-
PDF
- Split View
-
Views
-
Cite
Cite
Charbel G Saad, Naim Slaiby, Gibran Atwi, Elie Azar, Nabil Moukarzel, Adenoid cystic carcinoma of the trachea: an uncommon presentation of a rare malignancy, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag190, https://doi.org/10.1093/jscr/rjag190
Close - Share Icon Share
Abstract
Adenoid cystic carcinoma (ACC) is a rare salivary gland tumor characterized by an indolent yet aggressive course, facilitated by perineural invasion, a propensity for delayed distant metastases, and recurrence after resection. A 61-year-old female presented with a three-month history of intermittent hoarseness and resting dysphagia. Contrast-enhanced computed tomography demonstrated a subglottic mass with proximal tracheal invasion, and ultrasound-guided neck fine-needle aspiration cytology was consistent with ACC. Surgical management included total laryngectomy with proximal tracheal and left thyroid lobe resection, followed by immediate tracheostomy. ACC was confirmed on histopathology with negative margins, and adjuvant radiotherapy (RT) was initiated to reduce locoregional recurrence risk. At one-year follow-up, the patient remains in remission with no evidence of recurrence or metastasis. Early symptom recognition and surgery with adjuvant radiotherapy remain central to local disease control, and long-term surveillance is essential given the potential for late recurrence.
Introduction
Adenoid cystic carcinoma (ACC) is a rare tumor arising from secretory glands. It accounts for 1% of all head and neck malignancies and approximately 10% of all salivary gland tumors [1]. While the major salivary glands are the most common site of origin, ACC can rarely originate in the tracheobronchial tree [2, 3].
Biologically, ACC is characterized by an indolent but relentless course. It is renowned for its tendency toward submucosal spread and perineural invasion, often extending longitudinally well beyond the macroscopically visible tumor [3, 4]. In a recent pooled analysis of 403 patients, positive margins were reported in 73.8% of resected cases, a significantly higher figure than historically appreciated [4].
We report a case of tracheal ACC highlighting these diagnostic pitfalls and the surgical management of complex laryngotracheal invasion.
Case report
A 61-year-old female presented with a 3-month history of intermittent hoarseness, resting dysphagia, and exertional dyspnea. Her medical history included infiltrating ductal carcinoma of the breast (ER+/PR+, HER2-), treated with lumpectomy, radiotherapy, and anastrozole.
Computed tomography (CT) revealed a posteriorly based left subglottic tumor extending inferiorly to the first six tracheal rings (Fig. 1). Endoscopy confirmed the mass encroaching on the tracheoesophageal groove. Given her history, the differential diagnosis included metastatic breast cancer versus a primary tracheal malignancy. Ultrasound-guided fine-needle aspiration of the lesion demonstrated cytology consistent with ACC; definitive diagnosis was confirmed on histopathological examination of the resection specimen.
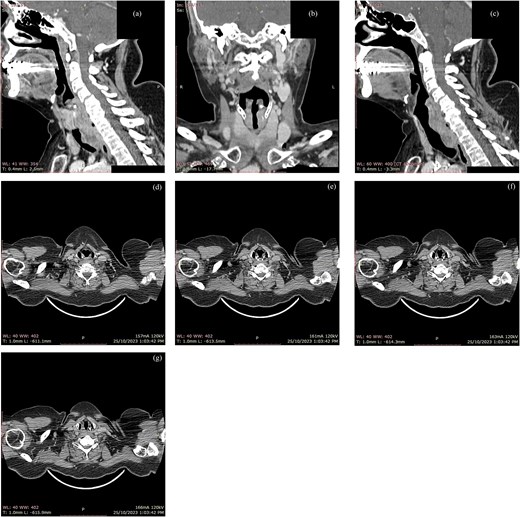
Contrast-enhanced CT scan of the neck demonstrating a left posterior subglottic mass with inferior extension into the proximal trachea. Sagittal (a, b) and coronal (c) reformats illustrate the craniocaudal extent. Sequential axial images (d–g) demonstrate eccentric left posterolateral subglottic/proximal tracheal wall thickening with luminal narrowing and extension into the adjacent left paratracheal soft tissues/tracheoesophageal groove region.
Intraoperatively, the tumor was found to involve the left thyroid lobe and extend significantly into the subglottis. A simple segmental resection with end-to-end anastomosis was deemed insufficient for oncologic clearance due to the extent of invasion. Consequently, a total laryngectomy with resection of the proximal trachea and left thyroid lobe was performed (Fig. 2). Frozen section analysis confirmed negative margins.
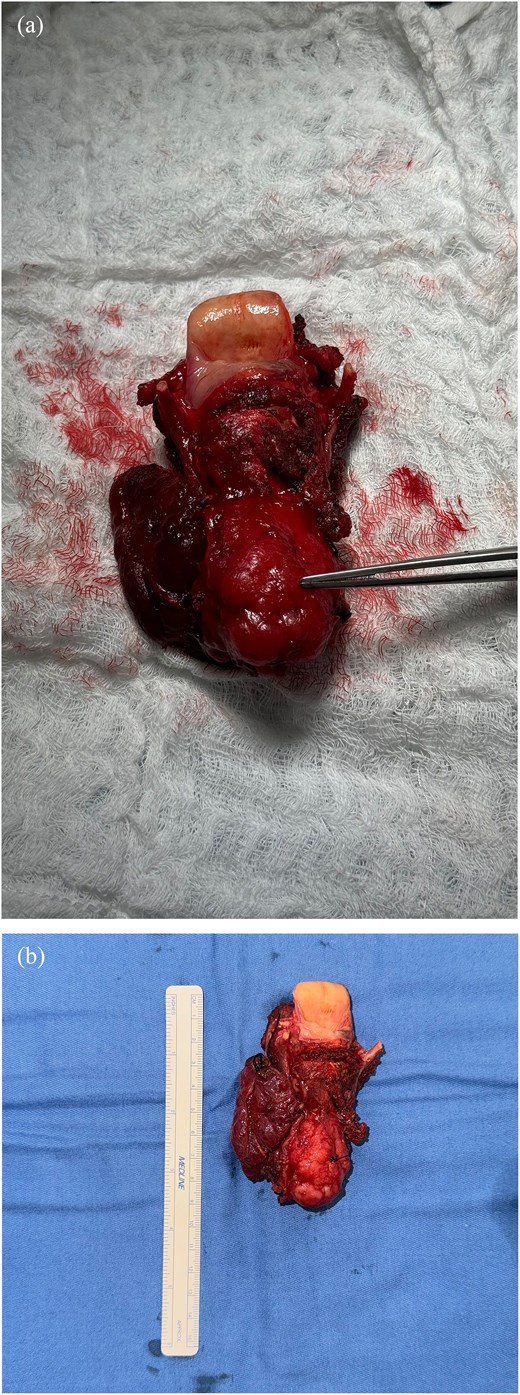
Gross specimen and histopathologic features of resected ACC. (a) Anterior view; (b) posterior view.
Histopathology revealed ACC (pT4a) with perineural invasion (Table 1). The tumor cells were negative for GATA3, ruling out metastatic breast cancer. The closest surgical margin was 8 mm. To mitigate the risk of locoregional recurrence, adjuvant RT was initiated. At one-year follow-up, the patient remains in remission with no evidence of recurrence.
| Focality . | Unifocal . |
|---|---|
| Size | Greatest dimension is 4 cm |
| Grade | Cribriform and solid pattern Grade 2. |
| Site | Infraglottic in the submucosa of the proximal trachea with extension to the thyroid gland and the left vocal cord |
| Extent | pT4a; involving deep extrinsic muscle and thyroid. |
| Margins | Negative |
| The closest surgical margin is the tracheal, with a clearance of 8 mm (including the resected slice for frozen section examination) | |
| Blood or lymphatic vessel invasion | Not identified |
| Perineural invasion | Present, multiple |
| Focality | Unifocal |
|---|---|
| Size | Greatest dimension is 4 cm |
| Grade | Cribriform and solid pattern Grade 2. |
| Site | Infraglottic in the submucosa of the proximal trachea with extension to the thyroid gland and the left vocal cord |
| Extent | pT4a; involving deep extrinsic muscle and thyroid. |
| Margins | Negative |
| The closest surgical margin is the tracheal, with a clearance of 8 mm (including the resected slice for frozen section examination) | |
| Blood or lymphatic vessel invasion | Not identified |
| Perineural invasion | Present, multiple |
Discussion
ACC is a rare but aggressive malignancy known for its indolent course. The 5-year overall survival for tracheal ACC is approximately 65% [2]. One of the main drivers for that is a delayed diagnosis, frequently due to the disease’s nonspecific presentation and tendency to mimic benign airway pathologies. Due to the slow caliber reduction of the airway, patients often present with non-specific symptoms such as dyspnea, hoarseness, wheezing, and intractable chronic cough which may delay diagnosis and treatment [5–9]. Similarly, our patient’s history of symptoms mirrors these reports.
Regarding imaging, FDG-PET/CT is routinely employed for staging, though the metabolic activity of ACC can be variable [6, 10]. Some cases demonstrate avid glucose uptake [9], whereas others exhibit only slightly increased or low-to-moderate metabolism [11, 12]. Emerging evidence suggests that ACCs may be intensely avid on Prostate-Specific Membrane Antigen (PSMA) PET scans; a recent direct comparison revealed a PSMA SUVmax of 12.5 versus an FDG SUVmax of only 3.6, a finding that could revolutionize the detection of skip lesions and distant metastases in patients with low FDG avidity [12].
Surgery remains the mainstay of treatment, performed in approximately 74.5% of cases [2, 4]. The gold standard for curative intent is wide surgical resection with negative margins (R0), which has been shown to offer the best long-term survival outcomes [4, 10]. However, achieving this goal is notoriously difficult due to ACC’s submucosal spread [2, 4]. A distinct and clinically significant pathway for spread is perineural invasion, which allows tumor cells to infiltrate along nerve sheaths, often leading to pain, cranial nerve paralysis, and late recurrences after resection [3, 4, 8].
Consequently, limited approaches such as segmental tracheal resections frequently result in positive margins in up to 73.8% of cases, often necessitating adjuvant therapies to compensate for incomplete excision [4, 11, 13]. Given the high rate of microscopic residual disease, RT is widely advocated for, even in cases with wide resections and negative margins, though only 56.7% of surgical patients receive adjuvant RT [2].
In this case, the tumor extended into the subglottic region and involved the thyroid gland (pT4a). Recognizing that a limited segmental resection would likely result in positive margins given this extensive local invasion, we opted for a more aggressive approach. Total laryngectomy with tracheal resection was performed to ensure complete tumor removal and secure clear margins, aligning with the principle that extensive laryngotracheal involvement often precludes simple resection and demands complex reconstruction for oncologic safety [2, 10].
For unresectable disease, concurrent carboplatin and paclitaxel with radiation (66 Gy) has been shown to achieve complete response and symptom relief in patients with extensive mediastinal involvement [7]. However, this efficacy is likely driven by radiosensitization; chemotherapy alone is generally considered ineffective due to the tumor’s poor sensitivity to cytotoxic agents, with response rates often remaining below 33% [2, 3].
Recent advancements in particle therapy offer new hope. Intensity-Modulated Proton Therapy significantly reduces the integral dose to the breast and lungs compared to photon therapy [6]. Furthermore, Nakamura et al. [14] (2024) reported long-term survival (>7 years) in unresectable cases treated with high-dose Proton Beam Therapy (74 Gy RBE), suggesting its potential as a potent alternative to radical surgery. Personalized Stereotactic Ablative Brachytherapy using Iodine-125 seeds achieved a 100% disease control rate in a salvage setting with a median follow up of 37.5 months [15]. Other forms of brachytherapy, such as intraluminal high-dose-rate (HDR) boosts, have also been successfully employed to control local recurrence [13].
Conclusion
The challenges of managing ACC with tracheal invasion are highlighted in this case report. Owing to its high recurrence rate and erratic behavior, an aggressive approach with surgical resection with RT remains the best evidence-based approach for achieving control of the disease. Long-term surveillance and follow-up are indispensable due to the risk of late recurrence and distant metastases. Novel therapeutical advances for the treatment of ACC remain needed.
Funding
No funding was received for this research.
Conflicts of interest
The authors have no conflicts of interest or disclosures to declare.
Ethical approval
The work presented in this article goes in accordance with the Declaration of Helsinki in 1964.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A signed consent form is held on file by the authors and is available for review by the journal’s editorial team upon request.
References
- neck
- ultrasonography
- computed tomography
- cancer
- deglutition disorders
- fine needle aspiration biopsy for cytology
- hoarseness
- adenoid cystic carcinoma
- follow-up
- laryngectomy
- neoplasm metastasis
- radiotherapy, adjuvant
- salivary gland neoplasms
- surgical procedures, operative
- thyroid
- trachea
- tracheostomy
- adenoid cystic carcinoma of trachea
- surveillance, medical
- metastasis, distant
- recurrence risk
- total laryngectomy
- histopathology tests
- disease remission
- indolent
- perineural invasion



