medical illness | 11 (68.8%) |
| Diabetes | 4 (25.0%) |
| Previous abdominal surgery | 5 (31.3%) |
| Charlson comorbidity index | |
| Charlson score 0 | 4 (25.0%) |
| Charlson score 1 | 2 (12.5%) |
| Charlson score 2 | 2 (12.5%) |
| Charlson score ≥ 3 | 8 (50.0%) |
| Biochemical parameters [normal range], mean (SD) | |
| White cell count (×109/l) [4.0–10.0] | 20.2 (± 11.3) |
| Neutrophil (×109/l) [2.0–7.0] | 17.4 (± 10.7) |
| C reactive protein (mg/l) [<4.9] | 218.4 (± 156.8) |
| Haemoglobin (g/l) [130–170] | 129.5 (± 26.2) |
| Platelets (×109/l) [150–400] | 308.4 (± 123.7) |
| Creatinine (μmol/l) [60–110] | 120.8 (± 65.5) |
| Urea (mmol/l) [4.0–9.0] | 10.2 (± 9.2) |
| Albumin (g/l) [33–48] | 29.8 (± 8.2) |
| Lactate (mmol/l) [<1.9] | 2.65 (± 1.8) |
| Sodium (mmol/l) [135–145] | 135.1 (± 4.8) |
| Glucose (mmol/l) [7.8–11.0] | 10.1 (± 5.8) |
| LRINEC score, mean (SD) | 5.9 (± 4.7) |
| LRINEC category | |
| Low risk (≤ 5) | 7 (43.8%) |
| Moderate risk (6–7) | 1 (6.2%) |
| High risk (≥ 8) | 8 (50.0%) |
| Imaging | |
| CT scan | 11 (68.8%) |
| Management | |
| ASA status | |
| ASA 1 | 3 (18.8%) |
| ASA 2 | 2 (12.5%) |
| ASA 3 | 6 (37.5%) |
| ASA 4 | 5 (31.3%) |
| Time to theatre [hours], mean (SD) | 14.7 (± 10.5) |
| Range | 2.8–32.3 |
| Operative time [hours], mean (SD) | 1.39 (± 0.9) |
| Range | 0.2–3.0 |
| Surgical technique | |
| Debridement | 16 (100%) |
| Flap | 0 (0.0%) |
| Number of operations, mean (SD) | 2.75 (± 2.6) |
| Post-operative outcomes | |
| Post-operative complications | 4 (25.0%) |
| ICU | |
| Admission | 10 (62.5%) |
| Mean length of stay (SD) | 11.1 (± 10.0) |
| Range | 1–28 |
| Total length of stay in hospital [days], mean (SD) | 19.9 (± 19.6) |
| Range | 1–64 |
| In-hospital mortality | 3 (18.8%) |
| n (%) | |
|---|---|
| Demographic information | |
| Age [years], mean (SD) | 54.9 (± 23.3) |
| Range | 16–90 |
| Gender | |
| Male | 12 (75.0%) |
| Female | 4 (25.0%) |
| Weight [kg], mean (SD) | 94.2 (± 36.8) |
| Range | 59–200 |
| Body mass index (BMI) [kg/m2], mean (SD) | 30.5 (± 10.5) |
| Range | 20.2–57.8 |
| Obesity | 5 (31.3%) |
| Ethnicity | |
| Caucasian | 10 (62.5%) |
| Middle Eastern | 3 (18.8%) |
| Indigenous | 1 (6.3%) |
| Asian | 2 (12.5%) |
| CALD | 7 (43.8%) |
| Smoker | 5 (31.3%) |
| Clinical presentation and symptomology | |
| Onset of symptoms till presentation to the emergency department [days], mean (SD) | 3.9 (± 2.9) |
| Range | 1–10 |
| Clinical signs | |
| Disproportionate pain at the site of injury | 12 (75.0%) |
| Swelling of the skin | 14 (87.5%) |
| Erythema of the skin | 14 (87.5%) |
| Skin crepitus | 1 (6.3%) |
| Haemorrhagic bullae or blisters | 1 (6.3%) |
| Symptoms | |
| Presence of abdominal pain | 1 (6.3%) |
| Site of Injury | |
| Peripheral | 9 (56.3%) |
| Central | 7 (43.8%) |
| Previous trauma to the site | 2 (12.5%) |
| Influenza like symptoms | 3 (18.8%) |
| Decreased urine output | 1 (6.3%) |
| SIRS | 8 (50.0%) |
| Sepsis | 2 (12.5%) |
| Medical comorbidities | |
| Pre-existing medical illness | 11 (68.8%) |
| Diabetes | 4 (25.0%) |
| Previous abdominal surgery | 5 (31.3%) |
| Charlson comorbidity index | |
| Charlson score 0 | 4 (25.0%) |
| Charlson score 1 | 2 (12.5%) |
| Charlson score 2 | 2 (12.5%) |
| Charlson score ≥ 3 | 8 (50.0%) |
| Biochemical parameters [normal range], mean (SD) | |
| White cell count (×109/l) [4.0–10.0] | 20.2 (± 11.3) |
| Neutrophil (×109/l) [2.0–7.0] | 17.4 (± 10.7) |
| C reactive protein (mg/l) [<4.9] | 218.4 (± 156.8) |
| Haemoglobin (g/l) [130–170] | 129.5 (± 26.2) |
| Platelets (×109/l) [150–400] | 308.4 (± 123.7) |
| Creatinine (μmol/l) [60–110] | 120.8 (± 65.5) |
| Urea (mmol/l) [4.0–9.0] | 10.2 (± 9.2) |
| Albumin (g/l) [33–48] | 29.8 (± 8.2) |
| Lactate (mmol/l) [<1.9] | 2.65 (± 1.8) |
| Sodium (mmol/l) [135–145] | 135.1 (± 4.8) |
| Glucose (mmol/l) [7.8–11.0] | 10.1 (± 5.8) |
| LRINEC score, mean (SD) | 5.9 (± 4.7) |
| LRINEC category | |
| Low risk (≤ 5) | 7 (43.8%) |
| Moderate risk (6–7) | 1 (6.2%) |
| High risk (≥ 8) | 8 (50.0%) |
| Imaging | |
| CT scan | 11 (68.8%) |
| Management | |
| ASA status | |
| ASA 1 | 3 (18.8%) |
| ASA 2 | 2 (12.5%) |
| ASA 3 | 6 (37.5%) |
| ASA 4 | 5 (31.3%) |
| Time to theatre [hours], mean (SD) | 14.7 (± 10.5) |
| Range | 2.8–32.3 |
| Operative time [hours], mean (SD) | 1.39 (± 0.9) |
| Range | 0.2–3.0 |
| Surgical technique | |
| Debridement | 16 (100%) |
| Flap | 0 (0.0%) |
| Number of operations, mean (SD) | 2.75 (± 2.6) |
| Post-operative outcomes | |
| Post-operative complications | 4 (25.0%) |
| ICU | |
| Admission | 10 (62.5%) |
| Mean length of stay (SD) | 11.1 (± 10.0) |
| Range | 1–28 |
| Total length of stay in hospital [days], mean (SD) | 19.9 (± 19.6) |
| Range | 1–64 |
| In-hospital mortality | 3 (18.8%) |
| . | COVID-19 cohort (2020) (n = 6) . | Control cohort (2017–2019) (n = 10) . | P value . |
|---|---|---|---|
| Demographic information | |||
| Age [years], mean (SD) | 73.8 (± 11.3) | 43.5 (± 21.1) | 0.008* |
| Male, n (%) | 5 (83.3%) | 7 (70.0%) | 1.000 |
| Weight [kg], mean (SD) | 91.2 (± 26.7) | 96.5 (± 44.6) | 0.948 |
| Body mass index (BMI) [kg/m2], mean (SD) | 27.4 (± 6.9) | 34.1 (± 13.3) | 0.391 |
| Obesity | 3 (50.0%) | 2 (28.6%) | 0.592 |
| CALD, n (%) | 2 (33.3%) | 5 (50.0%) | 0.633 |
| Smoker | 4 (40.0%) | 1 (16.7%) | 0.588 |
| Clinical presentation and symptomology | |||
| Onset of symptoms till presentation to the emergency department [days], mean (SD) | 6.5 (± 2.3) | 2.4 (± 2.1) | 0.006* |
| Clinical signs | |||
| Disproportionate pain at the site of injury | 4 (66.7%) | 8 (80.0%) | 0.604 |
| Swelling of the skin | 5 (83.3%) | 9 (90.0%) | 1.000 |
| Erythema of the skin | 5 (83.3%) | 9 (90.0%) | 1.000 |
| Skin crepitus | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Haemorrhagic bullae or blisters | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Symptoms | |||
| Presence of abdominal pain | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Site of Injury | |||
| Peripheral | 3 (50.0%) | 6 (60.0%) | 1.000 |
| Central | 3 (50.0%) | 4 (40.0%) | 1.000 |
| Previous trauma to the site | 0 (0.0%) | 2 (20.0%) | 0.500 |
| Influenza like symptoms | 0 (0.0%) | 3 (30.0%) | 0.250 |
| Decreased urine output | 1 (16.7%) | 0 (0.0%) | 0.375 |
| SIRS | 4 (66.7%) | 4 (40.0%) | 0.608 |
| Sepsis | 1 (16.7%) | 1 (10.0%) | 1.000 |
| Medical comorbidities | |||
| Pre-existing medical illness | 6 (100.0%) | 5 (50.0%) | 0.093 |
| Diabetes | 2 (33.3%) | 2 (20.0%) | 0.604 |
| Previous abdominal surgery | 3 (50.0%) | 2 (20.0%) | 0.604 |
| Charlson comorbidity index | |||
| Charlson score 0 | 0 (0.0%) | 4 (40.0%) | 0.234 |
| Charlson score 1 | 0 (0.0%) | 2 (20.0%) | 0.500 |
| Charlson score 2 | 0 (0.0%) | 2 (22.2%) | 0.500 |
| Charlson score ≥ 3 | 6 (100.0%) | 2 (20.0%) | 0.007* |
| Biochemical parameters [normal range], mean (SD) | |||
| White cell count (×109/l) [4.0–10.0] | 20.4 (± 13.8) | 20.1 (± 10.4) | 0.745 |
| Neutrophil (×109/l) [2.0–7.0] | 18.2 (± 13.5) | 17.0 (± 10.4) | 1.000 |
| C reactive protein (mg/l) [<4.9] | 317.8 (±184.6) | 168.6 (± 122.0) | 0.111 |
| Haemoglobin (g/l) [130–170] | 126.0 (± 32.9) | 131.6 (± 23.0) | 0.704 |
| Platelets (×109/l) [150–400] | 307.5 (± 162.0) | 309.0 (± 104.4) | 0.745 |
| Creatinine (μmol/l) [60–110] | 172.5 (± 70.8) | 89.7 (± 38.9) | 0.009* |
| Urea (mmol/l) [4.0–9.0] | 18.0 (± 10.4) | 5.5 (± 4.0) | 0.003* |
| Albumin (g/l) [33–48] | 28.5 (± 10.0) | 30.4 (± 7.9) | 0.799 |
| Lactate (mmol/l) [<1.9] | 3.68 (± 2.0) | 1.76 (± 1.0) | 0.015* |
| Sodium (mmol/l) [135–145] | 135.5 (± 4.5) | 134.8 (± 5.2) | 0.827 |
| Glucose (mmol/l) [7.8–11.0] | 12.0 (± 8.0) | 8.7 (± 3.4) | 0.651 |
| LRINEC score, mean (SD) | 7.3 (± 5.3) | 5.1 (± 4.4) | 0.529 |
| LRINEC category, n (%) | |||
| Low risk (≤5) | 2 (33.3%) | 5 (50.0%) | 0.633 |
| Moderate risk (6–7) | 1 (16.7%) | 0 (0.0%) | 0.375 |
| High risk (≥8) | 3 (50.0%) | 5 (50.0%) | 1.000 |
| Imaging | |||
| CT scan | 6 (100%) | 5 (50.0%) | 0.093 |
| Management | |||
| ASA status | |||
| ASA 1 | 0 (0.0%) | 3 (18.8%) | 0.250 |
| ASA 2 | 0 (0.0%) | 2 (12.5%) | 0.500 |
| ASA 3 | 2 (12.5%) | 4 (25.0%) | 1.000 |
| ASA 4 | 4 (25.0%) | 1 (6.3%) | 0.036* |
| Time to theatre [hours], mean (SD) | 9.8 (± 7.3) | 17.6 (± 11.3) | 0.193 |
| Operative time [hours], mean (SD) | 2.2 (± 0.8) | 0.9 (± 0.6) | 0.013* |
| Surgical technique | |||
| Debridement | 6 (100.0%) | 10 (100.0%) | 1.000 |
| Number of operations, mean (SD) | 3.7 (± 3.4) | 2.2 (± 2.0) | 0.296 |
| Post-operative outcomes | |||
| Post-operative complications | 3 (50.0%) | 1 (10.0%) | 0.118 |
| ICU | |||
| Admission | 6 (100.0%) | 4 (40.0%) | 0.034* |
| Mean length of stay (SD) | 13.3 (± 11.7) | 7.8 (± 7.0) | 0.454 |
| Total length of stay in hospital [days], mean (SD) | 20.7 (± 11.5) | 19.5 (± 23.8) | 0.385 |
| In-hospital mortality | 3 (50.0%) | 0 (0.0%) | 0.036* |
| COVID-19 cohort (2020) (n = 6) | Control cohort (2017–2019) (n = 10) | P value | |
|---|---|---|---|
| Demographic information | |||
| Age [years], mean (SD) | 73.8 (± 11.3) | 43.5 (± 21.1) | 0.008* |
| Male, n (%) | 5 (83.3%) | 7 (70.0%) | 1.000 |
| Weight [kg], mean (SD) | 91.2 (± 26.7) | 96.5 (± 44.6) | 0.948 |
| Body mass index (BMI) [kg/m2], mean (SD) | 27.4 (± 6.9) | 34.1 (± 13.3) | 0.391 |
| Obesity | 3 (50.0%) | 2 (28.6%) | 0.592 |
| CALD, n (%) | 2 (33.3%) | 5 (50.0%) | 0.633 |
| Smoker | 4 (40.0%) | 1 (16.7%) | 0.588 |
| Clinical presentation and symptomology | |||
| Onset of symptoms till presentation to the emergency department [days], mean (SD) | 6.5 (± 2.3) | 2.4 (± 2.1) | 0.006* |
| Clinical signs | |||
| Disproportionate pain at the site of injury | 4 (66.7%) | 8 (80.0%) | 0.604 |
| Swelling of the skin | 5 (83.3%) | 9 (90.0%) | 1.000 |
| Erythema of the skin | 5 (83.3%) | 9 (90.0%) | 1.000 |
| Skin crepitus | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Haemorrhagic bullae or blisters | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Symptoms | |||
| Presence of abdominal pain | 1 (16.7%) | 0 (0.0%) | 0.375 |
| Site of Injury | |||
| Peripheral | 3 (50.0%) | 6 (60.0%) | 1.000 |
| Central | 3 (50.0%) | 4 (40.0%) | 1.000 |
| Previous trauma to the site | 0 (0.0%) | 2 (20.0%) | 0.500 |
| Influenza like symptoms | 0 (0.0%) | 3 (30.0%) | 0.250 |
| Decreased urine output | 1 (16.7%) | 0 (0.0%) | 0.375 |
| SIRS | 4 (66.7%) | 4 (40.0%) | 0.608 |
| Sepsis | 1 (16.7%) | 1 (10.0%) | 1.000 |
| Medical comorbidities | |||
| Pre-existing medical illness | 6 (100.0%) | 5 (50.0%) | 0.093 |
| Diabetes | 2 (33.3%) | 2 (20.0%) | 0.604 |
| Previous abdominal surgery | 3 (50.0%) | 2 (20.0%) | 0.604 |
| Charlson comorbidity index | |||
| Charlson score 0 | 0 (0.0%) | 4 (40.0%) | 0.234 |
| Charlson score 1 | 0 (0.0%) | 2 (20.0%) | 0.500 |
| Charlson score 2 | 0 (0.0%) | 2 (22.2%) | 0.500 |
| Charlson score ≥ 3 | 6 (100.0%) | 2 (20.0%) | 0.007* |
| Biochemical parameters [normal range], mean (SD) | |||
| White cell count (×109/l) [4.0–10.0] | 20.4 (± 13.8) | 20.1 (± 10.4) | 0.745 |
| Neutrophil (×109/l) [2.0–7.0] | 18.2 (± 13.5) | 17.0 (± 10.4) | 1.000 |
| C reactive protein (mg/l) [<4.9] | 317.8 (±184.6) | 168.6 (± 122.0) | 0.111 |
| Haemoglobin (g/l) [130–170] | 126.0 (± 32.9) | 131.6 (± 23.0) | 0.704 |
| Platelets (×109/l) [150–400] | 307.5 (± 162.0) | 309.0 (± 104.4) | 0.745 |
| Creatinine (μmol/l) [60–110] | 172.5 (± 70.8) | 89.7 (± 38.9) | 0.009* |
| Urea (mmol/l) [4.0–9.0] | 18.0 (± 10.4) | 5.5 (± 4.0) | 0.003* |
| Albumin (g/l) [33–48] | 28.5 (± 10.0) | 30.4 (± 7.9) | 0.799 |
| Lactate (mmol/l) [<1.9] | 3.68 (± 2.0) | 1.76 (± 1.0) | 0.015* |
| Sodium (mmol/l) [135–145] | 135.5 (± 4.5) | 134.8 (± 5.2) | 0.827 |
| Glucose (mmol/l) [7.8–11.0] | 12.0 (± 8.0) | 8.7 (± 3.4) | 0.651 |
| LRINEC score, mean (SD) | 7.3 (± 5.3) | 5.1 (± 4.4) | 0.529 |
| LRINEC category, n (%) | |||
| Low risk (≤5) | 2 (33.3%) | 5 (50.0%) | 0.633 |
| Moderate risk (6–7) | 1 (16.7%) | 0 (0.0%) | 0.375 |
| High risk (≥8) | 3 (50.0%) | 5 (50.0%) | 1.000 |
| Imaging | |||
| CT scan | 6 (100%) | 5 (50.0%) | 0.093 |
| Management | |||
| ASA status | |||
| ASA 1 | 0 (0.0%) | 3 (18.8%) | 0.250 |
| ASA 2 | 0 (0.0%) | 2 (12.5%) | 0.500 |
| ASA 3 | 2 (12.5%) | 4 (25.0%) | 1.000 |
| ASA 4 | 4 (25.0%) | 1 (6.3%) | 0.036* |
| Time to theatre [hours], mean (SD) | 9.8 (± 7.3) | 17.6 (± 11.3) | 0.193 |
| Operative time [hours], mean (SD) | 2.2 (± 0.8) | 0.9 (± 0.6) | 0.013* |
| Surgical technique | |||
| Debridement | 6 (100.0%) | 10 (100.0%) | 1.000 |
| Number of operations, mean (SD) | 3.7 (± 3.4) | 2.2 (± 2.0) | 0.296 |
| Post-operative outcomes | |||
| Post-operative complications | 3 (50.0%) | 1 (10.0%) | 0.118 |
| ICU | |||
| Admission | 6 (100.0%) | 4 (40.0%) | 0.034* |
| Mean length of stay (SD) | 13.3 (± 11.7) | 7.8 (± 7.0) | 0.454 |
| Total length of stay in hospital [days], mean (SD) | 20.7 (± 11.5) | 19.5 (± 23.8) | 0.385 |
| In-hospital mortality | 3 (50.0%) | 0 (0.0%) | 0.036* |
The average onset of symptoms till ED presentation was 3.9 (± 2.9) days. The COVID-19 group had a significantly longer mean onset of symptoms till ED presentation of 4.1 days compared with the control group (6.5 vs 2.4 days, P = 0.006). Most patients presented with disproportionate pain at the site of injury (75.0%), swelling (87.5%) and skin erythema (87.5%, Table 2). Skin crepitus and haemorrhagic bullae were only recorded in one patient (6.3%), respectively. The upper and lower limbs accounted for 56.3% of NF presentations. Four patients of the control group (40.0%) and four patients of the COVID-19 group (66.7%) presented with systemic inflammatory response syndrome (SIRS) (P = 0.608). Only one patient from each cohort met the quick sequential organ failure assessment (qSOFA) criteria for sepsis.
The COVID-19 cohort had almost twice the value of creatinine levels compared to their counterparts (172.5 vs 89.7 μmol/l, P = 0.009), almost triple the urea levels (18.0 vs 5.5 mmol/l, P = 0.003) and double the lactate levels (3.68 vs 1.76 mmol/l, P = 0.015, Table 3). The mean LRINEC score was 5.9 with half the total cohort at high risk of NF (LRINEC ≥8). The COVID-19 group had a significantly longer mean operative time (2.2 vs 0.9 h, P = 0.013). There was a higher proportion of Grade 4 ASA in the COVID-19 group compared with the control group (25.0% vs 6.3%, P = 0.036).
A total of four patients (25.0%) suffered post-operative complications: three cases of sepsis and one case of pneumonia and acute renal failure with no significant differences between groups. All patients in the COVID-19 group were admitted to ICU post-operatively compared to only 4 out of 10 in the control group (100.0% vs 40.0%, P = 0.034). The average ICU LoS was 11.1 days. The total in-hospital LoS was 19.9 days with no significant differences between groups (P = 0.385).
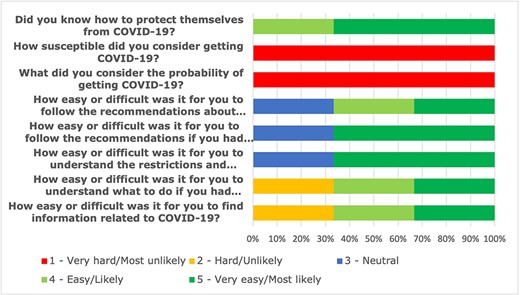
Three out of six patients of the COVID-19 group voluntarily participated in the questionnaire. The remaining patients or their next-of-kin declined. The patients were not infected with COVID-19 and had no known contacts with COVID-19. None of them lived in the immediate environment with suspected or confirmed COVID-19 cases (Table 4). They believed that their chance of contracting the virus was low and they were not susceptible to COVID-19 (Fig. 1). All participants stated that they felt ‘the virus was far away from them’ and they knew how to protect themselves from the virus with the preventive measures used, including hand washing for at least 20 s and wearing masks in public (Fig. 1). Most patients stated that it was easy to follow the instructions in case they contracted COVID-19. Only one patient found it was difficult to find information related to the COVID-19; whereas, another patient did not know what to do if he had COVID-19 due to his prolonged hospitalization since the beginning of the pandemic. All patients stated that the COVID-19 pandemic did not prevent them from presenting to ED (Table 4).
Demographics of qualitative component and summary of participants’ response to bimodal questions
| Patient demographics . | |
|---|---|
| Age [years], mean (± SD) . | 67.3 (± 9.03) . |
| Gender | |
| Male, n (%) | 3 (100.0%) |
| Ethnicity | |
| Caucasian, n (%) | 3 (100.0%) |
| CALD# | 0 (0.0%) |
| Body mass index (BMI) [kg/m2], mean (± SD) | 31.7 (± 7.1) |
| Weight [kg], mean (± SD) | 94.3 (± 15.1) |
| Negative COVID-19 test, n (%) | 3 (100.0%) |
| Living in the immediate environment with suspected/confirmed COVID-19 cases, n (%) | 0 (0.0%) |
| Bimodal questions | |
| Did you someone who died from COVID-19? | |
| Yes | 3 (100.0%) |
| No | 0 (0.0%) |
| Before presenting to the emergency department, were there any reasons in not coming due to COVID-19 or otherwise? | |
| Yes | 3 (100.0%) |
| No | 0 (0.0%) |
| Patient demographics | |
|---|---|
| Age [years], mean (± SD) | 67.3 (± 9.03) |
| Gender | |
| Male, n (%) | 3 (100.0%) |
| Ethnicity | |
| Caucasian, n (%) | 3 (100.0%) |
| CALD# | 0 (0.0%) |
| Body mass index (BMI) [kg/m2], mean (± SD) | 31.7 (± 7.1) |
| Weight [kg], mean (± SD) | 94.3 (± 15.1) |
| Negative COVID-19 test, n (%) | 3 (100.0%) |
| Living in the immediate environment with suspected/confirmed COVID-19 cases, n (%) | 0 (0.0%) |
| Bimodal questions | |
| Did you someone who died from COVID-19? | |
| Yes | 3 (100.0%) |
| No | 0 (0.0%) |
| Before presenting to the emergency department, were there any reasons in not coming due to COVID-19 or otherwise? | |
| Yes | 3 (100.0%) |
| No | 0 (0.0%) |
DISCUSSION
The COVID-19 pandemic has significantly impacted the predictable course of NF resulting in delayed presentations and worse clinical outcomes. Our experience was congruent with the overall decline in ED visits between March and May 2020 in Western Sydney Local Health District and Australia, with a drop of 25 and 38%, respectively [9, 10]. We identified that the COVID-19 group had a significantly longer mean onset of symptoms of NF before hospital presentation, which was greater than the average time of 4.5 days (range 1.0–13.3 days) reported in a NF systemic review and meta-analysis [7]. The COVID-19 cohort was also significantly older compared to the control with all patients having a higher Charlson comorbidity index (Table 3). Although interviews attempted to understand the ‘time and delay’ response from COVID, the cohort denied any reason for delay to ED with sound knowledge of personal protection. The patients overall had sufficient access to information, which Lim et al. found to normally reduce population-level anxiety and promote positive behavioural changes during an outbreak [11]. Other reasons that were more insightful from the progress notes and history taking identified that patients were more focused on respiratory symptoms of COVID-19, and other non-specific symptoms were considered less seriously [12].
In NF, the evolution of signs and symptoms can be non-specific and often missed adding confusion to the patient, but mainly the clinician. At the early stage of NF, symptoms commonly include swelling (75%), pain (72%) and erythema (72%) [5, 13]. In our study, we found comparable proportions with swelling (87.5%), pain (75%) and erythema (87.5%). These findings are identical to those generally found in patients with soft tissue infections and can lead to difficulty in making early diagnosis often requiring early multidisciplinary involvement. In a retrospective study of 22 patients, Wang et al. reported that patients with NF experienced more tenderness on palpation of apparently unaffected adjacent skin compared with clinically affected skin in cellulitis [14]. Therefore, disproportionate pain in a superficial soft tissue infection remains a surgical hallmark for consideration of NF in spite of limited data on sensitivity and specificity for the diagnosis [15]. We found no significant differences in the clinical symptoms or signs of NF for both groups, however, the most prevalent features overall were swelling, erythema and disproportionate pain.
We did not report any missed diagnosis of NF in the COVID-19 group based on clinical accruement with the literature reporting rates from 41.0 to 96.0% [13]. The COVID-19 group had higher proportions of SIRS response reflected by significantly elevated biochemical results including double creatinine, triple urea and double lactate levels (Table 3). Khamnuan et al.’s reported age greater than 60 years (relative risk (RR) = 1.39) and elevated creatinine levels (>1.6 mg/dl or > 141.47 μmol/l, RR = 3.06) to significantly predict increased mortality rate in NF [16]. In addition, the LRINEC score is an adjunct biochemical tool that may assist clinicians to identify and stratify the risk of NF in patients with suspected soft tissue infection [8]. The average LRINEC score of all NF patients in our study was 5.9, and it was comparable to that of a systematic review (6.06) [17].
The management of NF consists of broad-spectrum antibiotics, haemodynamic support and early surgical exploration and debridement of necrotic tissue in a timely manner [15]. Emergency surgical debridement should follow within 12–15 h after admission [18], where a delay of debridement greater than 24 h has shown to increase the mortality rate by 9-fold [8, 18]. In a systematic review and meta-analysis, Nawijn et al. found that mortality rate was statistically significantly lower when surgery was performed within 12 h after presentation compared with surgical treatment delayed by >12 h (odd ratio (OR) = 0.41) [7]. In our study, the average time to surgical debridement from initial presentation was 14.7 h without delay between groups, which was comparable to Quah et al.’s mean time to theatre of 16.2 h [19]. However, we identified a significantly longer operative time of 1.3 h in the COVID-19 group compared to the control group. Matsuyama et al.’s retrospective study of 562 patients reported that increased mortality and morbidity rates were associated with duration of surgery of >2 h [20].
We found multiple factors contributing to the increased operative time that included patient’s comorbidities, prolonged anaesthetic and operative setup and COVID-19 tests beforehand [21]. We identified that the COVID-19 group were largely more unwell with multiple comorbidities reflected by a higher proportion of Grade 4 ASA status. Khadabadi et al. [22] and Cuerva et al. [23] have also reported an increase in operative and anaesthetic time during the COVID-19 pandemic for trauma surgeries and caesarean sections, respectively. The mortality rate for the COVID-19 group was significantly greater than its counterparts, where all COVID-19 patients did not survive (50.0% vs 0.0%, P = 0.036). This mortality rate was substantially higher than that of recent studies from the pre-COVID era ranging from 5.8 to 25.8% [19, 24–26].
To the best of our knowledge, this is the first focused analysis on NF in Australia during COVID-19 and internationally that highlights the importance of timely diagnosis and lockdown measures. There were several limitations in the study principally related to conceivable retrospective bias and a small population sample reflective of the rare disease. A multiple centre study with a larger population size would add further insight into how the nature of the clinical presentations has changed.
CONCLUSION
The COVID-19 pandemic delayed presentations of NF to hospital likely influenced by the lockdown and changes in health seeking behaviours. A sound understanding of public knowledge of the virus and delayed presentations due to prioritization of other symptoms impacted timing of presentations with significantly increased operative time, greater ICU admissions and a higher mortality rate.
ACKNOWLEDGMENTS
We would like to extend our acknowledgement to the emergency and general surgical department at Bankstown-Lidcombe hospital and the medical administration who assisted in the collection of the data.
AUTHORS' CONTRIBUTIONS
The authors contributed to the conception and design of the manuscript, revised it critically for important intellectual content, approved the final version to be published and agreed to be accountable for all aspects of the work.
CONFLICT OF INTEREST STATEMENT
No authors have any competing interests. There is no source of financial or other support and no financial or professional relationships, which may pose a competing interest.
FUNDING
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
ETHICAL APPROVAL
This project has been approved by the Human Research Ethics Committee with no restrictions (reference, 2021/ETH0746).



