-
PDF
- Split View
-
Views
-
Cite
Cite
Joana Raquel Rodrigues Gaspar, Paula Marques, Isabel Mesquita, Mário Marcos, Jorge Santos, Carlos Nogueira, Laparoscopic treatment of a gastro-gastric fistula after Roux-en-Y gastric bypass—report of two cases, Journal of Surgical Case Reports, Volume 2020, Issue 12, December 2020, rjaa478, https://doi.org/10.1093/jscr/rjaa478
Close - Share Icon Share
Abstract
The most frequently performed bariatric surgery is the laparoscopic Roux-en-Y gastric bypass (LRYGB). An uncommon complication of LRYGB is gastro-gastric fistula (GGF). Possible causes of GGF include incomplete transection of the stomach during the initial surgery, staple-line leaks in the post-operative period and marginal ulcers. The optimal management of GGF is still under debate, with medical, endoscopic and surgical treatment modalities available. The authors present two cases of a GGF successfully managed with a laparoscopic surgical approach, after failed medical and endoscopic treatment.
INTRODUCTION
Obesity has become a major health issue worldwide [1–3]. Obese patients are at risk for several diseases including diabetes, arterial hypertension, dyslipidaemia, metabolic syndrome, sleep apnoea, deep vein thrombosis and cancer [1, 2].
For morbidly obese patients, bariatric surgery remains the only reliable treatment to achieve appropriate and sustained weight loss [4–6]. The most frequently performed bariatric procedure is the laparoscopic Roux-en-Y gastric bypass (LRYGB) due to its excellent results and low morbidity rate [1–7].
A well-documented complication of LRYGB is gastro-gastric fistula (GGF) with an estimated incidence ranging from 0 to 6% [1–9]. The most widely accepted aetiologies for GGF are incomplete transection of the stomach during the initial surgery, staple-line leaks in the post-operative period and marginal ulcers [1, 3–7].
GGT may be completely asymptomatic or present itself with non-specific symptoms including abdominal pain, nausea, vomiting, gas bloat, pyrosis and inadequate weight loss or weight regain [1, 3–9]. While asymptomatic and mildly symptomatic patients can generally be treated conservatively, the optimal management strategy for patients with severe or persistent symptoms is yet to be defined [3, 4, 6–8]. Several treatment strategies (including endoscopic, surgical and combined procedures) have been proposed to manage these patients [1, 3, 4, 6–10].
The authors present two cases of a GGF successfully treated with laparoscopic surgery, after failed medical and endoscopic treatment.
CASE 1
A 44-year-old male, former smoker (total pack-year of 25), with sleep apnoea and a previous history of laparoscopic adjustable gastric banding in 2003, underwent conversion to LRYGB in October 2016 due to weight regain (body mass index (BMI) of 53.3 kg/m2 previously to reoperation).
The patient presented to the emergency room with complains of persistent epigastric pain and occasional vomiting in May 2019. At the time, the patient had a BMI of 26.1 kg/m2, having lost a total of 85 kg since the LRYGB. The only abnormal finding upon physical examination was epigastric tenderness. The laboratory tests were unremarkable.
The computed tomography (CT) scan with oral contrast revealed a GGF (see Fig. 1). The upper gastrointestinal endoscopy confirmed a GGF and also demonstrated a stenosis of the gastro-jejunal anastomosis (GJA) (see Fig. 2).
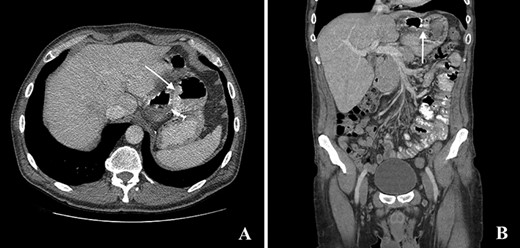
Computed tomography with intravenous contrast images demonstrating a gastro-gastric fistula (arrows). A: axial reconstruction; B: sagittal reconstruction.
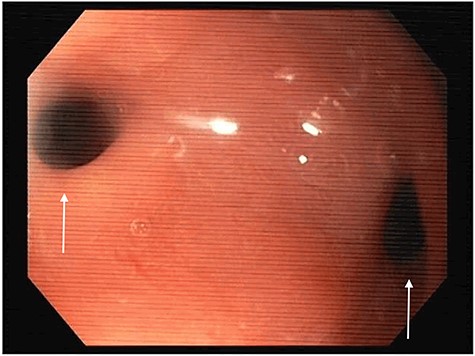
Esophagogastroduodenoscopy image showing a gastro-gastric fistula (on the right) and stenosis of the gastro-jejunal anastomosis (on the left), marked with arrows.
The stenosis of the GJA was dilated with a balloon and the proximal orifice of the GGF was closed with clips, endoscopically (see Fig. 3). Oral feeding was reintroduced with tolerance and the patient was discharged, 2 days after the procedure, medicated with oral proton pump inhibitors (PPIs).
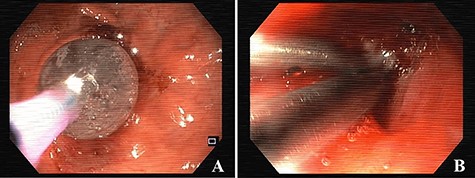
Esophagogastroduodenoscopy images showing dilation of the gastro-jejunal anastomosis (A) and closure of gastro-gastric fistula with clips (B).
Three months after endoscopic treatment, the patient started complaining of epigastric pain again, without other gastrointestinal symptoms or weight regain. An upper gastrointestinal (GI) contrast swallow study showed recurrence of the GGF and GJA stenosis (see Fig. 4).
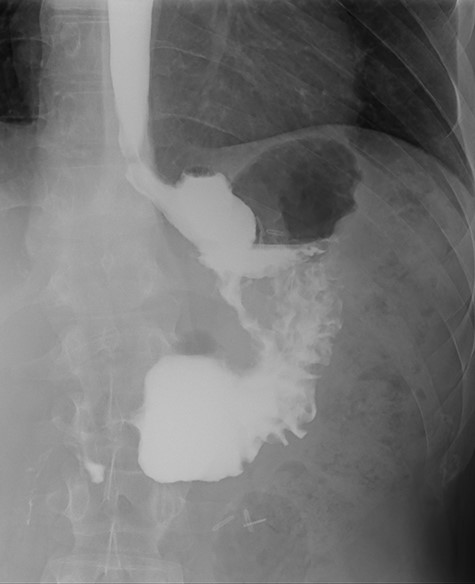
Upper gastrointestinal contrast swallow study image where it is possible to see a gastro-gastric fistula with opacification of the excluded stomach.
Due to the failure of the endoscopic treatment, the patient underwent laparoscopic surgery, immediately after a new endoscopic dilatation of the GJA. Proximal gastrectomy (including remnant gastrectomy with unblock resection of the GGF and GJA) followed by reconstruction of the GJA was performed (see Fig. 5). The post-operative period was uneventful and the patient was discharged 6 days later.
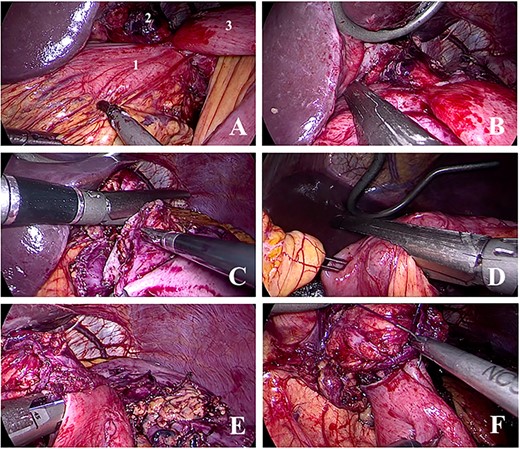
Images from the surgical procedure. A: Identification of the excluded stomach (1), gastric pouch (2) and jejunum (3). B: Section of the gastric remnant distal to the fistula. C: Section of gastric pouch proximal to the gastro-jejunal anastomosis and the gastro-gastric fistula. D: Section of the jejunum distal to the gastro-jejunal anastomosis. E: Reconstruction of the gastro-jejunal anastomosis—mechanical part. F: Reconstruction of the gastro-jejunal anastomosis—manual part (closure of the orifice from the stapler).
At the 6-month follow-up, the patient remains asymptomatic and has a stable BMI.
CASE 2
A 47-year-old male, non-smoker, with multiple comorbidities (including arterial hypertension, dyslipidaemia, type 2 diabetes and sleep apnoea), underwent LRYGB and a partial atypical gastrectomy due to morbid obesity (BMI 42.4 kg/m2) and a leiomyoma of the gastric fundus in May 2015.
A partial dehiscence of the GJA was diagnosed 3 days after the initial surgery and a second surgery was performed—closing of the dehiscence with sutures and insertion of a jejunostomy tube for enteral feeding.
The patient had a prolonged hospital stay due to complications, including organ disfunction associated with sepsis, pneumonia, central venous catheter infections and oesophageal fistula (originating in the superior limit of the staple line of the atypical gastrectomy of the fundus).
Several attempts to occlude the oesophageal fistula with endoscopic stents were performed. Initial stenting with a covered biodegradable stent was performed 8 weeks after the reoperation. One week later, distal migration of the stent was identified; due to partial incorporation, it was not possible to reposition the stent. A second attempt, using a completely recovered metal stent, was performed one week later. Five days after the second attempt, proximal migration of the second stent was identified and the stent was removed.
Due to resolution of the sepsis and associated organ dysfunctions, in spite of the persisting oesophageal fistula, the patient was discharged with exclusive enteral feeding through the jejunostomy tube, 12 weeks after the LRYGB, with a BMI of 29.6 kg/m2.
A last endoscopic treatment attempt of the oesophageal fistula was made 8 weeks after hospital discharge (October 2015), whereby a covered biodegradable stent was used and stabilized in place with clips (see Fig. 6). The success of the last endoscopic approach was confirmed with upper GI contrast swallow study (see Fig. 7). Oral feeding was reintroduced with tolerance.
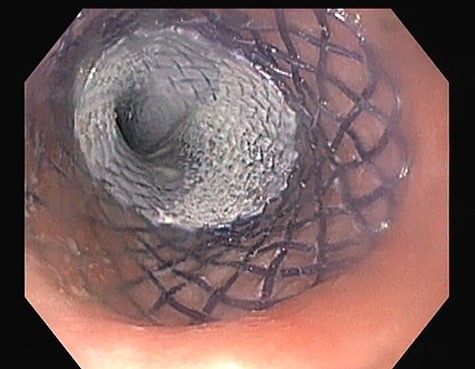
Esophagogastroduodenoscopy image showing a covered biodegradable stent occluding the proximal orifice of the oesophageal fistula.
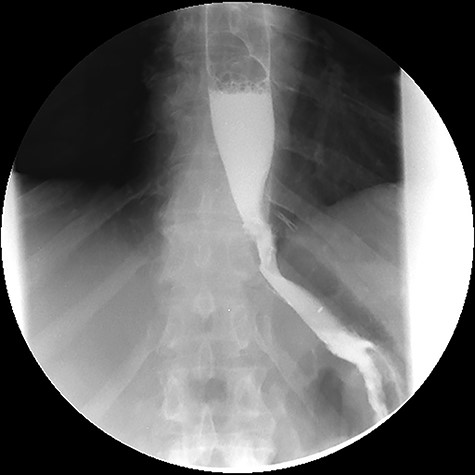
Upper gastrointestinal contrast swallow study image where is possible to see normal progression of contrast from the oesophagus to the stomach without leaks.
In December 2015, the patient presented to the emergency room complaining of pain in the left hypochondrium and anorexia. Upon physical examination, the only abnormal finding was tenderness to palpation of the left hypochondrium. The laboratory tests revealed an elevated white blood cells count (18.88 × 103/μL) and an elevated C-reactive protein (263.66 mg/L). A CT scan of the abdomen revealed a GGF with an associated abscess (see Fig. 8).
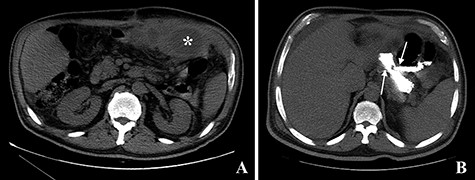
Computed tomography images. A: Image obtained prior to administration of contrast showing an abscess on the left upper quadrant of the abdomen (asterisk). B: Image obtained after administration of oral contrast showing progression of contrast through the gastro-jejunal anastomosis and through the gastro-gastric fistula (arrows).
After treatment of the abdominal abscess with percutaneous drainage and antibiotics, an upper GI contrast swallow study confirmed persistency of the GGF without any spillage of contrast to the abdominal cavity (see Fig. 9). The patient was discharged, medicated with oral PPIs.
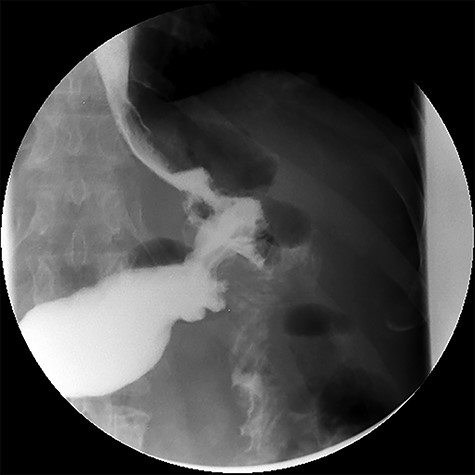
Upper gastrointestinal contrast swallow study image showing progression of contrast from the oesophagus to the gastric pouch and from the gastric pouch to the excluded stomach, confirming a gastro-gastric fistula.
After 18 months of follow-up, due to weight regain (BMI 37.1 kg/m2) in the presence of a GGF, the patient underwent laparoscopic surgery. Proximal gastrectomy (including remnant gastrectomy with unblock resection of the GGF) was performed. There were no complications in the post-operative period and the patient was discharged after 6 days.
At the 24-month follow-up, an upper endoscopy confirmed integrity of the stomach pouch and GJA without any GGF (see Fig. 10). Thirty-six months after surgical treatment of the GGF, the patient remains asymptomatic and has a stable BMI.
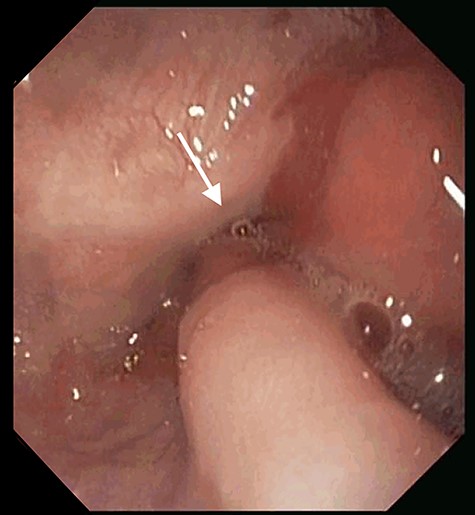
Esophagogastroduodenoscopy image showing a normal gastric pouch with only one orifice corresponding to the gastro-jejunal anastomosis (arrow).
DISCUSSION
Albeit uncommon, GGF is an important complication of LRYGB. Some authors defend that asymptomatic or mildly symptomatic patients with adequate weight loss may be treated conservatively or require no treatment at all [3, 4, 6–8]. Arguably, the incidence of GGF may actually be much higher than reported since diagnosis can only be made with appropriate radiological or endoscopic exams, which may not be routinely performed after LRYGB [7]. This supports a conservative initial approach of uncomplicated GGF.
Several endoscopic techniques have been described to address GGF, including endoclips, plugs, fibrin sealant, covered oesophageal stents, endoscopic suturing systems and coagulation [3, 10]. These techniques seem feasible and safe but are associated with low success rates [4, 6, 10]. Nevertheless, since a second surgery may carry a higher risk for potential life threating complications, an initial endoscopic approach is reasonable and should be attempted [3, 4, 6, 10].
Surgery remains the gold standard of care for large GGF and persistent GGF after medical and endoscopic treatments [3, 4, 6]. The best and most consistent outcomes described throughout the literature are obtained with remnant gastrectomy with or without revision of the GJA [4, 6–9].
Combined approaches (including surgery, endoscopy and fluoroscopy) have been described and seem promising but further studies need to be carried out to prove their effectiveness and safety [1, 4].
CONFLICT OF INTEREST STATEMENT
None declared.



