-
PDF
- Split View
-
Views
-
Cite
Cite
Andrea Naim, Anik Sarkar, Management of strangulated, perforated gastrothorax with resultant tension pneumothorax secondary to an iatrogenic diaphragmatic hernia: a case report, Journal of Surgical Case Reports, Volume 2026, Issue 1, January 2026, rjaf1033, https://doi.org/10.1093/jscr/rjaf1033
Close - Share Icon Share
Abstract
Diaphragmatic hernias arise when a defect in the diaphragmatic musculotendinous framework allows the abnormal protrusion of abdominal contents into the thoracic cavity, which can be further complicated by visceral incarceration, strangulation, and perforation. Whilst the vast majority occur congenitally or are acquired through trauma, diaphragmatic hernias arising from iatrogenic insults represent an exceedingly rare but important clinical entity. Diagnostic challenges arise in large part due to their tendency towards delayed presentation, often with varied, nonspecific and subacute clinical manifestations, as well as equivocal or indeterminate radiographic findings. The report herein represents the inaugural account of an iatrogenic diaphragmatic hernia arising following thoracoscopic pleural endometriosis removal, which was subsequently complicated by gastric strangulation and perforation, culminating in a life-threatening tension pneumothorax. We describe a safe, effective approach to managing this surgical emergency using an entirely laparoscopic technique and relate our findings to the broader literature pertaining to complicated diaphragmatic hernias.
Introduction
Diaphragmatic hernias (DH) arise when a defect in the musculotendinous framework of the diaphragm allows the abnormal protrusion of abdominal contents into the thoracic cavity. Most DHs are congenital, attributed to abnormal diaphragmatic embryogenesis. Acquired DHs typically arise later in life, usually from blunt or penetrating traumatic injury. Iatrogenic diaphragmatic hernias (IDH) are an exceptionally rare subtype of acquired DH, as evidenced by being exclusively reported in limited case reports and series. Most reported cases have occurred following abdominal surgery, with paediatric liver transplantation or resection being the commonest aetiologies [1, 2].
We present the unique case of a 29-year-old woman who developed an IDH following video-assisted thoracoscopic surgery (VATS) for diaphragmatic endometriosis removal, which was further complicated by tension pneumothorax secondary to gastric perforation. To our knowledge, this is the first documented account of such an occurrence. We describe a safe, effective approach to managing this surgical emergency using an entirely laparoscopic technique and contextualize our findings within broader research as it pertains to the management of complicated DHs.
Case report
A 29-year-old female presented in February 2025 with sudden-onset, severe left-sided pleuritic chest pain, nausea, and vomiting. Her background was significant for endometriosis with diaphragmatic involvement. Having failed to achieve appreciable symptom control with medical therapies, she had travelled overseas in September 2024, where she underwent bilateral VATS and excision of multiple endometriosis lesions from the diaphragmatic pleural surface.
On initial examination, she was haemodynamically stable, with epigastric and left upper quadrant tenderness, but no peritonism. A venous blood gas showed a lactate of 2.7, but no acid–base disturbance or respiratory failure. Chest X-ray (CXR) revealed left hemidiaphragm effacement and a large air-fluid level in the left hemithorax (Fig. 1). A thoracoabdominal computed tomography (CT) scan revealed a markedly dilated stomach displaced into the left hemithorax, suggestive of a large, incarcerated left DH (Fig. 2). A nasogastric tube (NGT) was inserted for immediate gastric decompression (Fig. 1). An emergency gastroscopy confirmed a DH with displacement of the whole stomach into the chest. Endoscopic pyloric cannulation was impossible due to the herniated stomach’s configuration, but there was no mucosal damage, transmural necrosis, or gastric volvulus. Therefore, NGT decompression was continued as a temporizing measure, with planned semi-urgent definitive repair within a few days.
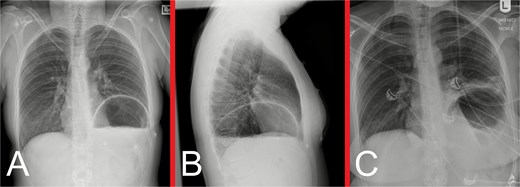
(A) Anteroposterior and (B) lateral view CXRs taken during the patient’s initial presentation, demonstrating effacement of the left hemidiaphragm outline, with a large air-fluid level within the left hemithorax and no evidence of a pneumothorax. (C) Anteroposterior view CXR following urgent NGT insertion for gastric decompression with its tip projecting over the stomach, with associated compressive atelectasis of the left lung.
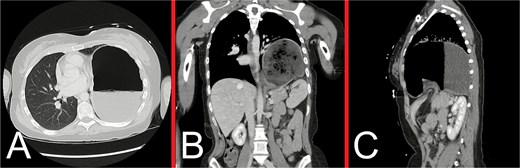
Representative CT images taken during the patient’s initial presentation. (A) Axial, (B) coronal, and (C) sagittal views illustrate a markedly distended stomach with an air-fluid level that is displaced into the left hemithorax, with normal mural enhancement and no evidence of gastric pneumatosis. The left lung is partly collapsed, but there is no evidence of an associated pleural effusion or pneumothorax. The free edge of the ruptured diaphragm is best appreciated on the sagittal view. These findings are consistent with a large, incarcerated left DH, with possible gastric volvulus.
On admission day three, she deteriorated acutely with worsening pain and dyspnoea, desaturations and haemodynamic instability, warranting new oxygen and inotropic support. An urgent bedside CXR showed a new, left tension pneumothorax (Fig. 3). Urgent beside decompression with intercostal catheter (ICC) insertion yielded large-volume gastric content output (Fig. 3). A repeat CT chest demonstrated a new, left hydropneumothorax, raising suspicion for gastric perforation and evolving mediastinitis (Fig. 4).
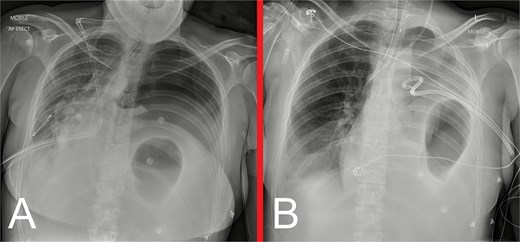
(A) Urgent bedside CXR demonstrating a new, large, left pneumothorax with significant rightward mediastinal shift, consistent with a tension pneumothorax. (B) Anteroposterior CXR following insertion of a left ICC via an anterior approach, showing restoration of the mediastinal structures to the midline, improved aeration of the right lung and near-complete collapse of the left lung. Obliteration of the left hemidiaphragm outline, left hydropneumothorax, and gastric herniation into the left hemithorax are also demonstrated.
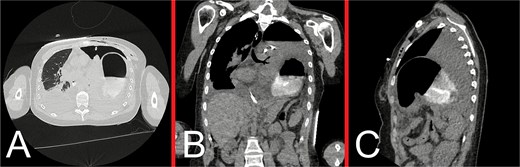
Representative CT images taken following decompression of the left tension pneumothorax with ICC insertion. (A) Axial, (B) coronal, and (C) sagittal views demonstrate a persistent left DH with associated gastrothorax. Since the CT obtained on initial presentation, there has been interval evolution of a left hydropneumothorax as evidenced by an air-fluid level outside the boundaries of the herniated stomach, and near-complete collapse of the underlying left lung. The tip of the pigtail ICC is located medially within the left hemithorax. These findings are suggestive of gastric strangulation and perforation arising as a secondary complication of the DH.
We proceeded with emergent surgical intervention the same day (Fig. 5). Using an entirely laparoscopic approach, the hernia sac was painstakingly mobilized, with extension of the diaphragmatic defect being necessary to reduce the stomach into the abdominal cavity. A 3 cm gastric fundus perforation was identified and definitively controlled with a stapled partial fundectomy. The thoracic and abdominal cavities were thoroughly irrigated, followed by insertion of two ICCs and two abdominal drains for ongoing contamination control. The DH defect was repaired primarily with a nonabsorbable suture and reinforced with an absorbable synthetic mesh. Gastroscopy confirmed viable gastric mucosa and facilitated NGT and nasojejunal tube (NJT) insertion. She was transferred to the Intensive Care Unit postoperatively and received broad-spectrum intravenous antibiotics (piperacillin-tazobactam and fluconazole) for presumed mediastinitis.
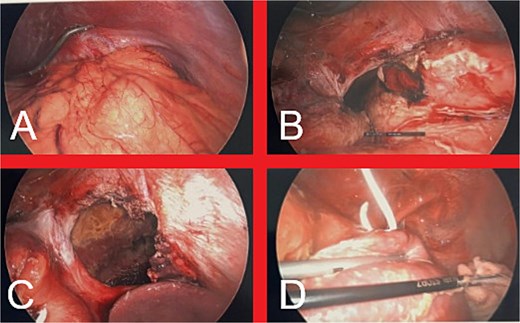
(A) Intraabdominal view of the DH upon initial laparoscopic entry into the abdomen. (B) Appearance of the stomach following dissection of the hernia sac and reduction of the stomach into the abdominal cavity, revealing a 3 cm perforation in the gastric fundus. (C) Final appearance of the large diaphragmatic defect prior to definitive repair. (D) Intrathoracic view of left hemithorax, demonstrating contamination with gastric contents, fibrin deposition on the left lung and the previously inserted ICC.
She was extubated and weaned off all inotropes on postoperative day one (POD1). NJT feeds were gradually introduced on POD2 until adequate oral nutrition was attainable. By POD5, the ICCs, abdominal drains and NGT had been sequentially removed, and she was transferred to the ward. By POD13, she was meeting her nutritional requirements orally, allowing NJT removal. She made a stable and steady postoperative recovery and was discharged home on POD17.
She remained well on outpatient follow-up at 2 weeks. Progress CXRs at 3 and 7 months showed satisfactory appearances, without evidence of hernia recurrence (Fig. 6).
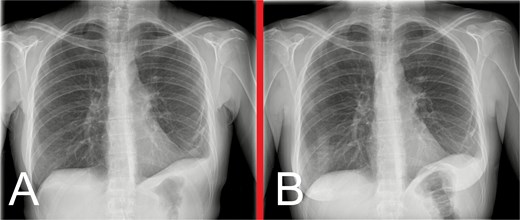
Progress anteroposterior view CXRs taken at (A) 3 months and (B) 7 months postoperatively, showing persistent, but stable, left hemidiaphragm elevation with associated generalized volume loss of the left lung, but no evidence of hernia recurrence.
Discussion
Awareness of IDHs as potential complications of thoracoabdominal surgery is vital, as delayed recognition and management may portend significant morbidity and mortality. IDH following minimally invasive diaphragmatic lesion excision is exceptionally rare, with only three cases reported following thoracoscopic or laparoscopic approaches [3–5]. We present the inaugural account of an IDH arising following VATS removal of diaphragmatic endometriosis, which was further distinguished by the evolution of a tension pneumothorax secondary to gastric perforation. Similar to our patient’s case, IDHs occurring in this setting are seemingly characterized by delayed presentation and nonspecific symptoms, often becoming evident several months following the initial insult and, occasionally, presenting incidentally on imaging [3–6].
The pathogenesis of IDH in this context is unknown, but it is theorized that inadvertent heat injury to deeper diaphragmatic layers from energy devices creates a weak point that gradually enlarges due to the continuous pleuroperitoneal pressure differential, allowing visceral herniation [3–6]. Despite diligent intraoperative inspection, significant damage may be macroscopically imperceptible. Moreover, even if an injury is repaired upon identification, an IDH may still occur [1]. Indeed, in our patient, the defects created following the excision of numerous pleural nodules had been closed primarily, albeit with an unknown suture type or closure method. This highlights the lack of evidence guiding optimal primary repair techniques to prevent postoperative IDH formation.
This is the first reported case of a tension pneumothorax occurring in conjunction with an IDH complicated by gastric perforation. Rare cases of tension faecopneumothorax complicating traumatic DH have been reported [7, 8]. Nishimura and Hayasaka reported a secondary pneumothorax in the setting of an IDH following thoracoabdominal aneurysm repair, with a concurrent adhesive small bowel obstruction. However, the site of perforation was in the distal small bowel, well away from the IDH [9]. They theorized that, because the IDH lacked a hernia sac, this allowed intraperitoneal air from the perforation site to leak intrathoracically via the IDH [9]. This free intercavitary communication likely prevented a tension pneumothorax.
Large DHs can easily be misdiagnosed as a tension pneumothorax on radiographic imaging, raising the risk of iatrogenic visceral injury from erroneous ICC insertion [10, 11]. Careful CXR interpretation is warranted, as these pathologies have subtle differences [10]. Imaging must always be interpreted within the clinical context, considering the presentation’s acuity, any history of trauma or surgery, and the presence of features of cardiorespiratory compromise. Tension pneumothorax is a clinical diagnosis and requires immediate intervention with needle thoracocentesis. If immediate measurable improvement does not occur after this, CXR should be performed before ICC insertion in case of a missed DH [10]. If the situation permits and the patient is not in extremis, NGT decompression should also be considered, as this may improve anatomical delineation and diagnostic clarity on further imaging prior to unnecessary and potentially deleterious ICC insertion [11].
Evidence-based guidance on IDH management is limited, but surgery is the mainstay of treatment for DHs complicated by established or imminent visceral incarceration, strangulation and/or perforation [12]. Both open (thoracotomy or laparotomy) and minimally invasive (thoracoscopic, laparoscopic, and robotic) surgical approaches have been described [3–6, 12–15]. Until more dedicated research becomes available, it is best to be led by current management guidelines for complicated traumatic and congenital DHs. Whilst the chosen approach is largely dictated by surgeon preference and expertise and resource availability, a laparoscopic approach is favoured, given its association with reduced morbidity, length-of-stay and likelihood of discharge to a skilled nursing facility [12, 15]. Thoracoscopy may be used adjunctly in cases of chronic herniation, where prominent visceropleural adhesions herald a risk of visceral perforation. Primary defect closure with a nonabsorbable suture should always be attempted where possible [12]. For larger defects (>3 cm) or where tension-free closure is unachievable, reinforcement with mesh is recommended, ensuring ≥1.5–2.5 cm defect edge overlap [12]. Whilst biological meshes offer the advantages of reduced rates of hernia recurrence, infection, and mesh displacement/migration, they are contraindicated in DH complicated by perforation. In these contaminated settings, absorbable synthetic mesh is favoured [12].
IDH evolution following thoracoabdominal surgery may be clinically silent, with radiographic evidence often becoming apparent long before symptoms occur. It is crucial to remain vigilant when reviewing surveillance postoperative imaging and be attentive to symptoms and signs that suggest gastrointestinal or cardiorespiratory compromise, in order to ensure timely diagnosis and intervention and prevent potentially life-threatening complications.
Acknowledgements
The authors would like to acknowledge the patient whose case is discussed herein for allowing us to share her story in the spirit of contributing to medical literature regarding this rare disease entity. We would also like to thank our family and friends for their continued and unconditional support.
Conflict of interest statement
All authors have no personal or financial conflicts of interest to declare with respect to the research, authorship, and/or publication of this article.
Funding
This work did not receive any funding or sponsorship from any third party agencies or institutions.
Consent
Informed consent was obtained from the patient for publication of the article herein.



