-
PDF
- Split View
-
Views
-
Cite
Cite
Santiago A Endara, Gerardo A Dávalos, Alberto Cardenas, Karina Gangotena, Alexis Flores, Sabine Dueñas, Gabriel A Molina, Dysphagia lusoria in a young patient with a concomitant aortic arch anomaly, successfully treated with surgery: a case report, Journal of Surgical Case Reports, Volume 2024, Issue 1, January 2024, rjad710, https://doi.org/10.1093/jscr/rjad710
Close - Share Icon Share
Abstract
Dysphagia is a common condition in clinical practice; however, an unusual type of dysphagia due to compression of the esophagus by an abnormal right subclavian artery may be discovered in a rare subset of patients. The prognosis and treatment will depend on the severity of the symptoms and the compromise of surrounding structures. We present the case of an 18-year-old female who presented with gradually progressive dysphagia. At first, it was treated as gastroesophageal reflux disease; nonetheless, the dysphagia became severe, and after a thorough evaluation, an aberrant right subclavian artery that compressed the esophagus was discovered along with a truncus bicaroticus. She was successfully treated with surgery without any complications. On follow-ups, she’s doing well.
Introduction
An aberrant right subclavian artery (ARSA) is a rare congenital anomaly of the aortic arch that can develop troublesome complications [1, 2]. When dysphagia appears, some patients may be managed with dietary modifications, yet patients with severe symptoms will need surgery [1, 3].
We present the case of an 18-year-old female; she presented to our hospital with progressive dysphagia after clinical evaluations and imaging tests. An ARSA that compressed the esophagus was discovered along with a truncus bicaroticus. After successful surgery, the patient fully recovered.
Case report
Patient is an 18-year-old female without past medical history. She suffered a 2-month gradually progressive dysphagia accompanied by mild reflux. At first, it was treated as gastroesophageal reflux disease (GERD), and proton pump inhibitors were given with poor results. Dysphagia became so severe at one point that she could not drink any liquid, including saliva; therefore, she presented at our hospital. Clinical examination, including palpation of her peripheral pulses, was unremarkable; therefore, additional exams were required. An esophagogram (Fig. 1) revealed an extrinsic compression above the level of the aortic arch. With these findings, a computed tomography angiography was requested and demonstrated aberrant anatomy of the aortic arch; the carotid arteries arise from one common trunk, the truncus bicaroticus, and an ARSA was found compressing the esophagus as it crossed behind it. There was no evidence of Kommerell’s diverticulum, aneurysmal dilation, or other abnormalities (Fig. 2A and B).
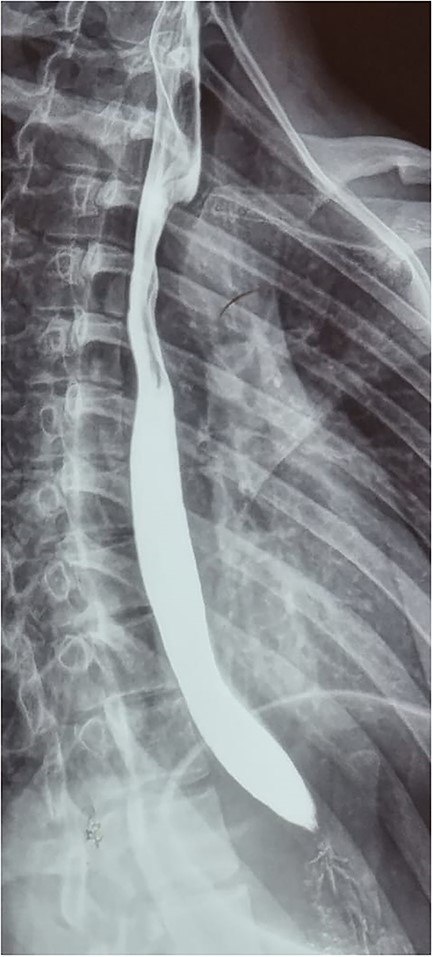
Esophagogram revealing an extrinsic compression above the level of the aortic arch.
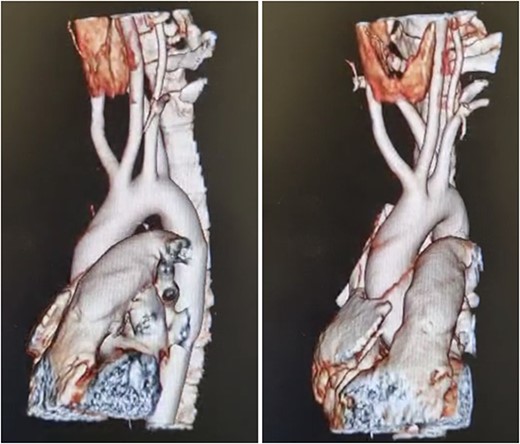
(A) CT, showing and ARSA compressing the esophagus, (B) CT, truncus bicaroticus and an ARSA.
Cardiothoracic assessment was required, and after discussing the surgical procedure and its potential complications, surgery was decided. A right posterolateral thoracotomy was done through the third intercostal space; this gave adequate exposure to the mediastinum. First, the esophagus was dissected to expose the ARSA. When found, the dissection continued proximally to the aortic arch and distally to achieve control of the aberrant vessel. The right subclavian artery arose from the posterior aspect of the aortic arch and was dissected entirely from the esophagus. Once it was free, the right subclavian artery was clamped, and the right upper limb was monitored; adequate circulation in the right arm and hand was tested after monitoring blood pressure, oxygen saturation, and clinically confirming good distal capillary refill time, and limb color.
Once adequate blood supply of the right upper limb has been confirmed, the right subclavian artery was divided with a vascular mechanical stapler (Covidien, Medtronic Dublin, Ireland), thereby achieving auto-retraction of the artery and freeing the esophagus. Afterward, the lung was re-expanded entirely, a pleural drain was placed, and the chest was appropriately closed (Fig. 3A, 3B, and 3C).
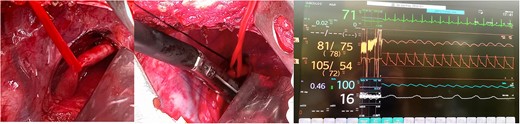
Intraoperative findings, (A) Dissected ARSA, (B) ARSA being clamped with mechanical suture, (C) Monitor showing comparative pressures between upper limbs.
Her postoperative course was uneventful; the right upper limb was closely monitored, and she had no vascular or motor impairment; her dysphagia disappeared, and she was able to eat a regular diet. She was discharged without any complications. On follow-ups, a right upper limb doppler was done, finding adequate circulation of the extremity.
Discussion
Congenital anomalies of the aortic arch, although rare (3%), can be associated with vascular rings, congenital heart disease, and anatomical abnormalities [1]. They can have important clinical implications since these variants can encircle and compress many organs, including the trachea or esophagus [1, 2]. The most common defect found in the adult population is the ARSA (0.3 to 3%) [1, 3]. This anomaly, which was first described in 1735 by Hunauld et al., occurs due to the involution of the fourth vascular arch and the right dorsal aorta, leaving the seventh intersegmental artery attached to the descending aorta; in some circumstances, the root of this aberrant artery can have a broad base, usually called Kommerell’s diverticulum [3]. The anatomical course of this anomalous artery can vary as it passes from the chest into the right arm [1, 4]. In most cases (80%), it will cross between the esophagus and the spine [2, 5]. As it happened to our patient.
ARSA can be associated with a concomitant congenital anomaly of the carotid arteries, either with a shared trunk or a close origin; if this happens, it will lead to a decreased distensibility of the esophagus as it is compressed posteriorly by the aberrant vessel leading to dysphagia, most commonly known as dysphagia lusoria [1, 3]. Bayford first described this disease in a young woman who died of permanent malnutrition due to long-term dysphagia [2, 6]. Other abnormalities associated with ARSA include the ipsilateral vertebral artery arising from the common carotid artery, anomalous point of entrance of the vertebral artery into the cervical spine, thoracic duct ending in the right subclavian, and non-recurrent inferior laryngeal nerve [1, 6]. In our patient, truncus bicaroticus with an ARSA was discovered.
ARSA is found more often in females, and when present, symptoms usually occur at the two extremes of life if compression of the esophagus or trachea arises [3, 7]. In children, respiratory symptoms are more common, and although dysphagia may be most frequent in adults, children may demonstrate other symptoms like dyspnea, GERD, retrosternal pain, cough, and weight loss [1]. On other occasions, ARSA does not cause any symptoms and can be discovered incidentally (up to 90%) [1, 4]. In our patient, progressive dysphagia accompanied by mild reflux was the main symptom.
Diagnosis will depend on the clinical manifestations; some patients will be asymptomatic, while others may present with asymmetrical radial pulses or other vascular abnormalities [1, 4]. A posterior mass may be visible on computed tomography (CT) scans [2]. If dysphagia or dyspnea appears, more invasive tests may reveal a posterior, rounded filling defect in the barium swallow or a pulsatile mass on the posterior aspect of the esophageal wall in an endoscopy [1, 7]. Nonetheless, the best imaging modalities will be thoracic aortograms, contrast-enhanced CT scans, and magnetic resonance imaging, which are always necessary for preoperative planning [1, 8]. As it was done in our patient.
Treatment will depend on the symptoms; ARSA can be carefully followed without intervention in an asymptomatic patient [2, 8]. However, if symptoms appear or there is a simultaneous aneurysmal formation, treatment should be considered due to the high risk of complications, including rupture, dissection, compression, and even embolization [1, 3, 5].
Several classical and endovascular treatments are available since the first successful surgery by Gross in 1946 [1, 9]. The classical approach involves the division and ligation of the right subclavian artery; the use of stents grafts and transposition of the right subclavian artery with carotid subclavian bypass will depend on the associated vascular anomalies like truncus bicaroticus that may compromise blood flow to the right upper limb, spine, and vertebrobasilar circulation [2, 4, 5]. Endovascular or hybrid techniques could be appropriate in particular cases, reducing morbidity and mortality; however, this approach is only suitable in cases without aneurysms because the origin of the ARSA cannot be ligated [1, 10]. Also, severe complications, including migration of the plugs, esophageal fistula, embolisms, and stroke, should be considered [2, 6].
In patients who won’t tolerate surgery, endoscopic dilation of the esophageal narrowing may be used as a palliative treatment [1, 2].
In our case, once ARSA was diagnosed, surgery was needed since dysphagia was severe; after surgery, the patient completely recovered and was able to eat and drink as usual and showed no neurological or vascular compromise.
Conclusions
Dysphagia is a common symptom seen in daily clinical practice and regretfully is not usually given the significance it deserves. A high degree of clinical insight is needed if dysphagia lusoria is not to be missed. If ARSA is symptomatic, it should always be corrected to overcome symptoms and prevent possible complications. In addition, when necessary, reconstruction of the blood flow to the upper extremity to maintain the arm’s viability and keep the spinal and vertebrobasilar circulation is of paramount importance.
Conflict of interest statement
None declared.
Funding
None declared.



