-
PDF
- Split View
-
Views
-
Cite
Cite
Brian Ng Hung Shin, Samuel X Tan, Anthony Griffin, Vijay Kanagarajah, Ailin Tan, Symptomatic scrotal-inguino-retroperitoneal lymphocele in a kidney transplant patient—to drain but how to drain?, Journal of Surgical Case Reports, Volume 2023, Issue 4, April 2023, rjad192, https://doi.org/10.1093/jscr/rjad192
Close - Share Icon Share
Abstract
Scrotal-inguino-retroperitoneal (SIR) lymphocele is a rare complication following kidney transplant. This entity is characterized by a tract originating in the retroperitoneal space, through the inguinal canal and scrotum following lymph hydrodissection. Systematic review investigating SIR lymphocele yielded cases with open fenestration of the sac into the peritoneum as treatment. We described a case report of a male in his 60s with a functioning kidney transplant and SIR lymphocele, which was successfully managed in the short term with percutaneous drainage of the collection. However, the collection recurred and computed tomography scan showed a multiloculated collection that prompted surgical management. Intraoperatively, the encapsulated fluid-filled tract was excised and a drain was placed, which was removed 48 h later. The patient wore a hernia belt for 6 weeks as support. He had no recurrence of his lymphocele following serial reviews for 9 months now.
INTRODUCTION
Scrotal-inguino-retroperitoneal (SIR) lymphocele is a rare complication following kidney transplant with an hourglass fluid communication in between these compartments with the narrow neck at the deep inguinal ring [1]. Unlike the other cases described in the literature that were managed by open fenestration of the sac into the peritoneum, our patient had an encapsulated fluid tract requiring a different approach to management.
CASE REPORT
A man in his 60s on haemodialysis for end-stage renal failure due to obstructive uropathy had an open donation-by-brain death kidney transplant. He had a right iliac fossa transverse incision with extraperitoneal dissection down to the right iliac vessels and end-to-side vascular anastomosis. A 15-French Blake drain was placed in the perimeter of the neoureterocystostomy joint. On postoperative Day 3, the drain was removed due to diminishing drain output down to 10 ml, and a scheduled renal transplant ultrasound on that day demonstrated no evidence of perinephric collection. The inguinal fluid pocket was localized with bedside ultrasound and 30 ml of clear fluid was aspirated with immediate symptom relief.
Three weeks postoperatively, the patient noticed a progressively enlarging right groin mass and associated discomfort. His examination was notable for a firm non-pulsatile non-reducible inguinal mass with no bowel sounds. He proceeded to have a CT Kidney, Ureter, Bladder (KUB), which revealed a small amount of perinephric collection (20 Hounsfield unit) marginating the posteromedial aspect of the transplant lower pole with a further fluid collection distending the right inguinal canal (10 Hounsfield unit) (see Fig. 1). The inguinal fluid pocket was localized with bedside ultrasound and 30 ml of clear fluid was aspirated with immediate symptom relief. The patient noticed reaccumulation of the fluid after a week and became symptomatic of pain again by the end of the third week.
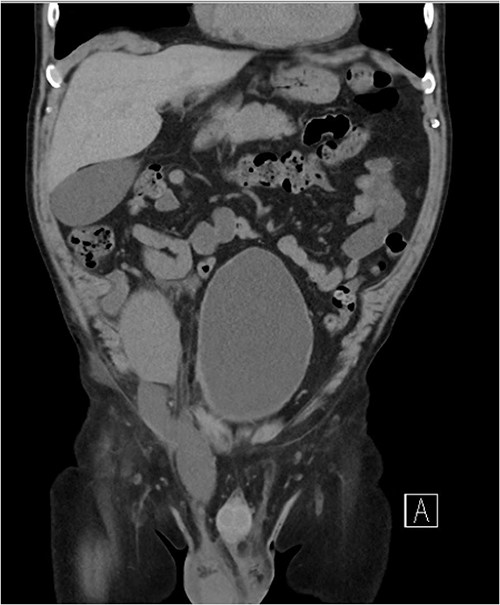
Coronal CT image showing a dominant fluid collection extending off the lower pole of the transplant kidney into the right inguinal canal.
Given the initial improvement in his symptom and in a bid to avoid invasive surgery, a formal ultrasound-guided aspiration was organized. The interventional radiology team deemed it unsafe to place a pigtail drain given the proximity of the lower pole collection to the hilum. Instead, 50 ml of clear fluid was aspirated with post-images showing both the inferior and inguinal canal collection completely drained.
Five days later, the patient had another ultrasound and an ensuing CT KUB given recurrence of a painful groin swelling overnight. It showed a reaccumulation of the pelvic fluid collection, which extended into the groin toward the scrotum with two pockets of fluid collection measuring 4.6 × 6.5 × 2.7 cm (42 ml) and 7.7 × 3.5 × 4.6 cm (66 ml) (Figs 2 and 3). We temporized the situation by aspirating 35 ml of fluid by the bedside under ultrasound guidance for symptom relief and booked him for an elective operation given the complex anatomy of the collection.
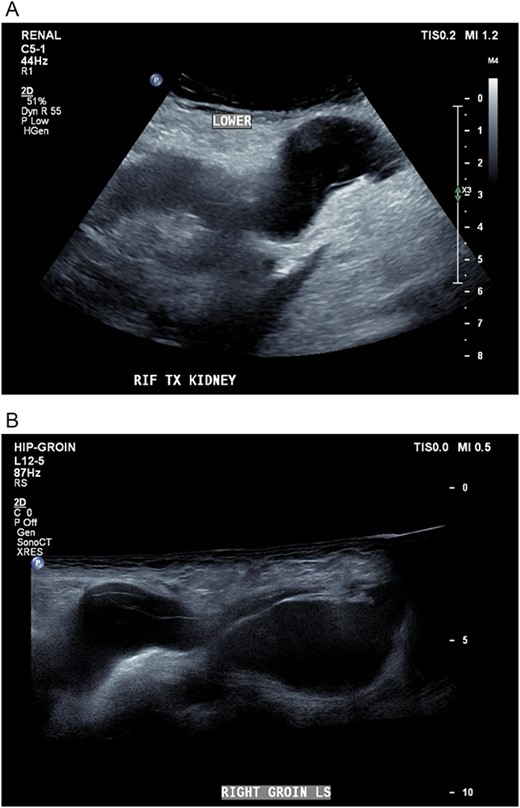
(A, B) Targeted ultrasound showing reaccumulation of a peri-transplant collection which appeared multiloculated measuring a total of 100 ml extending into the right groin.
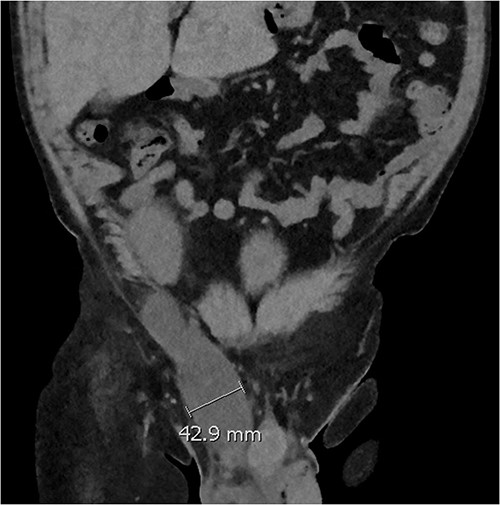
Coronal CT scan showing lobulated low-density fluid collection adjacent to the lower pole of the transplant kidney in the right iliac fossa and extending into the superficial inguinal canal tracking along the inguinal and scrotal hernia with a maximal width of 43 mm in transverse diameter.
Intraoperatively, an incision was made over the inguinal canal inferior to the transplant incision. We dissected down onto the external oblique aponeurosis which bear a lymphocele sac which was densely adhered to the cord structures. We fenestrated the sac to evacuate the fluid, skeletonize it from the cord structures and ligated its proximal and distal ends. In addition, a small fluid containing indirect inguinal hernia was also identified. The hernia sac was opened, fluid evacuated and the sac was subsequently excised with its ends ligated. On completion, a 10-French Bellovac drain was placed in the cavity of dissection. Postoperatively, the patient progressed well. The drain was removed on Day 4 as it remained dry throughout. The patient was discharged with a hernia binder to support the area. He had no recurrence of his lymphocele following serial reviews for 9 months now.
DISCUSSION
The incidence of pelvic lymphocele development following kidney transplantation varies between 3.8 and 32% [2, 3] with the mean time to development reported at 121 days (range 35–631) [4]. No consistent risk factors have been reported for its formation [5]. In comparison, scroto-inguino lymphocele is rarer with only five published cases, which occurred at varying time points after transplant [6].
The pathogenesis in our case is likely peri-transplant lymph fluid, which dissected along tissue planes through the inguinal canal and into the scrotum. The lymph either leaked from the injured lymphatics of the transplanted kidney and/or recipient’s injured iliac lymphatic vessels and accumulated secondary to an imbalance between leakage and absorptive capacity of the pouch surface [7]. Unique to our case, we aimed to shift the balance toward absorption by decreasing the volume of the sump fluid via percutaneous drainage. Although the effect was not sustained, ultrasound-guided aspiration or pigtail drain placement is a reasonable intermediate step prior to considering revision surgery.
Trigger for operative intervention in our case was a third recurrence of the collection which appeared multiloculated. There are no data available on predictors of revisional surgery for SIR lymphocele. Extrapolating from the data for post-traumatic seromas, aspiration volume of >50 ml and presence of a pseudo capsule as in our case are triggers for operative intervention [8, 9]. We decided to excise the pseudo-capsule given it is an important risk factor for recurrence.
CONCLUSION
Percutaneous drainage of SIR lymphocele is an effective short-term management option. Definitive surgery includes open fenestration; however, the presence of a fluid sac warrants excision given it is a risk factor for recurrence.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.



