-
PDF
- Split View
-
Views
-
Cite
Cite
Galia Jadkarim, Raghad H Alsharif, Fatmah Kahtani, Meral Altayeb, Faiz Zagzoog, Shaza Alsharif, Rana Ajabnoor, Malignant adenomyoepithelioma of the breast: a rare case report and literature review, Journal of Surgical Case Reports, Volume 2026, Issue 1, January 2026, rjaf1039, https://doi.org/10.1093/jscr/rjaf1039
Close - Share Icon Share
Abstract
Adenomyoepithelioma (AME) of the breast is a rare biphasic tumor composed of epithelial and myoepithelial elements, with malignant transformation being exceptionally uncommon. We report a case of pure malignant adenomyoepithelioma (M-AME) in a 51-year-old woman who presented with a gradually enlarging left breast mass. Imaging revealed a 3.6 cm lesion, and core biopsy showed biphasic proliferation with myoepithelial atypia and high mitotic activity. Wide local excision with oncoplastic reduction achieved negative margins, and histopathology confirmed pure M-AME (pT2 pN0), triple-negative for estrogen, progesterone, and Human Epidermal growth factor Receptor 2, with a Ki-67 index of 35%–40%. The patient received adjuvant radiotherapy and remains disease-free at 1 year. This case highlights the diagnostic challenges of M-AME, underscores the importance of immunohistochemistry and complete excision with negative margins, and adds to the limited literature on this exceptionally rare breast neoplasm.
Introduction
Adenomyoepithelioma (AME) of the breast is an uncommon biphasic tumor composed of both epithelial and myoepithelial elements. Although typically benign, AME has the potential for malignant transformation in one or both cellular components, giving rise to malignant adenomyoepithelioma (M-AME) [1, 2].
According to a recent systematic review by Chen et al. [3], only 167 cases of M-AME have been documented in the literature, underscoring its extreme rarity, and the limited understanding of its biological behavior [4]. Because of this scarcity, establishing accurate diagnosis and optimal management strategies remains difficult. Current evidence is mainly derived from individual case reports and small series.
Surgical excision with negative margins continues to be the cornerstone of treatment, as lymph-node involvement and distant metastasis are relatively uncommon [1, 5, 6].
Here, we present a case of pure malignant adenomyoepithelioma, not associated with conventional invasive ductal carcinoma of no special type, incidentally, detected during radiologic evaluation performed prior to reduction mammoplasty. The aim of reporting this case is to contribute to the limited body of literature on this exceptionally rare tumor and to share our experience in broadening the understanding of its clinicopathological characteristics.
Case presentation
A 51-year-old multiparous woman, postmenopausal for 3 years, with type 2 diabetes mellitus on oral agents, hypertension, dyslipidemia, and obesity (preoperative body mass index (BMI) 46 kg/m2; post-laparoscopic sleeve gastrectomy BMI 38 kg/m2), was referred from a primary healthcare center to our breast surgery clinic for evaluation of a left breast lump that had been noticed 6 months earlier. The mass had gradually increased in size and was occasionally associated with mild pain when lying on the left side. She denied nipple discharge, skin changes, fever, weight loss, or other systemic symptoms. She was a nonsmoker with no family history of breast cancer.
Her gynecologic history included menarche at 11 years and first pregnancy at 16 years. She had six full-term deliveries, breast-fed only her first child for 1 month, used oral contraceptive pills after each pregnancy, and had never received hormone replacement therapy. Previous surgeries included two cesarean sections, open cholecystectomy at age 17, and laparoscopic sleeve gastrectomy in October 2023.
Physical examination revealed large, pendulous breasts (bra size 46, double D cup) with grade III ptosis. The left breast contained a firm, hard, mobile 4 × 4 cm mass in the upper outer quadrant, not fixed to skin or muscle. The nipple–areola complex was normal, and no skin changes were observed. The left axilla contained mobile lymph nodes, while the right breast and axilla were normal.
Breast ultrasound showed a fatty background with a 3.6 × 3.3 × 3.0 cm round, heterogeneous hypoechoic mass at 2 o’clock with indistinct margins and internal vascularity, and a single prominent but benign appearing left axillary node (Fig. 1A and B). Mammography revealed almost entirely fatty parenchyma (ACR A) with a hyperdense left upper outer quadrant mass, without calcifications or architectural distortion (BI-RADS 4C on the left, 1 on the right) (Fig. 2A and B).
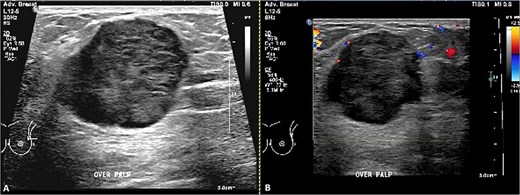
Ultrasound of the left breast. (A) Grayscale and (B) color Doppler images demonstrate a vertically oriented, microlobulated, heterogeneously hypoechoic mass at the 3 o’clock position in the distal left breast, showing peripheral rim vascularity.
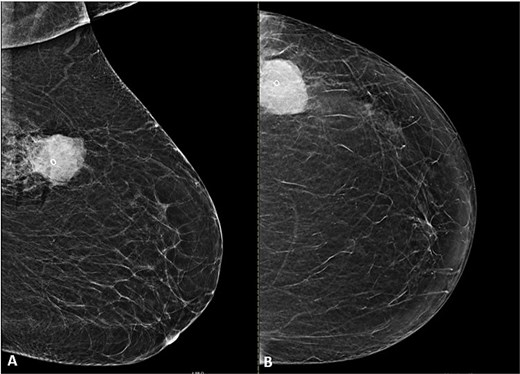
Mammogram of the left breast. (A) Craniocaudal (CC) and (B) mediolateral oblique (MLO) views demonstrate a high-density, microlobulated mass containing a post-biopsy clip with surrounding post-procedural changes.
Core needle biopsy demonstrated a biphasic epithelial and myoepithelial proliferation in a fibromyxoid background with prominent myoepithelial atypia, including enlarged hyperchromatic nuclei and frequent mitoses (Fig. 3A–C). Immunohistochemistry showed positivity for p63, calponin, CK5/6, SOX10, and S100 in the myoepithelial component, and CK7 positivity in both luminal epithelial and myoepithelial cells, more prominent in the luminal cells (Fig. 3D). Estrogen (ER), progesterone (PR), and Human Epidermal growth factor Receptor 2 were negative (HER2 score 0), and Ki-67 was 25%–30%. The biopsy was reported as atypical biphasic epithelial and myoepithelial neoplasm, with differential diagnoses of atypical adenomyoepithelioma (A-AME), malignant adenomyoepithelioma (M-AME), and metaplastic carcinoma, and recommended excision for definitive diagnosis. The left axillary node core needle biopsy was negative. Radiology–pathology correlation was concordant, and complete excision was planned. Because of symptomatic macromastia causing neck and back discomfort and bra-strap grooving, a therapeutic left oncoplastic lumpectomy with symmetrical right reduction mammoplasty was performed. The left breast specimen included the tumor and three axillary nodes. Histopathology revealed a well-circumscribed biphasic tumor composed of epithelial and myoepithelial cells forming glands and tubules, with alternating atypical and non-atypical areas in a fibromyxoid background (Fig. 4A and B). The atypical regions exhibited pleomorphic, large, hyperchromatic nuclei and frequent mitotic figures (up to 11 per 10 high-power fields, including atypical forms) with extensive necrosis (Fig. 5A–C). No components of conventional invasive ductal or lobular carcinoma, ductal carcinoma in situ, or lobular carcinoma in situ were identified. There was no lymphovascular or perineural invasion. Repeat myoepithelial markers staining on the excision confirmed the same pattern seen in the biopsy (Fig. 5D). The final diagnosis was pure malignant adenomyoepithelioma measuring 45 × 36 × 36 mm. Surgical margins were clear, the closest being the deep and superior margins at 3 mm. Biomarker testing confirmed ER-, PR-, and HER2-negative status with Ki-67 35%–40%. All three axillary nodes were negative, and the final pathological stage was pT2 pN0 (AJCC 8th edition).
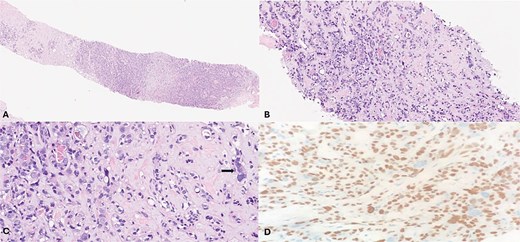
(A) (Hematoxylin & eosin (H&E), 2×) low-power view shows distinct neoplasm with infiltrating tumor cells set in a fibromyxoid stroma. (B) & (C) (H&E, 10×, 20×) show a biphasic neoplastic pattern composed of small, compressed tubules lined by two distinct cell populations: Inner round luminal cells and outer polygonal-to-spindled cells with clear cytoplasm. Occasional atypical cells are also noted (arrow). D P63 immunohistochemistry (20×) highlights the prominent myoepithelial cells and focally the atypical cells.
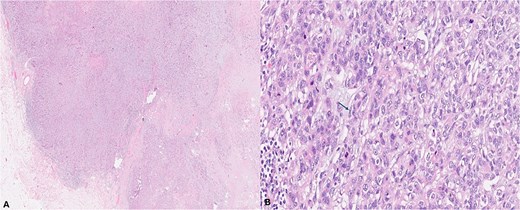
(A) (H&E, 2×) low-power view shows a well-defined neoplastic mass with areas demonstrating both pushing and focally infiltrative borders. (B) (H&E, 20×) high-power view shows neoplastic small, compressed tubules lined by two distinct cell populations with minimal atypia: Inner round luminal epithelial cells and outer polygonal-to-spindled myoepithelial cells with clear cytoplasm (arrow).
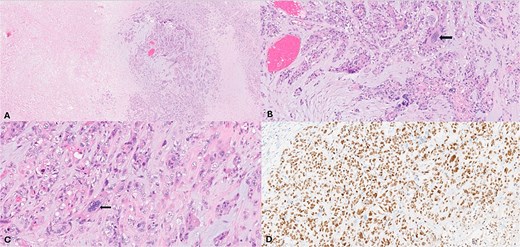
(A) (H&E, 2×) low power view show the neoplasm exhibiting necrosis. (B) & (C) (H&E 10×, 40×) intermediate and high-power views showing neoplastic compressed tubules and trabeculae in a fibro-myxoid matrix background lined by two distinct cell populations with some cells exhibiting marked atypia (arrow). (D) P63 immunohistochemistry (10×) highlights the prominent myoepithelial cells and the atypical cells.
The postoperative course was uneventful, and wound healing was satisfactory. Staging CT of the chest, abdomen, and pelvis, as well as bone scan, revealed no metastatic disease. The multidisciplinary tumor board recommended no further axillary surgery. The patient received adjuvant radiotherapy without systemic therapy. At 1-year follow-up, she remains alive and disease-free, satisfied with the cosmetic outcome, and continues on annual surveillance.
Discussion
Malignant adenomyoepithelioma (M-AME) is a rare breast tumor characterized by malignant epithelial and myoepithelial proliferation with variable behavior [1–5]. Diagnosis is challenging due to nonspecific imaging and biphasic histology; thus, immunohistochemistry (p63, CK5/6, calponin, SOX10) is essential [2–5]. Chen et al. [3] reviewed 167 cases, reporting a mean age of 61 years, mean tumor size of 35 mm, high mitotic rate (10/10 HPF), and mean Ki-67 ≈ 31%, with most tumors being triple-negative. Distant metastasis occurred in 21% and local recurrence in 17%, emphasizing complete excision with negative margins as the mainstay of treatment. Recurrence correlated with high-grade features and incomplete excision, while metastases involved lung, bone, and soft tissue [3–6]. Axillary node metastasis is uncommon (~10–15%) and sentinel biopsy is preferred over routine dissection [1, 4, 5, 7]. The benefit of radiotherapy or chemotherapy remains unclear, with limited evidence supporting their use in node-negative, margin-negative disease [1, 4–6]. Molecular studies show frequent PI3K/AKT/mTOR pathway mutations (PIK3CA, AKT1, HRAS), suggesting potential targets for therapy [8]. This case aligns with reported clinicopathologic features, highlights diagnostic challenges, and underscores the importance of clear margins, selective axillary staging, and long-term multidisciplinary follow-up.
Conflict of interest statement
The authors have no conflict of interest to declare.
Funding
No funding provided.



