-
PDF
- Split View
-
Views
-
Cite
Cite
Pavel Markov, Grigory Karmazanovsky, Andrey Lysenko, Ovanes Arutyunov, Dmitry Kalinin, Alexander Burmistrov, Giant dilatation of pancreatic duct associated with congenital anomaly of the pancreatic ductal system and small neuroendocrine tumor, Journal of Surgical Case Reports, Volume 2024, Issue 2, February 2024, rjae030, https://doi.org/10.1093/jscr/rjae030
Close - Share Icon Share
Abstract
Dilatation of the main pancreatic duct (MPD) ˃50 mm is a rare phenomenon. We present the case of successful surgical treatment a 63-year-old female patient who presented epigastric pain and weight loss of 10 kg in 6 months. Based on preoperative examination, the dilatation of MPD was 75 mm. The preliminary diagnosis was main duct intraductal papillary mucinous neoplasm. Extended pylorus-preserving pancreatoduodenectomy with resection of the body of the pancreas was performed. According to the results of pathological and immunohistochemical studies, performed by independent experts, the final diagnosis of congenital anomaly of the ductal system and neuroendocrine tumor of the accessory pancreatic duct was made.
Introduction
Dilatation of the main pancreatic duct (MPD) is not a specific symptom and can be a result of a congenital anomaly of the ductal system or the result of various diseases: intraductal papillary mucinous tumor (IPMN), chronic pancreatitis, pancreatic neuroendocrine tumors (NETs), pancreatic adenocarcinoma [1–3].
The average value of MPD dilatation in various pathologies is 7 mm within the range of 4–35 mm [1]. Cases with MPD dilatation of more than 50 mm are rare [2, 4].
Generally, significant dilatation of the MPD is observed in IPNM of the pancreas when the average diameter of the MPD reaches 9.03 ± 4.33 mm [5].
We present a rare case of giant dilatation of the pancreatic duct up to 100 mm, associated with a congenital anomaly of the ductal system and NET of small size at the minor duodenal papilla which simulated IPMN.
Case report
A 63-year-old female patient complained on epigastric pain and weight loss of 10 kg in 6 months. Over the past 2 years, the patient underwent regular abdominal ultrasound for cystic lesion in the pancreas.
CT scan demonstrated enormous cystic dilatation of the MPD up to 75 mm with total atrophy of the pancreatic head and body parenchyma with dilatation of the branch ducts in the tail up to 6 mm (Fig. 1).
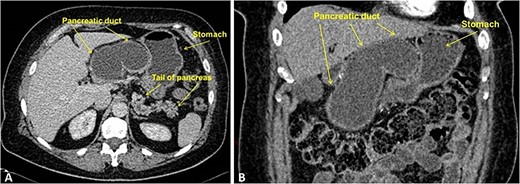
Contrast-enhanced CT, portal-venous phase: (A) axial; (B) frontal. The images demonstrate marked smooth cystic dilation of the head and neck region of the pancreas with duct dilation into the body (arrowheads), without mural nodules or side branch dilatation.
MRI showed cystic lesion in the pancreatic head, neck, and body, connected with the MPD, the maximum diameter of 75 mm, with the parenchyma only in the pancreatic tail (Fig. 2).
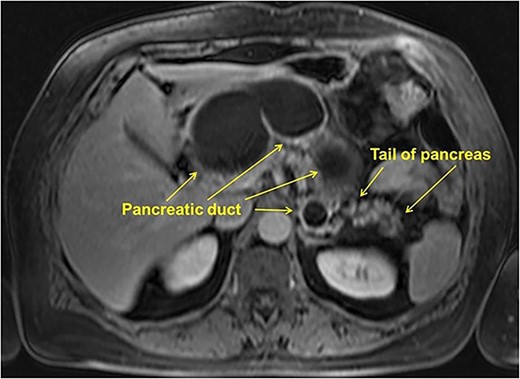
Endoscopic ultrasound (EUS) showed no mural thickening or nodules in the dilated segments of pancreatic duct. No pathological changes were detected during gastroduodenoscopy and colonoscopy. The tumor markers were normal: CEA—1.9 ng/ml; СA-19-9-1.95 U/mL, no signs of diabetes mellitus were detected. Thus, the preoperative diagnosis was main duct IPMN (MD-IPMN) and surgical intervention was performed.
Intraoperatively, it was revealed that the pancreatic head and body were markedly enlarged and presented as a cystic lesion, with a maximum width of up to 120 mm, the tail was without any macroscopic changes (Fig. 3). No tumor spread or distant metastasis were found.
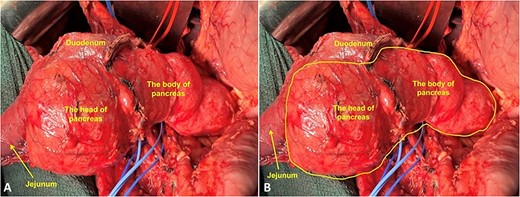
Intraoperative photo. (A) Fully mobilized head of the pancreas with the duodenum; (B) Yellow line marks the border of cystic lesions in the pancreas.
After mobilization of the pancreatoduodenal complex, an extended pylorus-preserving pancreatoduodenectomy was performed, with resection of the body of the pancreas.
Frozen section revealed no presence of tumor cells in the cut edge of the pancreas, with number of acinar cells only 10%. No complications were encountered and the patient was discharged in satisfactory condition on the 12th postoperative day.
Grossly, MPD was significantly dilatated (100 mm in the transverse dimension), its inner surface was smooth and shiny (Fig. 4). There were two orifices along the lateral surface of the MPD: one blindly ended in the pancreatic tissue, without connection with the major duodenal papilla, into which only the common bile duct (CBD) flowed (Fig. 4B and C). The second one communicated with the minor papilla, located 2.5 cm proximal to the major duodenal papilla.
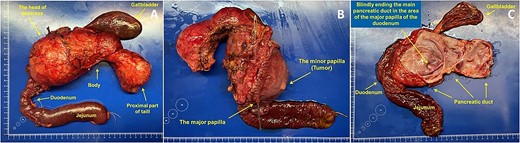
Image of the gross specimen: (A) general view; (B) rear view. Longitudinally dissected duodenum. Probe in the lumen of the CBD. (C) MPD opened longitudinally, the lumens of the gallbladder, duodenum, and jejunum were opened.
Pathological findings revealed intraductal papillary proliferation of the MPD wall. The parenchyma of pancreatic head, neck, and body was completely atrophied.
Mucosa of the MPD was represented by a flat cylindrical epithelium, partially with areas of squamous metaplasia. There were foci of fibrosis and lymphocytic infiltrates in the subepithelial zone. Elements of ovarian-like stroma were not detected during immunohistochemical analysis of progesterone receptor (clone 1E2, Ventana).
In the area of minor papilla there was a tumor measuring 7 × 5 mm with a solid, trabecular, and nested structure of medium-sized polygonal cells with rounded nuclei with fine chromatin (Fig. 5A–C). The tumor spreads to the mucous membrane, submucosa, and proper muscular layer of the duodenum, as well as to the terminal sections of the accessory pancreatic duct, with stenosis of the latter. Mitotic count is 1 per 2 mm2. In six out of 24 removed regional lymph nodes, there were metastases of the tumor (Fig. 5D).
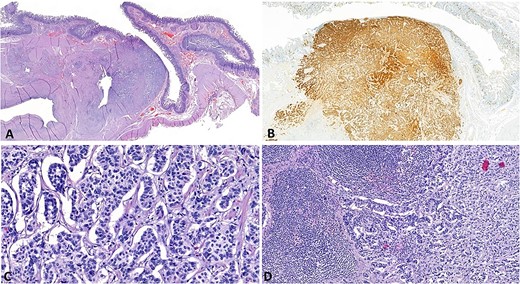
Microphoto: (A) NET in the area of the minor papilla (H and E, ×12); (B) cytoplasmic expression of chromogranin A (clone DAK-A3, DAKO) by tumor cells and enteroendocrine cells of the duodenal mucosa (DAB, H × 30); (C) Solid, trabecular and nested structure tumor of medium-sized polygonal cells with rounded nuclei with fine chromatin. Mitotic count is up to 1 per 2 mm2 (H and E, × 40); (D) Metastasis of the NET to a regional lymph node (H and E, × 200).
Immunohistochemical analysis showed diffusely positive trabecular growth for chromogranin A (clone DAK-A3, DAKO) and diffuse membrane expression for CD 56 (clone 123C3.D5, CellMarque). Gastrin (polyclonal, DAKO) and serotonin (clone 5HT-H209, DAKO) was not expressed. The Ki-67 labeling index was <1%. Thus, the final diagnosis of congenital anomaly of the ductal system and NET (G1) of the ampulla of the accessory pancreatic duct was made.
Preoperative CT and MRI images were reviewed by two independent experts. However, it was not possible to visualize the NET according to the radiological examinations.
The patient was examined 12 months after surgery. There were not any complaints, diabetes mellitus was not diagnosed. Clinical and laboratory tests showed that there was exocrine pancreatic insufficiency which manifested as steatorrhea (fecal elastase 30.3 μg/g). It was compensated by daily intake of pancreatin at a dose of 100.000 IU/day.
Discussion
There are only two cases of a significant dilatation of the MPD with a diameter of more than 50 mm in the current literature.
Congenital dilatation of the MPD without an obvious pathological cause is an extremely rare phenomenon, we found a single description of such a case [2].
A 36-year-old patient with symptoms of intestinal obstruction and significantly dilated MPD by CT scan, MRI, and EUS with no evidence of IPMN. During a year follow-up there was no any changes and authors suggested that there was a congenital dilatation of the MPD.
Koschny R. et al. [4] published a case of MPD dilatation up to 50 mm. Abdominal ultrasound showed biliary hypertension with a distal block and an enlargement of the MPD. The patient was diagnosed with MD-IPMN and underwent pancreatoduodenectomy. Histological examination confirmed the presence of IPMN with a low level of dysplasia.
IPMN is the most common reason for significant MPD dilatation. At the same time, a correlation was noted between the degree of duct dilatation and the likelihood of malignant tumor development [6]. According to Hamada T. et al. [6] among all patients with IPMN, the number of patients with MPD dilatation of more than 5 mm is 10%, and 26% of them had malignancy. And if with MPD diameter from 5 to 10 mm the frequency of cancer diagnosis for 5 years was 10%, then with a MPD diameter ˃10 mm the same value was 33.3%.
In our case, the cause of significant dilatation of the MPD was small size NET, which caused compression of the accessory pancreatic duct in combination with an anomaly in the structure of the MPD, in which only the CBD flowed into the major duodenal papilla, and the MPD communicated with the duodenum only through minor papilla, which, in turn, was squeezed by the tumor.
The literature describes some cases of small size NET, accompanied by dilatation of the MPD [3, 7–9].
Kiyonaga M. et al. [7] published a case of a 26-year-old patient with NET of the pancreatic head that was the result of compression and fibrosis of the MPD. In our case, there was also a compression of the pancreatic duct, but the accessory pancreatic duct, associated with the anomaly of ductal system.
Another case has been described by Ishii T. et al. [9]. The patient was diagnosed with a non-functioning NET with a diameter of ˂10 mm, which caused the dilatation of the MPD up to 7 mm. The operation was performed after dynamic observation, when the tumor reached the size of 10 mm. After pancreatoduodenectomy, pathomorphological examination confirmed NET, and the proliferation of fibrous tissue around caused stenosis of the MPD with its dilatation.
According to Kawamoto S. et al. [8] and Shi C. et al. [10] MPD dilatation is caused mainly by NETs producing serotonin. In these cases, there is the stromal fibrosis in the tissues around the tumor that led to stenosis of the duct with its dilatation and atrophy of the pancreatic parenchyma. Thus, when studying specimens of removed NETs in patients with MPD dilatation, in five (83%) cases out of six, tumor production of serotonin was detected. When analyzing specimens of removed NETs in patients without MPD dilatation, tumor production of serotonin was found only in three (6%) of 47.
Moreover, the development of fibrosis with subsequent stenosis and dilatation of the MPD does not depend on the size of the tumor and can be observed with very small tumors. Thus, in two out of six patients with NET and MPD dilatation, as in our case, the tumor was not detectable on preoperative CT scan. It is recommended by many authors to carry a thorough search for small tumors, in particular NET, if an idiopathic dilatation of the MPD is detected [8, 10].
In our case, the tumor did not produce serotonin, and it can be assumed that not only compression of the outflow duct by the tumor, but also a congenital anomaly of development played a role in the pathogenesis of a marked dilatation of the MPD.
Conclusion
To our knowledge, this is the first case of such a significant dilatation of the pancreatic duct, caused by combination of a congenital anomaly in the development of the pancreatic ductal system and small NET.
Conflict of interest statement
Authors declare no conflict of interest.
Funding
None declared.
Patient informed consent
Obtained.



