-
PDF
- Split View
-
Views
-
Cite
Cite
Yahya Alwatari, Renato Roriz-Silva, Roel Bolckmans, Guilherme M Campos, Intrathoracic sleeve gastrectomy migration with gastric volvulus treated with laparoscopic repair and conversion to gastric bypass, Journal of Surgical Case Reports, Volume 2020, Issue 8, August 2020, rjaa234, https://doi.org/10.1093/jscr/rjaa234
Close - Share Icon Share
Abstract
A 43 years old female with laparoscopic sleeve gastrectomy (SG) and an ‘anterior’ hiatal hernia repair 11 years ago, presented with 3 years history dysphagia and heartburn. Upper gastrointestinal barium showed an almost complete intrathoracic migration of the SG with a partial organoaxial volvulus. Upper endoscopy revealed a 10 cm hiatal hernia with grade B esophagitis. Laparoscopic revision surgery with reduction of the gastric sleeve, standard posterior hiatal hernia repair, resection of the narrowed remnant of the SG and conversion to a gastric bypass was performed. No postoperative complications occurred. The patient is asymptomatic at 2 years of follow-up. We present the technical standards for the management and discuss the suspected pathophysiology of this rare but challenging condition.
INTRODUCTION
In the last decade, sleeve gastrectomy (SG) has become the most commonly performed bariatric surgery in the USA and worldwide [1]. It has been estimated that 1.9 million patients underwent bariatric surgery in the USA between 1993 and 2016 with 113 thousand SG in 2016 alone [2]. While complications after SG are rare, from 25 to 75% of patients are found to have de novo or persistent gastroesophageal reflux disease (GERD) with or without esophageal mucosal injury after SG [3]. Thus, strategies to prevent and reduce GERD rates after LSG are being actively sought. One commonly quoted technical modification is to search for and repair esophageal hiatal hernias (HH) [4]. However, recurrences can occur that may result in the migration of the SG to the chest [5]. We report a case of almost complete intrathoracic SG migration with a partial organoaxial volvulus, erosive esophagitis, successfully managed with laparoscopic hiatal hernia reduction and repair, distal gastrectomy and conversion to Roux-en-Y gastric bypass.
CASE REPORT
Patient is a 43-year-old female with past medical history of a SG and an ‘anterior’ hiatal hernia repair at an outside facility 11 years ago for a body mass index (BMI) of 74.3 Kg/m2 at index SG. The reported hiatal hernia repair at index SG involved approximation of the right and left crura with one stitch anterior to the esophagus without a formal standard posterior or mediastinum esophageal dissection. She presented to our clinic with 3 years history of progressive dysphagia and heartburn. Preoperative upper gastrointestinal (UGI) series and computed tomography (CT) scan demonstrated the migration of almost all of the sleeved stomach into the thoracic cavity with a partial organoaxial volvulus (Fig. 1 A and B). UGI endoscopy showed 10 cm hiatal hernia with intrathoracic migration of the SG with esophagitis grade B (Los Angeles classification).
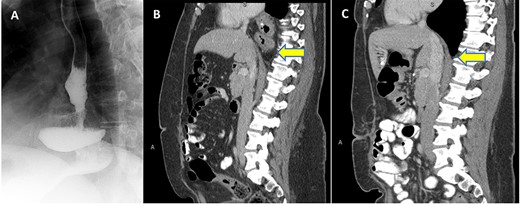
(A) UGI series with SLG in an intrathoracic position; (B) Preoperative CT scan in sagittal view with evidence thoracic migration of the stomach with partial organoaxial volvulus; (C) CT scan 1 year postoperatively confirming intact repair.
The patient underwent an elective laparoscopic surgical revision. Intraoperatively, majority of the SG had herniated into supradiaphragmatic position through the diaphragmatic esophageal hiatus and had a partial organoaxial volvulus (Fig. 2). Lysis of adhesion was carried with full mobilization of the SG and distal esophagus were achieved. The intrathoracic SG was reduced followed by the excision of the hernia sac with preservation of anterior and posterior vagus nerves and the left gastric artery (Fig. 3). The esophageal hiatus diaphragmatic defect was closed with four figure of eight stitches posterior to the esophagus using braided zero polyester (Fig. 4A). A relative narrowing of the mid portion of the SG at the incisura and an excess of gastric fundus was present. A 4 cm long gastric pouch was created in a standard fashion and the remnant of the SG was excised (Fig. 4B and C). Then a standard jejuno-jejunostomy (J-J) was done with a biliopancreatic limb of 60 cm and alimentary limb of 100 cm and the gastro-jejunal anastomosis was created using a 25 mm/4.8 mm circular stapler and reinforced with Lambert sutures of 3–0 polyglactin (Fig. 4D). The J-J and Peterson mesenteric defects were closed. Intraoperative leak test was negative.
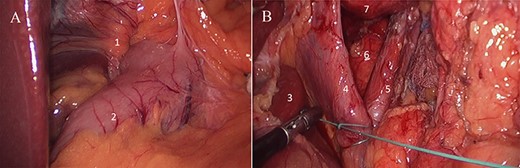
(A) Laparoscopic view of gastric sleeve herniation through esophageal hiatus (1. right crura of diaphragm covered by gastro-hepatic ligament just lateral to esophageal hiatus, 2. gastric antrum); (B) laparoscopic view of dissected esophageal hiatus (3. caudate lobe, 4. right crura of diaphragm, 5. left crura of diaphragm, 6. aorta, 7. distal esophagus).
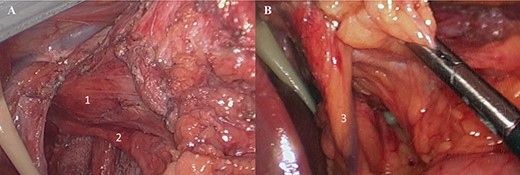
(A) Laparoscopic view (1: distal esophagus, 2. posterior vagus nerve); (B) 3. left gastric artery.
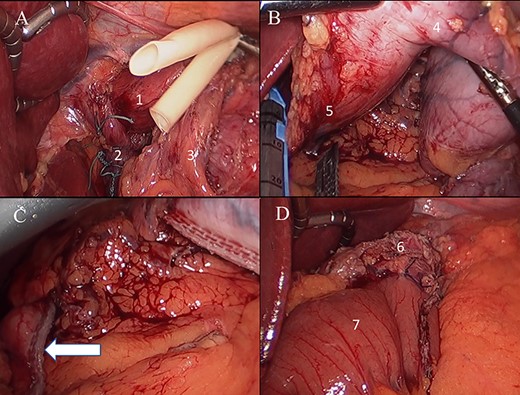
(A) Laparoscopic view posterior hiatal hernia repair (1: distal esophagus, 2: final aspect closed esophageal hiatus, 3: left gastric artery); (B) excision of gastric sleeve remnant (4. sleeve remnant, 5. fist portion of duodenum); (C) duodenal stump staple line; (D) final aspect of gastrojejunostomy (6: gastric pouch, 7: alimentary limb).
The recovery was uneventful and the patient was discharged home on postoperative day 3. The patient was symptoms free upon 2 years follow-up with BMI of 33. CT scan 1 year postoperatively was performed for symptoms not related to surgery confirmed an intact repair (Fig. 1C).
DISCUSSION
With the increasing utilization of SG, more patients with relatively common side effects such as GERD, and other somewhat rare anatomical complications such as strictures, ulcerations and HH have been observed [6]. Small esophageal hiatus sliding herniation of the SG are thought to be associated with post-SG GERD, thus repair of the HH at the index surgery has been advocated by many [4]. There have been reports of acute intrathoracic migration and/or incarceration of the SG which necessitates emergent revision and surgical repair in the early postoperative period [7,8]. Gastric narrowing, progressive enlargement of the esophageal hiatal orifice, division of natural attachments such as the phreno-esophageal membrane and the removal of gastric fundus in close proximity to the angle of His, postoperative weight loss and decrease in the visceral fat and are possible factors associated with the intrathoracic migration of the stomach [9]. Our patient, likely experienced a slow migration of the SG, as the patient was asymptomatic for 8 years after SG. We found only two case reports and one case series of three patients with a complete intrathoracic SG migration [9]. Our case was unique with the recurrence of a large hiatal hernia with chronic intrathoracic migration. Dysphagia can be associated of GERD symptoms and likely increases in the symptoms’ in patients with migrated SG [5].
Of note, our patient had an ‘anterior’ hiatoplasty during the index procedure, which we consider an inadequate repair of a hiatal herniation. Some authors have suggested that previous hiatal hernia repair might accelerate postoperative SG migration into the chest cavity given hiatal dissection which can lead phreno-esophageal membrane disruption, gastric sleeve instability and associated loss of the antireflux mechanism [5]. We believe that a formal and complete anterior, posterior and mediastinal esophageal mobilization should be done for an appropriate hiatal hernia repair, as to ensure appropriate abdominal esophageal length without tension and allow for adequate crural closure posterior to the esophagus at the index SG. However, if a SG migrates into the chest with accompanying GERD, the repair should always involve a complete esophageal mobilization, hiatal hernia repair and we propose that a conversion to a gastric bypass, if possible, is indicated and will be more durable and effective in controlling GERD than hiatal hernia repair alone. An esophageal mobilization and hiatal hernia repair alone in cases of GERD and HH after SG is likely to fail given the tubular nature of SG.
CONCLUSION
Patient with SG can present with intrathoracic sleeve migration even in the absence of peri-operatively recognized hiatal hernia. Several factors are associated with increased risk of sleeve migration post SG. Laparoscopic reduction and repair of the hiatal hernia and conversion of SG to gastric bypass is a valuable option for the treatment of reflux and dysphagia symptoms in patients with intrathoracic SG migration.
AUTHORS’ CONTRIBUTIONS
Drs Y.A., R.R.-S. and R.B. were responsible for drafting the report and conducting literature review. Dr. G.M.C supervised writing manuscript. All authors contributed to drafting the manuscript and/or revising it critically.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.



