-
PDF
- Split View
-
Views
-
Cite
Cite
Simone Oliver Senica, Marko Bogovic, Katja Schmidhofer, Bernhard Kienesberger, Gabriele Gritsch-Olipp, Johannes Schalamon, Rising detection of congenital pulmonary airway malformations and a pragmatic institutional algorithm: a four-patient thoracoscopic case series, Journal of Surgical Case Reports, Volume 2026, Issue 4, April 2026, rjag260, https://doi.org/10.1093/jscr/rjag260
Close - Share Icon Share
Abstract
Congenital pulmonary airway malformations (CPAM) are rare lung anomalies increasingly detected through prenatal imaging. Management of asymptomatic patients remains controversial, particularly regarding imaging strategy, timing of surgery, and malignancy risk. We report a four-patient case series treated at a regional pediatric surgical center between February and June 2025, representing an increased local incidence. All patients were asymptomatic postnatally and underwent thoracic computed tomography at a mean age of 13 months. Elective thoracoscopic lower lobectomy was performed in all cases at a mean age of 18.3 months. Histopathology revealed CPAM type 2 in three patients and CPAM type 4 in one asymptomatic child. Two postoperative pneumothoraces required temporary drainage; no long-term complications occurred. Our findings support elective thoracoscopic resection for confirmed CPAM. The detection of CPAM type 4 highlights the limitations of radiologic subtype differentiation, supporting early surgery in asymptomatic patients. We propose a management algorithm applicable to secondary centers.
Introduction
Congenital pulmonary airway malformations (CPAM), formerly known as congenital cystic adenomatoid malformations, constitute a spectrum of rare developmental anomalies of the lower respiratory tract resulting from abnormal branching of the tracheobronchial tree during fetal lung development. The incidence was previously reported to be ~1 in 30 000 live births, although recent studies suggest that the true prevalence may be higher, since the numbers are rising due to improved prenatal detection [1].
With the widespread use of high-resolution prenatal ultrasonography and increasing availability of fetal magnetic resonance imaging (MRI), most CPAM are now diagnosed antenatally [2]. However, postnatal confirmation remains essential for definitive diagnosis and surgical planning, most commonly by computed tomography (CT) [1, 3]. Only 10%–20% of neonates present with respiratory distress at birth; the majority remain asymptomatic during infancy [4].
The management of symptomatic CPAM is undisputed and requires surgical resection. However, the optimal strategy for asymptomatic patients remains highly controversial [1, 5]. Conservative management with observation has been proposed by some authors, whereas others recommend elective resection to prevent infection, pneumothorax, and malignant transformation [1, 6]. In particular, CPAM type 4 is associated with pleuropulmonary blastoma, a highly malignant pediatric tumour [7]. Differentiation between CPAM subtypes based solely on imaging remains unreliable. Histological differentiation is still the gold standard for this purpose [8].
Between February and June 2025, our institution diagnosed and treated four cases of CPAM, corresponding to a notable increase in local detection. This prompted the establishment of a structured institutional diagnostic and therapeutic pathway. The aims of this study are to report this regional clustering, to describe our postnatal imaging strategy, to demonstrate the safety and feasibility of universal thoracoscopic management in asymptomatic patients, and to emphasize the limitations of radiologic subtype prediction through histopathological correlation.
Materials and methods
This retrospective case series includes all patients with a prenatal diagnosis of CPAM who underwent surgical treatment at the Department of Pediatric and Adolescent Surgery, LKH Klagenfurt, between February and June 2025. LKH Klagenfurt is a regional referral center for pediatric surgery serving a predominantly rural population. The study was conducted in accordance with institutional ethical standards and the Declaration of Helsinki. Informed consent was waived due the retrospective nature of this work. Data were extracted from electronic medical records and included prenatal imaging, gestational age, birth weight, lesion localization, postnatal imaging, clinical course, surgical details, histopathological results, postoperative complications, length of hospital stay, and follow-up outcomes.
Case series
Patient characteristics and prenatal diagnosis
Four children (three males, one female) were included. Mean gestational age at birth was 37 weeks (range 36–39 weeks), and mean birth weight was 3100 g (range 2900–3400 g). All four patients had been diagnosed prenatally by routine obstetric ultrasound. Fetal MRI was performed in all cases and supported the diagnosis of CPAM.
Lesions were localized in the right lower lobe in three patients and in the left lower lobe in one patient. No patient showed signs of fetal hydrops, mediastinal shift, or polyhydramnios. In utero interventions were not required.
Postnatal clinical course and imaging strategy
All neonates were clinically asymptomatic after birth. There were no episodes of respiratory distress, desaturation, or feeding difficulties. Cardiopulmonary examinations were normal in all patients. Routine postnatal chest radiography was unremarkable in all four infants, with no visible cystic lesions or lung asymmetry. These findings confirmed the limited diagnostic value of conventional radiography in asymptomatic CPAM. Definitive postnatal imaging was performed using contrast-enhanced chest CT at a mean age of 13 months (range 11–15 months). CT imaging provided clear visualization of the cystic malformations, precise lobe localization, and detailed vascular anatomy (Fig. 1). All CT-scans were performed under conscious sedation using oral midazolam. Postnatal MRI was deliberately avoided to prevent the need for general anesthesia in infancy, as its additional diagnostic benefit over CT was considered limited for surgical planning.
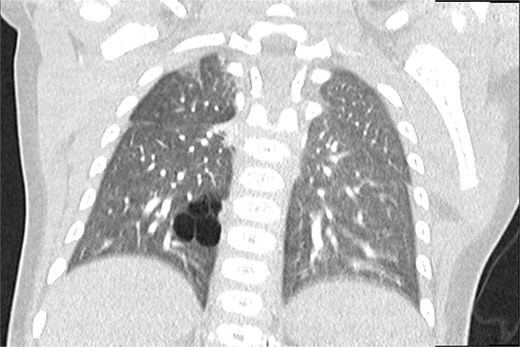
Chest CT (coronal plane) showing a CPAM in the lower lobe of the right lung.
Surgical technique
All patients underwent elective thoracoscopic lower lobectomy at a mean age of 18.3 months (range 16–21 months). Procedures were performed under general anesthesia with selective single-lung ventilation. The thoracoscopic approach included three ports placed under direct visualization. Careful dissection was performed to identify and ligate the segmental pulmonary artery branches, pulmonary veins, and bronchus of the affected lobe (Fig. 2). The specimen was retrieved in an endoscopic bag, hemostasis was verified, and a chest drain was placed in all cases. No conversions to thoracotomy were required. Mean operative time was 110 minutes (range 95–135 minutes). There were no intraoperative complications.
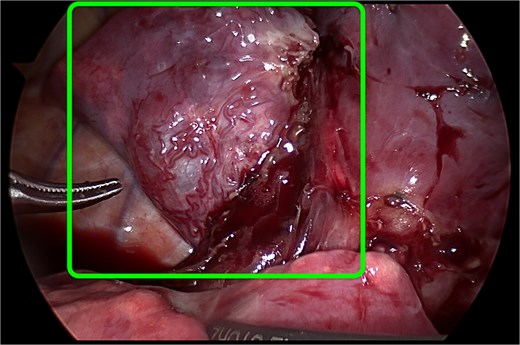
Thoracoscopic view of the CPAM in the lower lobe of the right lung. The malformation is highlighted by the green contour.
Histopathological findings
Histopathological examination confirmed CPAM type 2 in three patients. One asymptomatic child was diagnosed with CPAM type 4. No malignant transformation was detected in any specimen (Fig. 3).
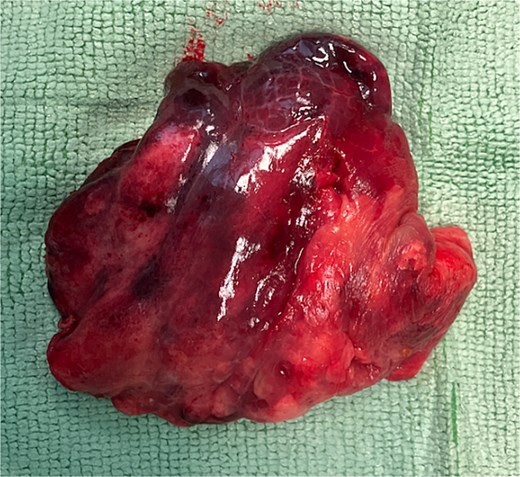
Postoperative course and follow-up
Two patients showed signs of a mild leakage from the surgical site requiring continued chest drainage for 4 and 6 days, respectively. The remaining two patients had uneventful postoperative courses without any detectable airflow and the chest drains could be removed on day two after surgery. The mean hospital stay was 7 days (range 5–10 days).
All patients were followed clinically and radiologically. At a mean follow-up of 9 months, all children were asymptomatic with no respiratory complaints, normal growth, and normal physical examination. Chest radiographs at follow-up showed full lung re-expansion.
Discussion
This case series provides several clinically relevant observations: a regional increase in CPAM detection, successful implementation of a pragmatic postnatal imaging strategy, consistent use of thoracoscopic resection in asymptomatic patients, and histopathological confirmation of CPAM type 4 in an asymptomatic child.
Regional increase in CPAM detection
Within a five-month period, four cases of CPAM were diagnosed and treated among ~4000 births in our catchment area, resulting in an incidence of ~1:1000. This rate appears higher than traditionally reported figures; however, given the small sample size and limited observation period, this finding should be interpreted with caution. It remains unclear whether this reflects a true epidemiological increase or is attributable to improved prenatal imaging and heightened diagnostic awareness. While our experience may suggest the potential value of structured institutional management pathways, these observations are preliminary and should be validated in larger cohorts before broader recommendations can be made [1].
Role of prenatal MRI
Although fetal MRI was performed in all patients in our series, it is not included as a routine step in our proposed algorithm. This reflects a shift in our practice, as prenatal MRI is not justified in all suspected CPAM cases. High-quality ultrasound is usually sufficient for diagnosis, while MRI should be reserved as an adjunct in selected cases with complex, atypical, or equivocal findings [9]. Based on both our experience and the available literature, we have therefore refined our approach to reserve fetal MRI for selected cases rather than incorporating it as a standard component of the diagnostic pathway.
Postnatal imaging: CT versus MRI
Postnatal diagnostic confirmation is essential for surgical planning and exclusion of differential diagnoses such as bronchopulmonary sequestration [3]. Chest radiography is widely available but has insufficient diagnostic sensitivity, as confirmed in all cases in our cohort.
CT remains the gold standard for postnatal assessment due to its excellent spatial resolution and ability to delineate vascular anatomy. However, radiation exposure in infancy raises legitimate concerns [10]. MRI avoids radiation but almost invariably requires sedation or general anesthesia in infants, introducing additional risk and logistical complexity [11].
Our institutional strategy involves a single CT examination at ~1 year of age in asymptomatic patients. This timing minimizes radiation exposure while ensuring reliable diagnosis and optimal surgical planning.
Management of asymptomatic CPAM
The management of asymptomatic CPAM remains one of the most debated topics in pediatric thoracic surgery [1]. Advocates of conservative management cite the low rate of early complications and wish to avoid surgical morbidity. However, standardized surveillance protocols are lacking, and there is no reliable method to predict which lesions will become symptomatic.
Casagrande et al. demonstrated that the risk of malignant degeneration cannot be eliminated by expectant observation [6, 12]. Moreover, delayed surgery may be technically more difficult due to recurrent infections, adhesions, and inflammatory changes [13].
Thoracoscopic lobectomy as standard therapy
Minimally invasive thoracic surgery has become the preferred approach for CPAM resection. Thoracoscopy offers superior visualization, reduced postoperative pain, shorter hospital stays, and excellent cosmetic and functional results [14].
In our cohort, thoracoscopic lobectomy was completed successfully in all cases without conversion. The low complication rate and favorable outcomes support the safety and reproducibility of this technique even in small-volume regional centers.
Radiologic–pathologic discrepancy and malignancy risk
A critical finding in our series was the identification of CPAM type 4 in a completely asymptomatic child. Type 4 CPAM is known to share histologic features with pleuropulmonary blastoma and is associated with the highest malignancy risk among CPAM subtypes.
Reliable radiologic differentiation between CPAM type 1 and type 4 remains difficult. Zeng et al. and Leblanc et al. have both highlighted the limitations of imaging-based subtype classification [15–17]. Our finding provides real-world confirmation of this limitation and strengthens the argument for early elective resection.
Considerations regarding follow up
Our surgical strategy included lobectomy, thereby minimizing the risk of malignancy through complete resection. Long term follow-up by chest x-ray is not recommended due to the limited sensitivity of radiography in detecting residual lesions. Postoperative CT may be useful in cases of atypical resection and or when histopathological findings raise suspicion of incomplete tumour removal [18]. Therefore, our protocol does not include routine follow-up imaging using radiographic modalities.
Proposed simplified algorithm for the management of CPAM
Based on our experience, we implemented the following protocol (Fig. 4):
Prenatal period: Clear sonographic diagnosis is sufficient; routine fetal MRI is not mandatory.
Neonatal period: Chest radiography is reserved for symptomatic infants only.
Asymptomatic infants: One CT scan at ~12 months of age.
Definitive treatment: Elective thoracoscopic lobectomy for all confirmed CPAM, regardless of symptoms.
Follow up after surgery: In cases of complete resection, long-term radiographic follow-up is not required. Clinical follow-up until adulthood should be limited to cases with respiratory symptoms.
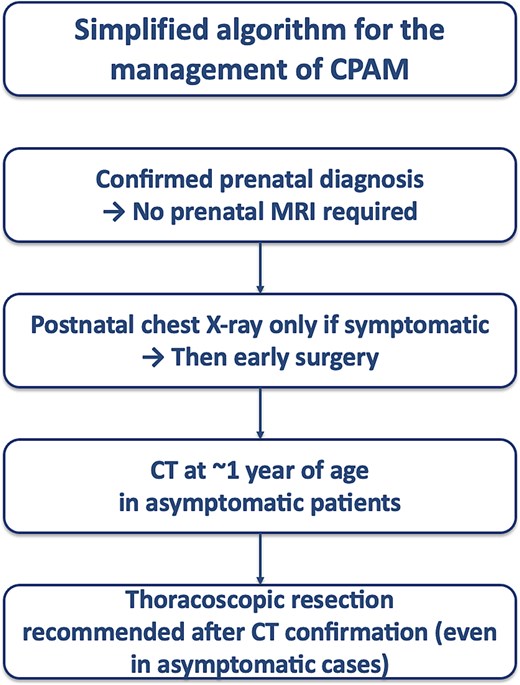
This approach balances diagnostic accuracy, radiation exposure, and anesthetic risk while providing definitive treatment before complications arise.
Limitations
The main limitation of this study is the small sample size inherent to rare congenital diseases. Furthermore, the retrospective design limits statistical analysis. Clinical long-term follow-up into adolescence and adulthood is still pending in our patients. Despite these limitations, the series offers clinically relevant insights due to complete pre- and postoperative documentation and histopathological confirmation.
Conclusion
This four-patient case series demonstrates that a streamlined imaging strategy combined with universal thoracoscopic resection is safe and effective for the management of CPAM, including asymptomatic lesions. The detection of CPAM type 4 in an asymptomatic child underlines the limitations of radiologic classification and it also supports early elective surgery for histological determination of the lesions. The observed regional increase in detection further highlights the need for standardized institutional protocols adaptable beyond large tertiary centers. Larger multicenter studies such as the CONNECT Trial may help refine guidelines and improve risk stratification [19].
Conflicts of interest
None declared.
Funding
None declared.



