-
PDF
- Split View
-
Views
-
Cite
Cite
Catherine Denkler, Helene M Sterbling, Hani Seoudi, Tension pneumoperitoneum following upper endoscopy, Journal of Surgical Case Reports, Volume 2020, Issue 11, November 2020, rjaa401, https://doi.org/10.1093/jscr/rjaa401
Close - Share Icon Share
Abstract
Perforation of the digestive tract is a rare complication of endoscopy. Massive accumulation of air within the peritoneum resulting in the abdominal compartment syndrome is much less common with <20 cases reported. In this report we present a case of jejunal perforation during an upper gastrointestinal endoscopy that resulted in tension physiology with mesenteric ischemia, severe acidosis, renal failure, coagulopathy and massive gastrointestinal hemorrhage. The patient had a sudden onset of shock as soon as her abdomen was decompressed, indicating that she possibly developed a reperfusion injury. She did not respond to resuscitative efforts and ultimately died.
INTRODUCTION
Iatrogenic perforation of the digestive tract after upper gastrointestinal (GI) endoscopy is a rare occurrence with incidences of <1% [1]. The management of pneumoperitoneum after endoscopic perforation can be non-operative or operative depending on the size of the perforation, clinical symptoms and hemodynamic stability. Tension pneumoperitoneum (TPP) has been defined as life threatening severe intra-abdominal hypertension due to massive accumulation of intraperitoneal air [2]. TPP can result in a constellation of clinical symptoms including abdominal distension, dyspnea and signs of venous congestion [2–4]. As TPP progresses, it can lead to hemodynamic instability and abdominal compartment syndrome (ACS), demanding urgent intervention.
CASE REPORT
A 46-year-old woman with a body mass index of 43 and a past surgical history of Roux-en-Y gastric bypass 6 years prior presented to the bariatrics clinic with a chief complaint of weight gain of over 30 pounds over a few months. An upper GI series was suspicious for a fistula between the gastric pouch and gastric remnant. The patient was referred to gastroenterology for upper endoscopy. Endoscopy was uneventful and detected no abnormalities. The gastro-jejunal anastomosis had an adequate caliber (Fig. 1), and there was no evidence of gastric fistula (Fig. 2).
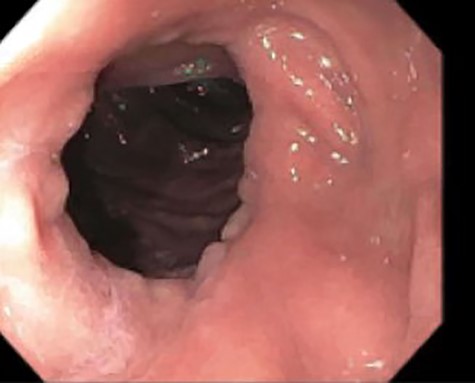
Esophagogastroduodenoscopy (EGD)–gastrojejunostomy, staple line, with no abnormal findings.
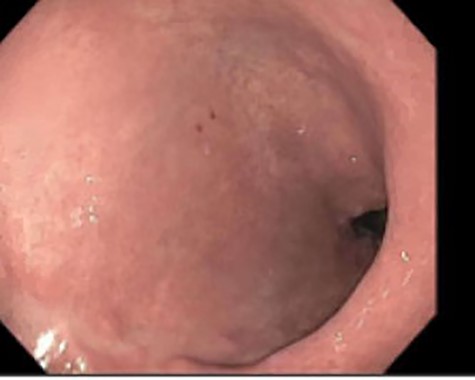
As the patient emerged from sedation, she experienced respiratory distress requiring intubation and her abdomen was noted to be distended. Abdominal X-ray revealed massive pneumoperitoneum (Fig. 3). The patient was taken to the intensive care unit (ICU) where she remained hemodynamically stable and with normal airway pressures. Attempts at Foley catheter placement in the ICU were unsuccessful. Approximately 2 h following transfer to the ICU she was taken to the operating room for exploratory laparotomy. A urologist was able to insert a Foley catheter before the operation and the bladder was noted to be empty.
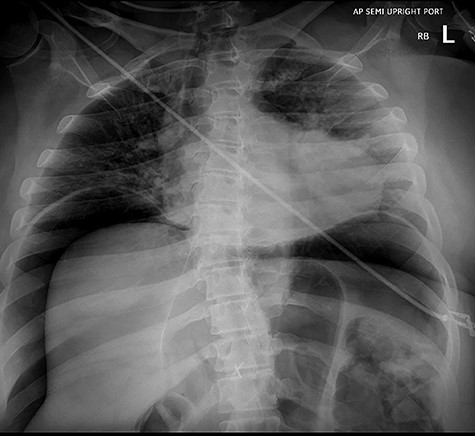
Abdominal X-ray revealing massive pneumoperitoneum after endoscopy.
Abdominal exploration was started laparoscopically. Upon incising the midline fascia a large amount of air came out under pressure. The patient abruptly became hypoxic, hypotensive and the previously normal end tidal CO2 was noted to have increased to 90 mmHg, despite the fact that abdominal insufflation with CO2 had not yet started. At this time, a midline incision was made to explore the abdomen. Diffuse bowel ischemia and distension of both the small and large intestines were observed. A 1-cm perforation with dark sanguineous output was noted at the jejuno-jejunal anastomosis staple line. A large amount of intraluminal blood was suctioned through the perforation. A segmental small bowel resection containing the anastomosis was performed (Fig. 4). Intraoperative blood testing revealed a hematocrit of 23%, lactate 7.8 mmol/l, potassium 8.0 mEq/l and blood glucose 3.0 mg/dl.
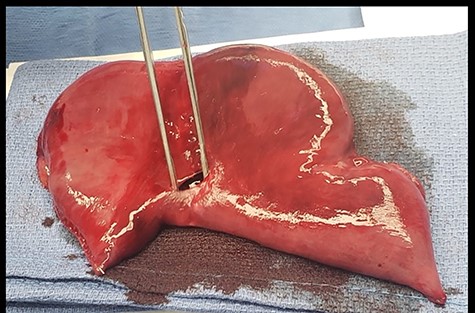
Resected specimen of jejuno-jejunal anastomosis with perforation.
Given the persistent dark discoloration of the large and small bowel consistent with a low-flow state, the abdomen was left open with the small bowel in discontinuity. Despite continued resuscitation and transfusion of blood products in the operating room, the patient remained hypotensive, hypothermic, coagulopathic and severely acidotic with lactate levels rising to 16 mmol/l. She was taken to the ICU where continued resuscitative measures were unsuccessful, and the patient died within hours.
DISCUSSION
TPP is a rare but potentially lethal complication of upper GI endoscopy. The sequence of events in this case suggests that bowel perforation happened as a result of intraluminal air accumulation during endoscopy leading to TPP and respiratory failure. Surgical consultation was obtained as soon as pneumoperitoneum was noted on the X-ray. The sudden onset of hypercarbia and hypotension combined with lactic acidosis and hyperkalemia suggested a reperfusion phenomenon coinciding with the release of the TPP. Severe hypoglycemia was most likely due to liver failure. The massive intraluminal bleeding observed was consistent with mucosal sloughing secondary to mesenteric ischemia. Ultimately, the combination of hemorrhagic shock and a fulminant reperfusion insult led to an irreversible systemic inflammatory response with multiple organ failure including acute lung injury, hepatic failure and severe coagulopathy.
Intra-abdominal pressure can be measured through the Foley catheter. Bladder pressure >20 mmHg represents intra-abdominal hypertension. Bladder pressure >30 mmHg is defined as ACS, which is typically associated with elevated airway pressures, respiratory failure and oliguria. This patient had respiratory failure, but her airway pressures were normal, and we could not assess her urine output or bladder pressures due to inability to insert a Foley catheter. Alternative methods for measurement of intra-abdominal pressure have been proposed using nasogastric and inferior vena cava pressure monitors and can be considered when bladder pressure is unobtainable [5].
In a report of post-colonoscopy TPP, Launey et al. [6] showed near-complete compression of the inferior vena cava, portal vein, abdominal aorta, mesenteric vessels, as well as total interruption of iliac arteries blood flow on computed tomography angiogram. A similar phenomenon likely occurred in our patient as evidenced by mottled lower extremities, fulminant multi-organ failure upon release of TPP and subsequent coagulopathy. Malbrain et al. [7] described the GI effects of increased intra-abdominal pressure including decreased mesenteric blood flow, decreased mucosal perfusion and intestinal edema, all of which were observed in our patient.
Published case reports have advocated for urgent needle decompression in TPP, highlighting its utility either as a definitive treatment of TPP or a bridging therapy to operative intervention [1–4]. In our patient we chose not to perform needle decompression, given that the airway pressures were normal and that she was hemodynamically intact.
In conclusion, TPP is a diagnosis that necessitates urgent recognition and treatment. Alternative intra-abdominal pressure measurement methods should be explored when bladder pressure is unobtainable and needle decompression should be considered early once the diagnosis is made. Left unaddressed TPP can progress to ACS, leading to end organ dysfunction. Decompression of TPP can unmask the dramatic consequences of multi-organ hypoperfusion potentially resulting in a fatal reperfusion injury. Appropriate resuscitative measures should be administered promptly as the decompression is performed.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.
References
- gastrointestinal tract vascular insufficiency
- acidosis
- blood coagulation disorders
- kidney failure
- upper gastrointestinal endoscopy
- endoscopy
- gastrointestinal bleeding
- reperfusion injury
- resuscitation
- shock
- abdomen
- jejunum
- peritoneum
- physiology
- pneumoperitoneum
- gastrointestinal tract
- abdominal compartment syndrome



