-
PDF
- Split View
-
Views
-
Cite
Cite
Bastiaan P. Vierhout, Jeroen M. Smit, Clark J. Zeebregts, Covering of an exposed vascular graft in the groin with an external oblique muscle rotational flap, Journal of Surgical Case Reports, Volume 2017, Issue 2, February 2017, rjx009, https://doi.org/10.1093/jscr/rjx009
Close - Share Icon Share
Abstract
Abdominal muscles, such as the oblique- and transverse muscles, find their blood supply from multiple segmental pedicles from the iliac artery. Besides its superior vascularization, its release is simple, leaving two abdominal muscles for securing abdominal wall strength. The release of the muscle and coverage of the graft requires partial muscle mobilization and is a minor reconstruction, but extension of the mobilization cranially enables coverage of larger defects. We present a case of an infected vascular graft in the groin successfully preserved through coverage with an external oblique muscle flap.
INTRODUCTION
Vascular graft salvage is successfully manageable with a muscle flap, following wound debridement and targeted antimicrobial therapy, in 78–84% of groin infections [1–3]. In this context, four types of flaps have been described, including transposition of the sartorius muscle [4, 5], the gracilis muscle [5, 6], the rectus abdominis and rectus femoris muscle [5]. In complicated cases these flaps may, however, not be feasible because of previous use or a compromised vascular supply as their pedicle is distally from the groin. We report a case in which the external oblique muscle was used to salvage an infected vascular graft.
CASE REPORT
An 81-year-old woman, who had been given an aortobifemoral Dacron prosthesis in 2007 for occlusive aortoiliac disease, was seen at our clinic with a persisting medial malleolar wound in September 2014. The wound was the result of a fall 2 months earlier. Thrombosis of the left iliac part of the aorta-bifemoral graft explained the insufficient healing. An unsuccessful attempt to recanalize the graft percutaneously required a reconstruction with a femoro-femoral crossover bypass in October 2014. She recovered well from this procedure and was discharged home 9 days later.
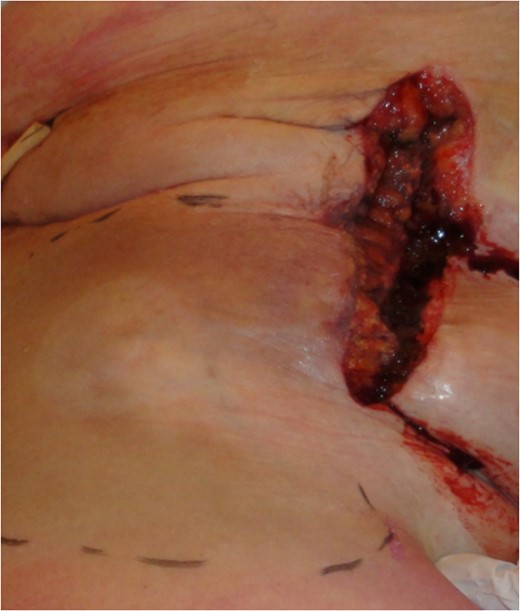
Macroscopic appearance after draining the hematoma and muscle transposition of the sartorius muscle.
Bacterial culture confirmed infection with aerobic- and anaerobic bacteria (Proteus vulgaris, Citrobacter koseri and Bacteroides fragilis). In line with the sensitivity of these microorganisms, treatment was started with Piperacilline/Tazobactam. Duplex ultrasound examination 1 week later showed a patent bypass and computed tomography 10 days thereafter did not show any fluid surrounding the graft. White blood cell count remained normal, and C-reactive protein (CRP) dropped from 111 mg/l to 52 mg/l after 2 months. However, a recurrent hematoma developed distally from the wound in the right groin.
Subsequently, a re-exploration of the right groin was performed. The hematoma did not seem macroscopically infected; it was drained through the previous incision and through a new incision laterally, thus diverting the infection away from the graft. The wounds were left open.
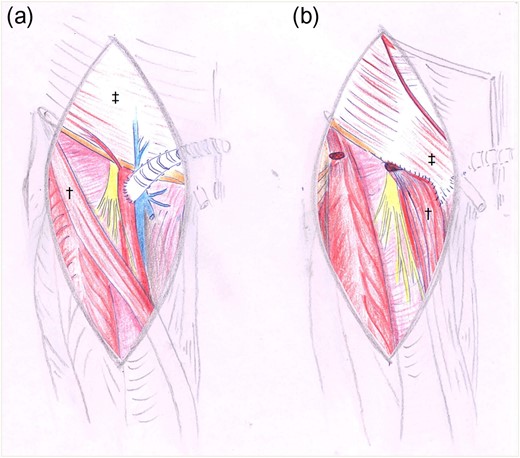
Drawings before (a) and after (b) reconstruction with sartorius†- and oblique external‡ flap.
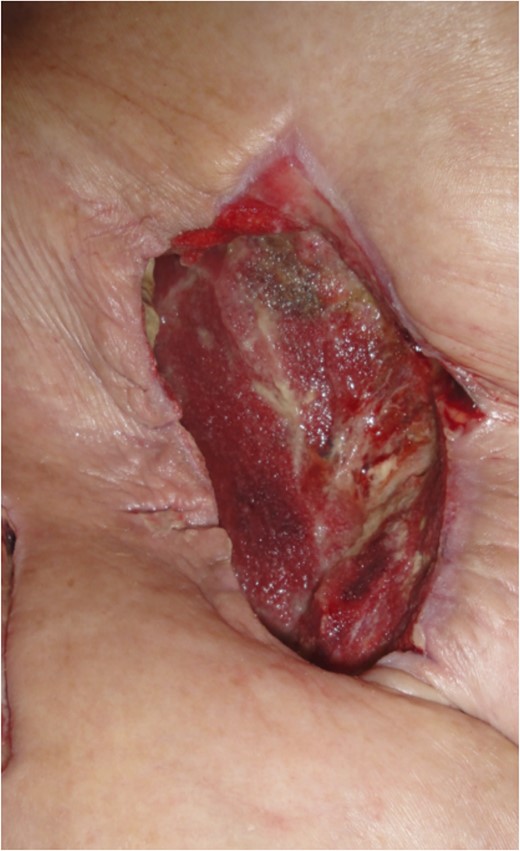
Appearance of the wound after removal of negative pressure, 4 days after the oblique external flap.
The patient was discharged home 6 days postoperatively and VAC-therapy was continued for 2 months, partially at home. The wound was closed and antibiotics were discontinued.
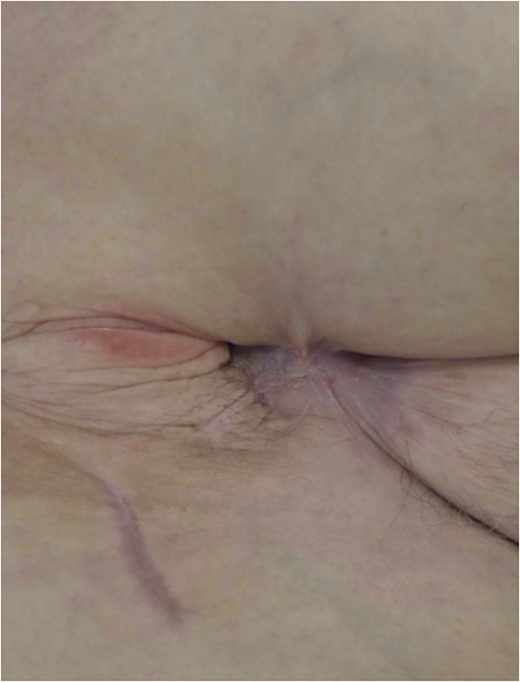
Approximately half a year post oblique flap; the wound is fully healed without a fistula.
DISCUSSION
This report indicates that in complicated cases coverage of an infected vascular graft in the groin with the external oblique muscle flap can help preserve the graft and assist in wound closure. The external oblique muscle is a Type V muscle with both dominant and multiple segmental vascular pedicles [7]. The dominant pedicles are one or two branches of either the deep circumflex iliac artery (94.7%) or the iliolumbar artery (5.3%) [8]. The segmental vascular supply is derived from the 5th through 12th posterior intercostal arteries. The use of the external oblique muscle has predominantly been described for chest wall reconstruction [9].
Advantages that the external oblique muscle flap offers are its simple release and its lateral vascularization, facilitating rotation. Even though it is a very thin muscle, it seems to have the ability to grow in an infected environment to form a large bulky protection of the synthetic bypasses. Three days after muscle rotation, the wound of our patient had significantly improved. The vascularization of the external oblique muscle and the non-smoking status of the patient contributed to the favorable outcome [10].
Because of the extension of the external oblique muscle cranially, further mobilization is possible, thus enabling coverage of larger areas in the infected groin.
One other advantage may be derived from its cranial position and its blood supply; the viability of the flap does not depend on the vascularization through a (infected) bypass graft, as is the case in sartorius-, rectus femoris- and gracilis flaps. Also, its blood supply is not dependent on the epigastric artery, essential for rectus femoris flaps. This epigastric/mammary artery is often used in coronary artery bypass surgery (CABG) in patients with peripheral artery disease.
To the best of our knowledge, external oblique muscle coverage of a vascular graft has not been described previously and is a feasible, effective, technically simple and safe procedure. It is an attractive option in complicated cases, in which previous muscle transpositions were unsuccessful. It can cover large vascular grafts in the groin, particular in patients with poor vascularization distally and in post-CABG patients. A larger series including functional long-term outcomes are required to confirm the above.
FUNDING
None of the authors has a financial interest in any of the products, devices or drugs mentioned in this manuscript.
ACKNOWLEDGEMENTS
We thank Paulien van Twuyver for proof reading and correcting the grammar and spelling in this article.



