-
PDF
- Split View
-
Views
-
Cite
Cite
Trần Thiết Sơn, Phan Tuấn Nghĩa, Đặng Phương Nam, Brachet Michel, Bey Eric, Single-stage elbow reconstruction using thinned anterolateral thigh flaps: technical insights from perforator anatomy, Journal of Surgical Case Reports, Volume 2026, Issue 4, April 2026, rjag222, https://doi.org/10.1093/jscr/rjag222
Close - Share Icon Share
Abstract
Reconstruction of complex soft-tissue defects around the elbow requires both durable coverage and preservation of joint mobility. Although the anterolateral thigh (ALT) flap is a versatile option, its bulk often limits range of motion (ROM) and results in less aesthetic outcomes. This study presents a refined, single-stage thinning technique for ALT flaps, guided by perforator anatomy. The thinning strategy was determined by the perforator’s characteristics. The orientation and number of perforators dictated the specific thinning method employed. Preservation of a fascial island surrounding the perforator maintained the flap’s blood supply. The clinical utility of this technique is illustrated by two representative cases involving severe post-traumatic and post-burn elbow contractures. Single-stage thinned ALT flaps, customized using perforator-based algorithms, offer a reliable, pliable, and aesthetically favorable solution for elbow reconstruction. This method enables immediate functional gliding of the underlying structures and reduces the need for secondary procedures, thereby effectively restoring joint mobility.
Introduction
The reconstruction of soft-tissue defects around the elbow remains a formidable challenge in plastic surgery. The requirement for a thin, pliable, and durable covering is paramount to facilitate the wide range of motion and the complex gliding mechanisms of the underlying tendons and nerves [1–3]. While the anterolateral thigh (ALT) flap has emerged as a workhorse for such reconstructions due to its versatile skin paddle and long pedicle, its inherent bulkiness often results in poor aesthetic contour and mechanical obstruction of the joint [2, 4]. In some cases, the ALT flap is thin enough that it is no longer a concern for surgeons. However, in many cases, a thick ALT flap still requires secondary debulking procedures to optimize functional outcomes. Recent advancements have shifted the focus toward immediate, single-stage thinning of the ALT flap [5]. However, the safety of aggressive thinning remains a subject of debate due to the risk of vascular compromise [6]. In this article, we describe a technique for reconstructing an elbow defect using the ALT flap, with a refined technique for thinning method. Central to our approach is a personalized thinning strategy guided by the perforator's anatomical structure.
Surgical technique
The surgical procedure begins with the radical excision of the elbow scar tissue, and the dimensions of the required defect coverage are measured.
The ALT flap is then harvested in the subfascial plane, allowing a meticulous survey of the perforator anatomy. For sufficiently thin ALT flaps (flap thickness 8–10 mm), a subsequent thinning step is not necessary. For flaps with a thickness of 12–20 mm, we will need to assess the anatomical structure of the perforating vessel to select the appropriate thinning option. Based on our experience, only perforators that follow a perpendicular or near-perpendicular course through the fascia are considered suitable candidates for immediate thinning. Once the suitable perforator is confirmed, the thinning strategy is determined based on the number of identified perforators. To ensure maximum flap safety, the thinning process is performed while the flap remains perfused by its donor source, allowing continuous blood circulation and immediate identification of any vascular compromise.
For extensive flaps supported by a single perpendicular perforator, only primary thinning is performed using blunt-tipped scissors to remove the deep adipose tissue while preserving a wide fascial island (2–5 cm) around the perforator to maintain the fascial vascular plexus. This plexus provides vascular connections between the perforator and the superficial fascial and subdermal vascular systems, thereby ensuring reliable perfusion of the distal part of the flap.
When the flap incorporates two or more independent perpendicular perforators, microdissection thinning is performed under magnification. This more aggressive approach involves the systematic removal of individual fat lobules within the superficial adipose layer; the redundancy provided by multiple perforators acts as a “vascular safety net,” allowing the flap to be thinned to 3–4 mm thickness. The fascial island surrounding the perforator is preserved at least 1 cm without concern for compromised perfusion of the large flap (Fig. 1).
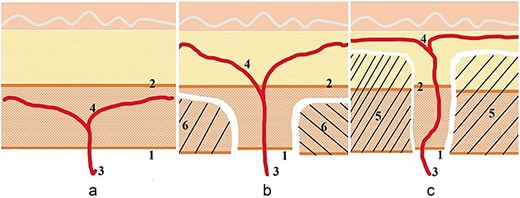
Illustration of the technique. (a) The perforator branching below the superficial fascia is not a candidate for thinning. (b) The perforator branches diagonally to both sides through the superficial fascia and can only be primarily thinned. The fascia island is left at 2–4 cm. (c) The perforator branches running perpendicular to the skin can be microdissected and thinned. The fascia island retains 1–2 cm.
Before pedicle division, flap viability was checked with three indicators: active bright red bleeding at the dermal border, capillary refill time under 2 s across the skin paddle, and maintaining a pink color. Only after confirming stable perfusion was the pedicle divided and transferred to the recipient site, where microvascular anastomosis was performed. The thinned ALT flap is shaped to match the contour, providing pliable coverage that mimics native skin and allows immediate gliding of underlying structures.
Cases presentation
Case 1
A 13-year-old boy developed a severe anterior elbow contracture after a degloving injury sustained in a traffic accident 5 months earlier. Previous debridement and split-thickness skin grafting were complicated by infection, resulting in a hypertrophic anterior scar with limited elbow motion (40° extension, 90° flexion) (Fig. 2A). Given the predominant anterior contracture, the anterior scar was completely excised, creating a 19 × 20-cm defect. Reconstruction was performed using a single-perforator ALT free flap harvested from the right thigh. The flap measured 10 × 18 cm, with an initial thickness of 18 mm (Fig. 2B). Primary thinning reduced the flap thickness to 7 mm while preserving a 5-cm fascial island around the perforator to ensure vascular reliability. Microvascular anastomoses were performed in an end-to-side fashion to the brachial artery and two concomitant veins. The donor site was resurfaced with a split-thickness skin graft. The flap survived without complications. At 4-month follow-up, elbow extension improved to 10°, with flexion maintained at 90°. The patient reported high functional and aesthetic satisfaction (Fig. 2C and D).
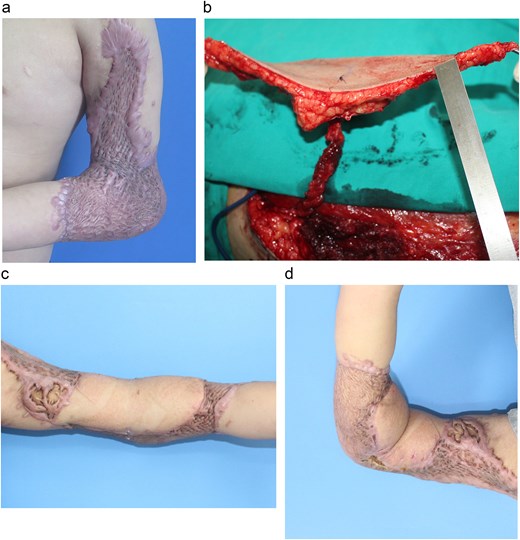
(a) Extensive post-burn contracture of the elbow with severe range-of-motion limitation. (b) Harvest of the ALT flap, showing the isolated vascular perforator. (c, d) Postoperative results show successful coverage and restoration of elbow function.
Case 2
A 67-year-old man developed severe necrosis of the anterior aspect of his left elbow several weeks after extravasation of Yttrium-90, a beta-emitting radionuclide used in nuclear medicine for the treatment of non-Hodgkin lymphoma. As is typical in radiation-induced injuries, no spontaneous healing was observed. The patient experienced severe pain and significant functional sequelae, particularly limitation of elbow mobility (Fig. 3A).
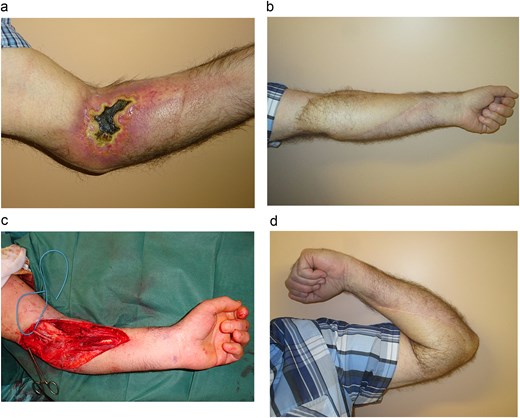
(a) The radial ulcer of the anterior elbow site. The patient experienced severe pain and significant functional sequelae, particularly limitation of elbow mobility. (b) The defect after resection was extensive. The elbow then received a 22 × 10 cm ALT flap, harvested without primary thinning, to cover the elbow defect. (c, d) Result after 2-year follow-up. The patient demonstrated excellent aesthetic and functional outcomes, with no recurrence of necrosis, a common complication of such radiation injuries.
Ten months after irradiation, the patient consented to surgical reconstruction. Extensive debridement was performed to release the contracture, followed by coverage of the large soft-tissue defect using an ALT flap measuring 22 × 10 cm (Fig. 2B). The flap was adequately thin and required no primary thinning. At the 2-year follow-up, the patient demonstrated excellent aesthetic and functional outcomes, with no recurrence of necrosis, a common complication of such radiation injuries (Fig. 3C and D).
Discussion
The reconstruction of complex elbow defects and post-burn contractures presents a unique challenge, requiring a balance between stable soft-tissue coverage and the preservation of joint mobility.
Elbow defect and reconstructive requirements
The elbow is a high-mobility joint with a thin subcutaneous layer and a wide range of motion. Pathological conditions, particularly post-burn scars, after tumor removal, or even the radiation ulcer, often lead to severe contractures that fix the joint in a dysfunctional position. Unlike other anatomical regions, the elbow requires a thinner coverage that can accommodate extreme skin stretching and folding without causing mechanical constriction. The primary goal of reconstruction is not merely wound closure but the restoration of functional gliding. An ideal tissue must be sufficiently thin to avoid mechanical bulk in the antecubital fossa and pliable enough to withstand constant stretch during flexion and extension. Furthermore, it must provide a durable surface to prevent recurrent adhesions between the skin and underlying tendons or nerves [1, 4, 7, 8].
Alternative reconstructive modalities
Various techniques have been utilized for elbow coverage, including skin grafts, local fasciocutaneous flaps, and regional pedicled flaps. Skin grafts are often prone to secondary contraction, leading to recurrent contractures. Local flaps, such as the antecubital or lateral arm flaps, are frequently limited by their size and the potential for donor-site morbidity in an already traumatized limb [3]. Regional pedicled flaps, while reliable, may not offer the necessary thinness or reach for extensive defects [1, 7, 9, 10]. The lateral arm flap offers similar tissue characteristics but is often limited by the dimensions of the skin paddle. The radial forearm flap provides a large surface area with high reliability; however, it requires sacrificing a major hand artery and leaves a conspicuous donor-site scar. Consequently, free tissue transfer has become the preferred choice for complex cases, with the ALT flap the most prominent “workhorse” due to its reliability and versatility [2, 11, 12].
Clinical rationale for perforator-based thinning in elbow reconstruction
The shift from conventional to thinned ALT flaps in elbow reconstruction is driven by the need to balance vascular reliability with the biomechanical requirements of the joint [12, 13]. Our clinical rationale for immediate thinning is grounded in the anatomical insight that flap survival depends less on total fat volume and more on the integrity of the perforator-to-subdermal plexus architecture. Strict selection of a perpendicular perforator orientation is essential to ensure vascular safety in our protocol. When a perforator courses perpendicular to the deep fascia, its branches extend deeper and then spread superficially within the subdermal layer. Consequently, thinning does not disrupt the subdermal vascular plexus, thereby preserving reliable perfusion of the entire flap. By identifying this orientation as a prerequisite, we minimize the risk of marginal necrosis, a common deterrent to immediate thinning. Furthermore, our strategy for managing large flaps with a single perforator addresses the "distal perfusion" dilemma. By maintaining a wide fascial island (2–5 cm), we are not merely removing fat but preserving a functional vascular plexus. This island protects the delicate lateral macrovessels that branch from the main perforator just above the fascia. In our experience, this is the only reliable way to ensure that the distal ends of an extensive flap (often exceeding 20 cm) remain well perfused. Conversely, the use of microdissection thinning in multi-perforator flaps leverages vascular redundancy. Having multiple inflow points creates a “safety net” that allows the surgeon to be more aggressive in thinning. This level of thinning, which brings the flap down to 3–5 mm, is transformative for elbow function. It ensures that the flap is pliable enough to glide over the olecranon and antecubital fossa during rapid movement. By eliminating the bulkiness that often restricts postoperative physical therapy, this approach directly translates to better long-term range-of-motion outcomes and a significant reduction in the psychological and financial burden of secondary debulking surgeries. In lean individuals or those with a low BMI, sufficiently thin ALT flaps can be used without primary thinning. To ensure flap viability after reconstruction, secondary thinning by liposuction or fat removal is often performed. In this case, the anatomical structure of the perforating vessel is not a concern.
Although the two cases presented here demonstrate excellent functional outcomes, this study is limited by its small sample size. However, these cases reflect the refined principles derived from our broader experience with over 150 thinned ALT flaps performed on the face, hand, and foot. In our larger series, we observed that adhering to a perforator-centric thinning algorithm significantly reduces the rate of partial necrosis. A larger, multicenter prospective study focused specifically on elbow contractures would help further validate the long-term functional advantages of this single-stage thinning technique compared with traditional multi-stage approaches.
Conclusion
The thinning ALT flap is a reliable and versatile option for reconstructing complex elbow defects. It provides thin, durable, and aesthetically acceptable coverage that conforms well to elbow anatomy and facilitates functional recovery.
Conflicts of interest
None declared.
Funding
None declared.



