-
PDF
- Split View
-
Views
-
Cite
Cite
Bayan O Besharah, Raghad A Ghazzawi, Haddad H AL-kaff, Sherif K Abdelmonim, Mohammad A Al-Essa, Reconstruction of facial dermatofibrosarcoma protuberans using an anterolateral thigh flap: a case report and literature review, Journal of Surgical Case Reports, Volume 2020, Issue 9, September 2020, rjaa340, https://doi.org/10.1093/jscr/rjaa340
Close - Share Icon Share
Abstract
Dermatofibrosarcoma protuberans is a rare, locally aggressive cutaneous malignancy with a high rate of recurrence if the head-and-neck region is involved. Treatment is challenging as it is difficult to avoid poor aesthetic and functional outcomes using the standard approach of complete resection with negative margins and subsequent reconstruction. This is especially challenging in cases where the face is involved and the defect is large after resection. There have been few cases of reconstruction using different flaps reported. We present the case of a young lady with a large cheek tumor, which was reconstructed using an anterolateral thigh flap. Excellent functional and aesthetic outcomes were achieved.
INTRODUCTION
Dermatofibrosarcoma protuberans (DFSP) is a rare cutaneous neoplasm of the dermis. It is slow growing but locally aggressive with the potential to deeply infiltrate underlying structures [1, 2]. Typically, DFSP develops between the ages of 25 and 50; however, it can occur at any age without difference in prevalence between genders [2]. Only 10–15% of cases affect the head-and-neck region; the trunk and extremities as the most common sites [3]. The involvement of the face is extremely rare and associated with high local recurrence, possibly due to the difficulty in operating on in this region because of complex vascular and neural structures. These mean that complete resection—the gold standard treatment—is challenging and the risk of disfiguration high [4]. Mohs micrographic surgery (MMS) or wide local excision (WLE) with negative surgical margins can be used for resection [3], and reconstruction is crucial for acceptable aesthetic and functional outcomes.
CASE REPORT
A 39-year-old Yamani woman was referred to us for right-cheek DFSP. The lesion began as a small nodule 2 years previously and increased in size over time. Recently, the patient experienced itching over her face, particularly around the lesion, with no associated pain or trismus. The patient had a history of facial trauma 20 years back, but no history of radiation treatment and no significant medical history.
Physical examination revealed a nodular mass of 4 |$\times$| 5 cm over the right cheek, surrounded by skin discoloration, and texture changes reaching up to the nasolabial fold anteriorly, lower eyelid superiorly and the level of the lower lip inferiorly. The nodule was hard and not fungating. Head-and-neck examinations including neck lymph nodes and cranial nerve assessments were unremarkable. All laboratory results were within normal ranges.
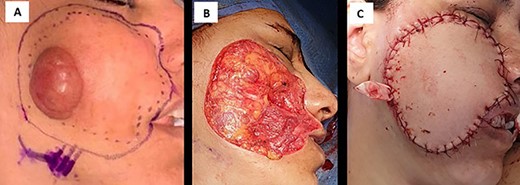
(A) intraoperative image shows right cheek mass, (B) postoperative defect in right cheek lesion and (C) anterolateral thigh flap completely covers the postsurgical defect.
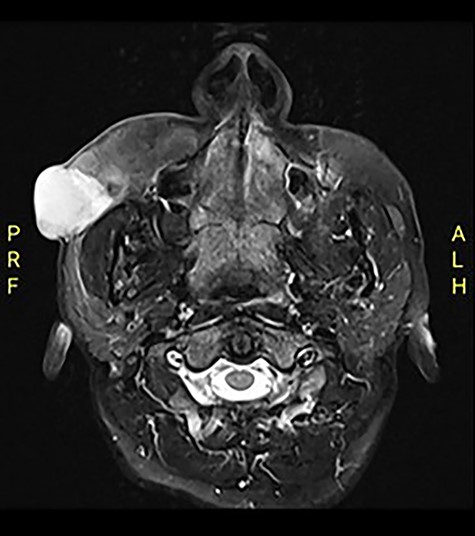
Computed tomography (CT) revealed an exophytic skin lesion on the right cheek reaching the buccal space with maximum transverse, anteroposterior and craniocaudal dimensions of 31, 28, 30 mm, respectively. No bone invasion was noted. Magnetic resonance imaging (MRI) indicated that the mass extended to the anterior surface of the masseter muscle, with parotid duct medialization due to mass effect (Fig. 1). The referred histopathology slide of the incisional biopsy was reviewed, and a diagnosis of DFSP was confirmed.
The case was discussed with a multidisciplinary team, and treatment options were discussed with the patient. WLE with negative margins was planned as we do not have access to MMS. Unfortunately, intraoperatively, the zygomatic branch of the facial nerve was found to be involved and had to be excised. An intraoperative frozen section confirmed negative margins, and the resulting defect was reconstructed using a harvested left antro-lateral thigh (ALT) flap. An anastomosis to the facial artery was created (Fig. 2) using the vein coupler for venous anastomosis. The donor site was closed primarily.
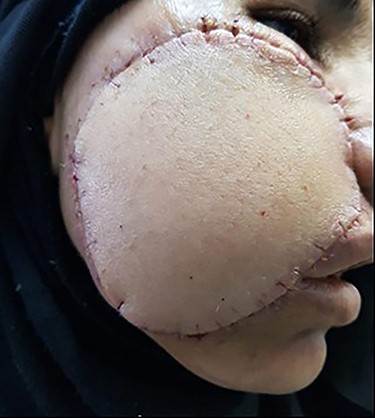
View of postoperative day 7 shows flap almost completely healed.
Histopathological examination confirmed a DFSP of 3 |$\times$| 2.5 |$\times$| 2.5 cm in size with close margins of less than 2 cm and reaching the deep-inked margin. There was no evidence of perineural or lymphovascular invasion.
The flap completely healed by postoperative day 14 with good face contouring (Fig. 3). All branches of the facial nerve were intact except the zygomatic branch, which was excised during the surgery, with the loss of nasolabial fold at the site of surgical incision without any impact on the eye. The patient underwent adjuvant radiotherapy for the right cheek lesion (60 Gy in 30 fractions) and followed for 6 months. There was no effect on visual filed, acuity and eye movement. Furthermore, no recurrence or residual disease was detected by the head CT scan. As the patient has a higher risk of local recurrence, she will be followed every 3 months for physical examination and every 6 months for radiological evaluation for the first 2 years and then every 6 months for the next 3 years, followed by annually for life.
DISCUSSION
DFSP is the most common cutaneous sarcoma; it accounts for 2–6% of all soft-tissue sarcomas [5]. The name DFSP was coined by Hoffman based on the nodular appearance of the tumors [6], which have indolent courses, initially appearing as reddish-blue to brown hard and indurated plaques. Lesions may increase to a typical size of up to 5 cm in diameter and may infiltrate underlying soft tissue. Tumors of over 10 cm are very uncommon [2, 6].
Summary for the reported cases of cheek DFSP reconstructed with regional or free flap; RFFF: radial forearm free flap, LTMC: lower trapezius musculocutaneous, PMMC: pectoralis major myocutaneous, ALT: antro-lateral thigh
| Study . | Tumor size . | Treatment . | Reconstruction . | Aesthetic function . |
|---|---|---|---|---|
| Foshee et al.1 | 3.7 × 4.8 cm | MMS | RFFF | Right eyebrow ptosis improved within 6 months |
| Wahid Terro et al.2 | 10 × 8 × 4 cm | WLE | LTMC flap | Limited neck range of motion improved within 7 weeks |
| Lokendra Kumar et al.3 | 15 × 10 × 13.7 cm | WLE | Supratrochlear flap + PMMC flap | Not elaborated |
| Gimena Castro Pérez et al.4 | 10 × 6 cm | WLE | PMMC flap | Not elaborated |
| R Van Der Rijt et al.7 | NA | WLE | RFFF | Not elaborated |
| Tommaso Agostini et al.10 | 4 × 5 cm | WLE | ALT free flap | Good contour but with color mismatch |
| Tommaso Agostini et al.10 | 7 × 5 cm | WLE | ALT free flap | Died with pulmonary embolism |
| Study | Tumor size | Treatment | Reconstruction | Aesthetic function |
|---|---|---|---|---|
| Foshee et al.1 | 3.7 × 4.8 cm | MMS | RFFF | Right eyebrow ptosis improved within 6 months |
| Wahid Terro et al.2 | 10 × 8 × 4 cm | WLE | LTMC flap | Limited neck range of motion improved within 7 weeks |
| Lokendra Kumar et al.3 | 15 × 10 × 13.7 cm | WLE | Supratrochlear flap + PMMC flap | Not elaborated |
| Gimena Castro Pérez et al.4 | 10 × 6 cm | WLE | PMMC flap | Not elaborated |
| R Van Der Rijt et al.7 | NA | WLE | RFFF | Not elaborated |
| Tommaso Agostini et al.10 | 4 × 5 cm | WLE | ALT free flap | Good contour but with color mismatch |
| Tommaso Agostini et al.10 | 7 × 5 cm | WLE | ALT free flap | Died with pulmonary embolism |
Their diagnosis is confirmed by pathological examination, while MRI is used to detect their extent of invasion and CT to confirm suspected bone invasion [6]. Treatment by MMS has advantages over WLE, particularly easier histological evaluation of borders enabling improved tissue preservation and a smaller surgical defect, with consequent improvements in the postoperative aesthetic appearance and reduced recurrence rate (1–8% for MMS versus 7.3–30.8% for WLE) [1, 4, 5].
The risk of adverse neurological defects associated with DFSP of the head and neck is high, especially when the face (H-zone) is involved. Injury from direct tumor invasion or during surgical excision can also result in facial nerve injury, which is categorized into grades estimate postoperative neurological outcomes. Typically, function of the facial nerve recovers within 6 months, although this depends on interconnections of the bilateral facial nerves [1]. Few reports have documented the postoperative status of the facial nerve after surgical resection of DFSP; therefore, it is important to publish information on this factor to enable surgeons to counsel patients about potential surgical complications [1].
Long-term follow-up is critical as 30% of local recurrences occur beyond 5 years postoperatively [7]. Van Der Rijt et al. [8] reported the risk of local recurrence to be 50–75%, with the majority appearing in the first 3 years postoperatively. However, distant metastasis is seen in only 4% of patients, mainly to the lungs and lymph nodes [3, 7].
Radiotherapy can be used as adjuvant therapy in cases with positive margins in the critical area when surgical re-excision is not possible. In addition, systemic therapy such as imatinib has been reported to have high efficacy in advanced cases [7].
The selection of reconstructions depends on the factors including skin laxity, smoking status, sun exposure, patient age, tumor location and size [9]. Primary closure can be performed for small cheek defects, while larger defects require further surgical treatment in order to avoid distortion of anatomical structures. For example, a local flap is suitable for small-to-moderate-sized cutaneous cheek defects, while complex or very large defects require distal flaps covering the functional and cosmetic parts of the reconstruction [9]. Only seven cases of large facial DFSPs have been reported in the literature; various types of distant flaps have been used to treat these, and the approaches are summarized in Table 1.
A study by Tommaso Agostini et al. [10] reported cheek reconstruction using a free ALT flap to improve functional and aesthetic outcomes; hence, better postoperative quality of life.
CONCLUSION
Facial DFSP is a rare disease; options of reconstructions depend on many factors and can affect the patient’s quality of life. Our case of large cheek DFSP was reconstructed by using ALT flap. The good aesthetic and functional outcomes were achieved.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.



