-
PDF
- Split View
-
Views
-
Cite
Cite
Ruthwik Duvuru, Ahmad AlAwadhi, Faisal Nawaz, Aftab Ahmed, Afnan W M Jobran, Aya Zazo, A rare case of gastric duplication cyst in a male paediatric Caucasian patient: a case report, Journal of Surgical Case Reports, Volume 2022, Issue 10, October 2022, rjac436, https://doi.org/10.1093/jscr/rjac436
Close - Share Icon Share
Abstract
Anywhere in the alimentary canal, you can find a gastric duplication cyst, a spherical muscle formation lined by mucosal membrane. It is an uncommon example of a group of congenital intestinal abnormalities. Gastric cysts typically develop on the stomach’s greater curvature. A Caucasian 4-year-old boy came in with his family after experiencing colicky central stomach pain for 2 days, along with vomiting for 4 days, decreased oral intake, a temperature of up to 38.5°C and regular bowel movements. A region of the transverse colon with degraded and inflammatory serosa covered in omentum with black necrotic sections was seen during the procedure.
INTRODUCTION
Gastric duplication cyst (GDC) is a spherical muscular structure which is lined by mucosal membrane and can be found across the alimentary canal. It is part of a rare set of congenital alimentary tract anomalies, with an incidence of 1 in 4500 births, and higher predisposition among males [1]. GDC represents 4% of all alimentary canal duplications, and about two-thirds manifest within the first year of life [2]. A study by Wieczorek et al. reported that 70% of all GDC cases have been diagnosed in the first 12 years of life [3].
Clinical diagnosis of the GDC is predominantly in young children with symptoms of abdominal pain, gastric outlet obstruction or a palpable abdominal mass [4]. Endoscopic ultrasound (EUS) is the preferred modality for the evaluation and diagnosis of GDC as it can distinguish between solid and cystic lesions. On EUS, duplication cysts appear as anechoic and homogenous lesions with regular margins arising from the submucosal layer [5].
Surgical resection is the preferred method of treatment for most duplications of the alimentary canal, except for duodenal duplications because of their close proximity to the pancreatic and biliary ductal system. In such cases, drainage may be performed; however, the presence of gastric mucosa should be considered to prevent ulceration [6]. Laparoscopic resection of GDC has been described in both newborns [7] and adults [8], and cannot only be diagnostic but also avoid unnecessary abdominal laparotomy [9]. Untreated GDC can lead to pancreatitis due to the proximity of cysts with the pancreatic duct, along with unwanted neoplastic changes reported in some cases [6]. Long-term monitoring of GDC is generally done to assess for pancreatitis and recurrent bleeding risks after mucosal stripping [6].
CASE PRESENTATION
A 4-year-old Caucasian boy presented with a history of central abdominal pain for 2 days that was colicky in nature, vomiting for the past 4 days, decreased oral intake and fevers up to 38.5°C. The surgical history is significant for a mini-laparotomy and appendectomy with an uneventful recovery. The medical history is otherwise unremarkable. Vitals on presentation were as follows: temperature: 38.5°C; SpO2: 100%; heart rate: 105 BPM; blood pressure: 111/66; Height: 151 cm; weight: 14.4 kg; BMI: 6.31. The patient’s immunizations were up to date. No family history of any inherited conditions or gastrointestinal anomalies was found. On examination, abdominal tenderness in the left hypochondriac and left iliac region with guarding was present.
An ultrasound scan of the abdomen showed a 3.2 cm mass in the left upper quadrant. Further assessment was required, thus prompting the use of an IV CT scan. The CT scan confirmed the presence of a cystic mass, measuring about 3 cm × 2.5 cm anteriorly in the left flank in the intervening mesenteric fat between the colon and the small bowel, with enhancing thick walls measuring up to 5 cm. Intrinsic homogenous fluid content was noted with marked surrounding inflammatory fatty streaking. A provisional diagnosis of abscess with an infected cyst or bowel duplication cyst was made.
After obtaining surgical consent, the patient underwent an excisional laparotomy. During the procedure, an area of the transverse colon was noted to have serosal erosion and inflammation, covered by the omentum with black necrotic parts (Fig. 1). A cyst was present in the mesocolon which contained dark fluid presumed to be blood. Postoperatively, the patient recovered well and was discharged 2 days later (Fig. 2).
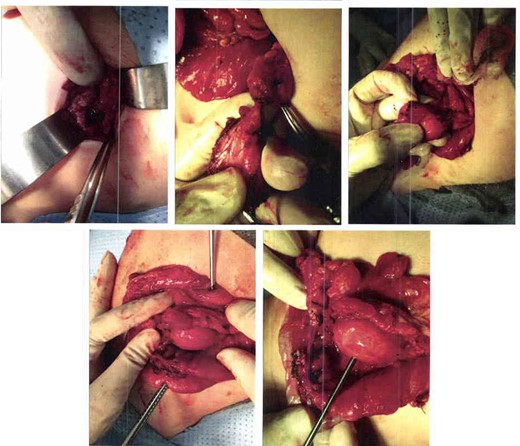
Excisional laparotomy, the area of the transverse colon as noted is inflamed and covered with necrotic part, below it a mesocolon cyst was noted too.
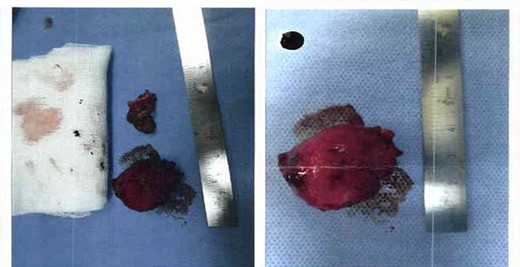
Excised cyst containing dark fluid assumed to be blood, it measured 3 cm by 2.5 cm, and sample was sent for histopathology to confirm diagnosis.
Histopathology
The biopsied specimen was sent to the lab for analysis in two parts with formalin-filled containers. The first container was labeled as ‘cyst’ and compromised a spherical fragment of purple-brown unoriented solid tissue with rubbery consistency measuring 3.2 cm × 2.5 cm × 0.3 cm. The specimen was inked in black. Serial sectioning shows a thick-walled cystic lesion, filled with transparent mucinous material and lined by benign antral type gastric mucosa. It was surrounded by a circular layer of smooth muscle tissue. Morphologic features of dysplasia or malignancy were not identified. The concurrent cytology showed acellular fluid.
The second container was designated as ‘omentum’ and was comprised of a single un-oriented fragment of thin membranous tissue with a smooth lining measuring 3 cm × 2.5 cm × 0.3 cm. The serially sectioned specimen showed leaf-shaped fragments of lobulated mature fibro-adipose tissue with a moderate degree of chronic inflammation, surface stromal fibrosis and focal mesothelial reactive proliferation. There was a single entrapped, small benign lymph node. Morphologic features of dysplasia or malignancy were not identified. Thus, a diagnosis of gastric cyst duplications was established.
The next follow-up was in 2 weeks for stitch removal and discussing the results of the histopathology and diagnosis with the parents. No further follow-ups were deemed necessary.
DISCUSSION
GDC is a rare anomaly that may occur at any site between the oral cavity and the rectum, with ileum being the most common site. Gastric cysts commonly occur on the greater curvature of the stomach [8].
GDC is essentially characterized by (i) a wall of the cyst which is continuous with the stomach wall; (ii) cyst is surrounded by smooth muscle which is continuous with that of the stomach and (iii) the cyst wall is lined by epithelium of gastric or any other type of gut mucosa. [2, 10, 11]. Our presented case exhibits similar characteristics.
Gastric duplications are usually diagnosed via pre-operative CT scan. In this patient, we had a provisional diagnosis of infected cyst or bowel duplication cyst. The histopathology report allowed for a confirmatory diagnosis of GDC [9].
The clinical presentation of gastric duplications varies from vague abdominal pain to nausea, vomiting, epigastric fullness, weight loss, anemia, dysphagia, dyspepsia with abdominal tenderness and also an epigastric mass on physical examination. However, from the above-mentioned characteristics, this patient solely presented with abdominal pain [12].
The go-to management of GDCs would be to completely excise the cyst to avoid any risk of possible complications such as obstruction, perforation, hemorrhage and malignancy [10]. In this patient, we completely excised the cyst which was subsequently sent to histopathology for confirmation.
CONCLUSION
In summary, gastric duplication is a rare diagnosis and may exhibit a gamut of possible symptoms upon presentation. However, due to the risk of malignancy, GDC should be part of the differential diagnoses for cystic lesions of the gastrointestinal tract. GDC should be treated surgically by complete excision due to the risk of malignant transformation. Further research is warranted to better understand the pathophysiology and risk factors to strategize timely interventions.
ACKNOWLEDGEMENT
The authors acknowledges the Dubai Hospital.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
DECLARATION OF PATIENT CONSENT
The authors declare that they have obtained the parents’ consent.



