-
PDF
- Split View
-
Views
-
Cite
Cite
Ryusuke Machino, Shinichiro Kobayashi, Tsutomu Tagawa, Ken Taniguchi, Kengo Kanetaka, Susumu Eguchi, Takeshi Nagayasu, Double vision approach applied by two teams for retrosternal gastric conduit for gastric tube cancer, Journal of Surgical Case Reports, Volume 2023, Issue 2, February 2023, rjad068, https://doi.org/10.1093/jscr/rjad068
Close - Share Icon Share
Abstract
Gastric tube cancer is classically treated with resection through a midline sternal incision. However, because of its invasiveness and limited reconstructive potential, transdiaphragmatic laparoscopic or thoracoscopic dissection of the gastric tube has been investigated. As resection from only the abdominal or thoracic cavity is difficult, we performed surgery with a thoracic surgeon approaching from the thoracic cavity and an abdominal surgeon simultaneously approaching from the cervical and abdominal regions. The gastric tube may be tightly adhered to the back of the sternum, cervicothoracic transition or thoracoabdominal transition. Dissection can be safely performed by operating from two directions simultaneously, the neck and chest or chest and abdomen, to successfully withdraw the gastric tube from the abdominal cavity. We performed this surgery in four cases. This collaborative operation provided a good surgical view and allowed for safe dissection of the gastric tube without requiring sternotomy.
INTRODUCTION
Using a gastric conduit as a substitute for the esophagus is a well-established reconstruction method after esophagectomy. However, gastric tube cancer (GTC) can develop subsequently in the raised stomach [1]. Previously, GTCs were rare owing to the poor prognosis of esophageal cancer (EC). However, GTC occurrence has increased in recent years, following the concurrent use of surgical and chemoradiation therapies for EC [2].
Okamoto et al. [3] reported that eight of 414 patients who underwent esophagectomy developed GTC, and the mean and median times from esophagectomy to GTC diagnosis were 55.8 and 56.8 months, respectively [2]. Most patients with GTC are diagnosed with advanced-stage disease [1, 4]. Recently, endoscopic treatment, including endoscopic mucosal resection, endoscopic submucosal dissection and partial or wedge resection of early stage GTC, has been successful as a less invasive treatment [1, 3–8]. However, endoscopy indication is limited to superficial or less-advanced tumors. The surgical approach for GTC resection depends on the reconstructive route used during esophagectomy. If reconstruction is performed using the retrosternal route, a median sternotomy is traditionally used that is highly invasive and predisposes patients to osteomyelitis [9, 10].
We present three cases of GTC successfully treated using an endoscopic retrosternal gastric conduit resection technique that approaches simultaneously from the chest and abdomen without a sternal incision.
CASE SERIES
We experienced three cases of GTC resection via the retrosternal route at Nagasaki University Hospital and National Hospital Organization Nagasaki Medical Center between 2019 and 2022. All patients were male with a mean age of 73 years at the time of surgery. Three patients underwent video-assisted thoracoscopic esophagectomy, and one patient underwent open esophagectomy. The time between esophagectomy and surgery for GTC ranged from 3 to 13 years, and the clinical stages of GTC were I, I, IIA and IIB. The operation times for the gastric tube resections ranged from 155 to 190 min. All patients were discharged without major complications. The postoperative follow-up times ranged from 3 months to 3 years without recurrence.
Under general anesthesia, the patients were placed in the supine position with the left upper arm slightly extended and a cushion placed to raise the back. Ports were inserted in three places at the left axilla and three places at the abdomen, as shown in Fig. 1. A collar incision was added to the neck (Fig. 1).
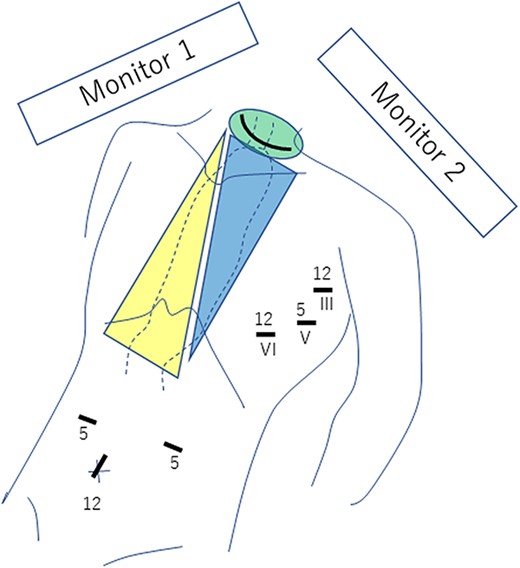
Three ports are placed in both the left chest wall and the abdomen, and a collar incision is performed in the neck. Roman numerals indicate the intercostal space, and the digits reflect the port size (in millimeters). The green area is dissected from the neck, the blue area from the chest and the yellow area from the abdomen. The thoracic surgeon views and operates with monitor 1 from the left side of the patient, while the abdominal surgeon views and operates with monitor 2 from the right side of the patient.
From the chest, the left wall to the posterior wall of the gastric tube was dissected by a chest surgeon, followed by the anterior wall to the right wall of the gastric tube from the abdomen. An abdominal surgeon then dissected the circumference of the esophagogastric anastomosis at the neck. The thoracoabdominal transition and the back of the sternum, where adhesions are particularly severe, were manipulated jointly using a two-directional view from the thoracic and abdominal cavities (Figs 2 and 3).
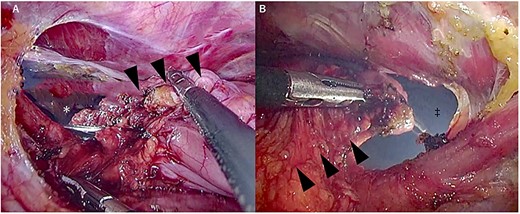
Manipulation around the thoracoabdominal transition. (A) Thoracic view, (B) Abdominal view. ▼ gastric tube, ▲ gastric tube, * abdominal cavity and ‡ thoracic cavity.
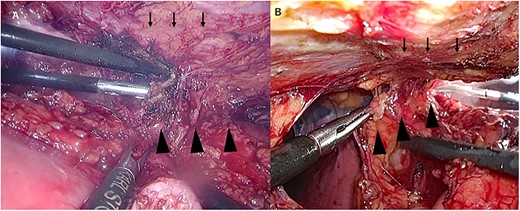
Manipulation around the sternum. (A) Thoracic view, (B) Abdominal view. ↓ sternum and ▲ gastric tube.
The cervicothoracic transition was also manipulated using a two-directional view from the neck and left thoracic cavity (Fig. 4).
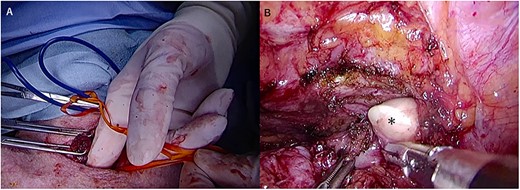
Manipulation around the cervicothoracic transition. (A) Cervical view, (B) Thoracic view. * Surgeon’s fingers during cervical manipulation.
The gastric tube was dissected at the neck in three patients, whereas in one patient, the adhesions at the cervicothoracic transition were so severe that dissection from the neck was judged to be dangerous; therefore, the gastric tube was dissected through the thoracic cavity, and the dissection was safely performed by pulling the end out through the neck. The gastric tube and lymph nodes were then pulled out completely into the abdominal cavity. The entire process of gastric conduit resection is shown in Supplementary Video 1. Subsequently, pedicled jejunal reconstruction via the presternal route and digestive tract anastomosis were performed, followed by vascular anastomosis in the neck performed by a plastic surgeon.
DISCUSSION
We propose a new surgical method for GTC resection using simultaneous thoracoscopy and laparoscopy.
Various methods can reduce the surgical invasiveness of a median sternotomy, such as lifting the sternum with a Kent retractor [11], transdiaphragmatic mediastinal manipulation under laparoscopy [12], hand-assisted thoracoscopic surgery via the right thoracic cavity [10], and thoracoscopic resection of the gastric tube via the left thoracic cavity [13]. However, no studies have reported combined resection for GTC using simultaneous thoracoscopic and laparoscopic approaches.
In our institution, EC surgery is usually performed by abdominal surgeons; however, thoracic surgeons are involved in the thoracic components of GTC resection. This surgical approach avoids the risks and pain associated with median sternotomy and has several advantages. First, the left thoracic cavity, a virgin surgical field, can be used for surgery using an artificial pneumothorax to obtain a good field of view. Moreover, although esophageal surgery is usually performed from the right thoracic cavity, thoracic surgeons often perform anterior mediastinum surgery from the left thoracic cavity, and thoracic surgeons are accustomed to approaching the mediastinum through the left thoracic cavity. Second, the operative field is closer to the mediastinum than that in a laparoscopic approach; because the approach is from the left side, the gastric tube does not interfere with the field of view compared with operation from the abdominal cavity, allowing relatively safe dissection around the innominate vein. The third and most useful advantage is the simultaneous operation from the thoracoabdominal region with double vision in areas with severe adhesions, such as the cervicothoracic transition, the back of the sternum, and the thoracoabdominal transition. Thus, this double vision approach can allow safe dissection in areas with a poor field of view or severe adhesions. Specifically, in the cervicothoracic transition area, neck manipulation of the dorsal side of the esophagogastric anastomosis, which normally has to be performed blindly owing to the presence of the gastric tube in the narrow surgical field, can be performed while viewing from the thoracic cavity. We believe this will lead to a reduction in surgical time.
A limitation to this approach is that the resulting opening between the thorax and abdomen causes difficulty in storing fluid in the thoracic cavity for the water sealing test. Thus, care must be taken not to damage the lung when inserting forceps. It is also difficult to use in combination with open abdominal surgery because the pneumothorax environment cannot be maintained.
This collaborative surgery for GTC with simultaneous access from the chest and abdomen/neck does not require a sternotomy, provides good surgical view from two directions at all times, and allows for safe dissection. Usually, abdominal surgeons perform GTC surgery; however, collaborating with thoracic surgeons may make the surgery safer.
AUTHORS’ CONTRIBUTIONS
Machino R: Conceptualization; Data curation; Investigation; Methodology; Visualization; Writing—original draft; Writing—review & editing. Kobayashi S: Conceptualization; Data curation; Methodology; Writing—review & editing. Tagawa T: Conceptualization; Data curation; Methodology; Project administration; Supervision. Taniguchi K: Conceptualization; Data curation; Supervision. Kanetaka K: Conceptualization; Data curation; Supervision. Eguchi S: Conceptualization; Supervision. Nagayasu T: Conceptualization; Supervision.
DATA AVAILABILITY
The datasets obtained during this study are available from the corresponding author on reasonable request.
FINANCIAL SUPPORT AND SPONSORSHIP
None.
CONFLICT OF INTEREST STATEMENT
None declared.
CONSENT FOR PUBLICATION
Informed consent was obtained from patients for the publication of this report and any accompanying images. Patient anonymities were maintained.



