-
PDF
- Split View
-
Views
-
Cite
Cite
Mishalle Rashid, Daniel Rashid, Ahan L Hunter, Robotic-assisted laparoscopic repair of a symptomatic uterine isthmocele following cesarean delivery: a case report, Journal of Surgical Case Reports, Volume 2026, Issue 4, April 2026, rjag288, https://doi.org/10.1093/jscr/rjag288
Close - Share Icon Share
Abstract
A uterine isthmocele, or cesarean scar defect, is a complication of cesarean delivery. This report presents a 36-year-old female patient with chronic pelvic pain and uterine bleeding years after a cesarean delivery. Initially, conservative treatment did not alleviate these symptoms. Upon failing conservative measures for symptom relief, she elected to undergo a definitive surgical repair. The patient underwent diagnostic hysteroscopy followed by robotic-assisted laparoscopic repair. Intraoperative evaluation revealed a 2–3 cm niche at the lower uterine segment, which was excised and reconstructed in two layers. Bilateral tubal patency and a watertight closure were confirmed with methylene blue instillation. The procedure was completed without complications and with minimal blood loss. Postoperative recovery was uncomplicated, and the patient reported early symptom improvement. This case demonstrates a minimally invasive surgical technique in a patient with a symptomatic isthmocele and serves to inform osteopathic practitioners of considerations when encountering patients with post-cesarean sequelae.
Introduction
Uterine isthmocele is a defect in the myometrium at the site of a previous cesarean incision and has been documented in 24%–70% of patients depending on imaging modality and diagnostic criteria, based on studies evaluating several hundred post-cesarean patients [1]. Although frequently asymptomatic, clinically significant defects may lead way to abnormal uterine bleeding, chronic pelvic pain, or secondary infertility [2]. The underlying mechanism is thought to involve inadequate myometrial healing and the development of a pouch-like indentation that retains menstrual blood [3].
Single-layer uterine closure, retroflexed uterine position, multiple cesarean deliveries, low transverse incisions, and other variations in surgical technique are associated risk factors [4, 5]. Double-layer closure has been associated with improved myometrial approximation and reduced niche formation [6]. Transvaginal ultrasonography and saline infusion sonohysterography are the most frequently used methods in order to evaluate the depth, width, and thickness of residual myometrium in the niche [7, 8]. Lower residual myometrial thickness, often cited at <3 mm, is often associated with higher symptom intensity [9].
Management options include hormone or surgical treatment. Hysteroscopic repair can be considered in smaller defects with existing uterine wall thickness, but defects in deeper regions with a thin (<3 mm) residual myometrial thickness (RMT) may need laparoscopic or robotic repair [10, 11]. Despite multiple operative approaches described in the literature, the optimal surgical technique for symptomatic isthmocele remains debated, particularly in patients with significant myometrial thinning. Additionally, robotic repair offers better vision, dexterity, and accuracy for complete removal and repair [12, 13].
This case report describes a patient with a symptomatic isthmocele refractory to conservative management and highlights the role of robotic-assisted laparoscopic repair as a minimally invasive option in this clinical setting.
Case report
The patient was a 36-year-old White woman presenting with chronic pelvic pain and abnormal uterine bleeding. Her menstrual cycles came every 28 days, with bleeding lasting 4–5 days followed by spotting and cramping. She also detailed intermittent right-sided pelvic pain. Symptoms gradually worsened despite the use of combined oral contraceptives.
History and examination
Past medical history included anxiety, major depressive disorder, insomnia, asthma, right ovarian cyst, and chronic pelvic pain. Surgical history included one low transverse cesarean delivery in 2020 and a benign cystoscopy in 2021. She was G1P1, sexually active, and used oral contraceptives. She smoked daily and denied alcohol or illicit drug use. Vital signs were normal. The abdomen was soft and non-tender. Pelvic examination findings were unremarkable in prior evaluations. Transvaginal US was unremarkable (Fig. 1).
Transvaginal ultrasound image of uterine cavity prior to isthmocele identification during laproscopic procedure.
Diagnostic evaluation
Evaluation suggested a symptomatic lower uterine segment defect consistent with isthmocele. Definitive assessment was planned through hysteroscopic and laparoscopic visualization.
Surgical procedure
On 18 March 2025, the patient underwent diagnostic hysteroscopy followed by robotic-assisted laparoscopic repair under general anesthesia. The hysteroscopy showed a defect in the lower uterine segment typical of a niche. Robotic laparoscopy was then performed with infraumbilical Veress entry and 8 mm trocar placement.
Pelvic survey revealed normal adnexa aside from the defect. A bladder flap was created, exposing a 2–3 cm indentation at the prior hysterotomy site. The defect was excised using cold scissors and electrocautery, and the specimen was removed in an EndoCatch bag.
The uterine wall was layered using two layers of 2-0 V-Loc barbed sutures. Transcervically, methylene blue dye was injected, which demonstrated the absence of leakage and bilateral tubal patency. Port sites were closed with 4-0 Monocryl. Estimated blood loss was 15 mL. No complications occurred.
Postoperative course
Recovery was uncomplicated. No postoperative imaging or additional diagnostic testing was performed following surgical repair. The patient was advised to avoid intercourse, strenuous activity, and heavy lifting for 2 weeks. She reported improved pelvic discomfort and reduced abnormal bleeding at 2-week postoperative follow-up.
Discussion
In the context of this patient’s presentation and management course, previously described characteristics of uterine isthmocele help frame the rationale for operative intervention. Uterine isthmocele is a postoperative complication of cesarean delivery associated with abnormal uterine bleeding, chronic pelvic pain, and infertility [14]. The pathophysiology is related to impaired myometrial healing that causes a pouch-like indentation in the anterior wall of the uterus.
Single-layer closure, retroflexed uterine position, and multiple prior cesarean deliveries are risk factors that enhance the likelihood of niche formation. Transvaginal ultrasonography is most often employed in diagnostic evaluation, with adjunct techniques used to further characterize niche morphology [7, 8, 15].
Operative management may be necessary in symptomatic patients. Small, shallow defects are amenable to hysteroscopic resection, but deeper defects or those with thin myometrium will require laparoscopic or robotic reconstruction. Robotic-assisted repair offers improved visualization and allows for precise complete excision of the defect with a layered closure.
Uterine isthmoceles have significant implications for future reproductive planning and obstetric risk beyond symptom burden, which represents further rationale for accurate diagnosis and appropriate management [1]. Defects involving considerable myometrial atrophy have also been linked with cesarean scar ectopic pregnancy, uterine rupture in subsequent gestations, and improper placentation, including disorders of the placenta accreta spectrum [4].
These are factors show the utility of restoring myometrial status for those desiring future fertility or experiencing symptoms [6]. While abnormal uterine bleeding and pelvic pain were this patient's main reason for surgical intervention, definitive surgical management also incorporates a preventative strategy for potential future sequelae related to subsequent cesarean scar defects if left uncorrected [1, 3].
Robotic-assisted laparoscopic management also enables precise fibrosis excision and subsequent layered myometrial repairs and reconstructions, potentially optimizing future uterine strength and minimizing future complications related to pregnancy or parturition [12–14]. While not formally examined in this case, this surgical management also incorporates a plan established in other studies demonstrating utility in optimizing existing residual myometrial tissue and existing uterine contour [6, 15].
These factors become essential in clinical decision making for osteopathic physicians involved in women’s health, minimally invasive gynecologic surgery, and perioperative care, as isthmocele represents not only a source of current symptoms but also a condition with meaningful implications for future gynecologic and obstetric health [1].
Clinical outcomes following minimally invasive scar defect repair are favorable, with improvement in abnormal bleeding, pelvic pain, and fertility outcomes [7]. This case corresponds to such findings and provides intraoperative visualization (Fig. 2) that show defect morphology and the reconstructive technique.
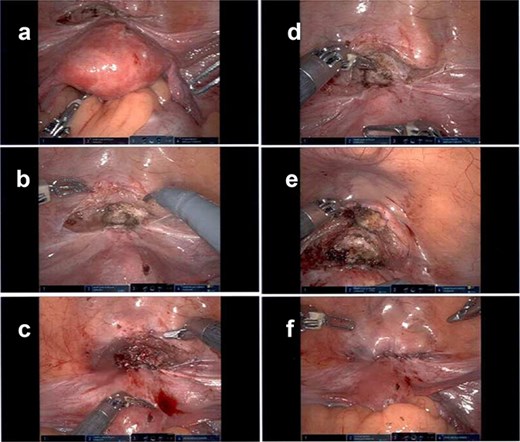
Laparoscopic intraoperative images demonstrating identification and repair of a uterine isthmocele defect; the figure depicts initial visualization of the isthmocele defect (a), closer inspection showing thinning of the myometrium (b), dissection and delineation of the defect margins (c), excision of fibrotic tissue within the defect (d), primary closure of the defect with suturing (e), and final appearance following complete repair (f).
Acknowledgements
The authors would like to thank the staff at About Women OB/GYN and Sentara Northern Virginia Medical Center for their support during data collection and patient care.
Conflicts of interest
None declared.
Funding
None declared.



