-
PDF
- Split View
-
Views
-
Cite
Cite
Masahiko Narita, Nobuyuki Akasaka, Kentaro Shirakura, Hana Fukuta, Tomoki Nakatsu, Hiroyuki Kamiya, Thoracic aortic rupture in a patient with repeated oral fluoroquinolone administration, Journal of Surgical Case Reports, Volume 2026, Issue 4, April 2026, rjaf1115, https://doi.org/10.1093/jscr/rjaf1115
Close - Share Icon Share
Abstract
Fluoroquinolones (FQs) are widely used antibiotics with a broad antibacterial spectrum. However, recent studies have raised concerns regarding the potential association of FQs with an increased risk of aortic aneurysm and aortic dissection. We report a case of an older patient with stable blood pressure and no aortic dilation who developed a thoracic aortic rupture following repeated FQ administration. Although a direct causal relationship between FQ exposure and aortic rupture remains unproven in this single case, this case suggests the need for careful monitoring for possible aortic events, particularly in older patients requiring repeated FQ therapy.
Introduction
Fluoroquinolones (FQs) are commonly used antibiotics with a broad antibacterial spectrum and high oral bioavailability. Their mechanism of action includes inhibition of bacterial DNA synthesis; however, they may cause connective tissue collagen degeneration [1]. To date, several studies have indicated that FQ use is associated with the risk of aortic rupture and aortic dissection (AA/AD) [2–4]; therefore, the United States Food and Drug Administration (FDA) has warned against FQ use in patients at increased risk of AA/AD [5]. However, recent studies have reported that FQs are not risk factors for AA/AD [6–7]. The association between FQ exposure and AA/AD is still debated, and further evidence and clinical experience are warranted to clarify this problem. Herein, we report a case in which a patient with stable daily blood pressure and a non-dilated aorta experienced thoracic aortic rupture following repeated administration of FQs.
Case report
An 81-year-old male patient was transferred to our institution with chest radiographic abnormalities and an increased inflammatory response. The patient had a fever 2 weeks before admission, was diagnosed with a urinary tract infection (UTI), and was treated with oral levofloxacin (LVFX) by the patient’s doctor. The fever reduced following the administration of LVFX; however, follow-up chest radiography revealed an abnormal shadow in the left lung field (Fig. 1A). On admission, the physical examination results were normal: afebrile, no haemodynamic instability, and no specific symptoms. Contrast-enhanced computed tomography (CECT) revealed a thoracic aortic rupture (Fig. 1B and C). Therefore, the patient underwent emergency thoracic endovascular aortic repair (TEVAR) with a Zenith Alpha (32 × 155 mm; W. L. Gore & Associates, Flagstaff, AZ). The operation was successfully performed with no obvious complications (Fig. 2).
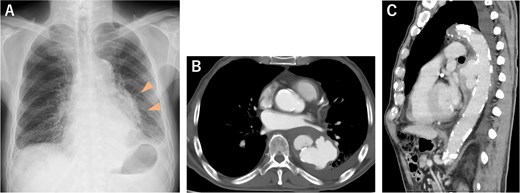
(A) Chest radiograph 3 days before admission showed an abnormal shadow in the left lung field (arrowheads). (B, C) CECT on admission revealed a descending thoracic aortic rupture.
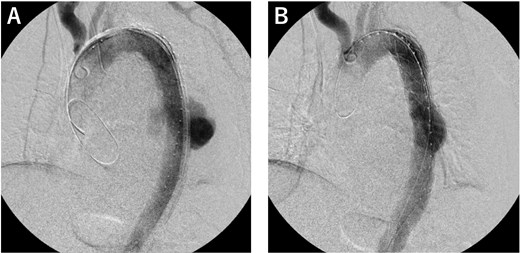
Operative findings. (A) We deployed a Zenith Alpha (32 × 155 mm; W. L. Gore & Associates, Flagstaff, AZ) in the descending aorta, covering the rupture site. (B) The final study confirmed loss of the contrast effect in the aneurysm and no obvious endoleak.
Postoperatively, we obtained additional information about the patient’s medical history. The patient had been bedridden for ~5 years due to severe dementia and required total assistance. A family member, who had a clinical nurse license, monitored medication use and daily blood pressure, which was stable; moreover, the patient’s antihypertensive drug had not been changed for a long time. Because the patient had a history of percutaneous coronary intervention at another hospital, the patient had undergone coronary computed tomography angiography follow-up. The images revealed that the maximum diameter of the descending aorta was 28 mm, and although arteriosclerotic changes were significant, the diameter had not changed for at least 3 years (Fig. 3A–C). The patient had long-term use of a urinary catheter for benign prostatic hypertrophy; therefore, the patient repeatedly developed UTIs and was treated with LVFX in almost all cases. In particular, 7 months before presentation, the patient received LVFX prescription four times, for 39 days in total (Fig. 4).
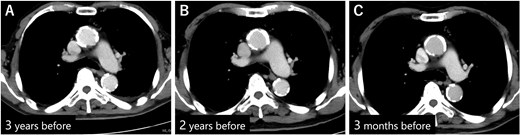
(A) CECT 3 years before presentation showed that the diameter of the descending aorta was 28 mm. (B) CECT 2 years before presentation. (C) CECT 3 months before presentation. Diameter of the descending aorta was unchanged during that period.
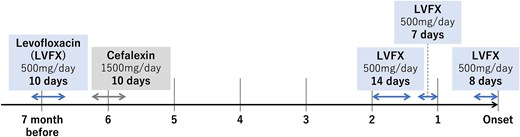
Antibiotic prescription history: The patient received levofloxacin for 39 days over 7 months, including 29 days within 2 months before presentation. LVFX, levofloxacin.
The patient had an uneventful postoperative course and was discharged on the 15th postoperative day.
Discussion
We encountered a patient who had been exposed to FQs and was treated for a thoracic aortic rupture with emergent TEVAR. This case is unique in that daily blood pressure control and medication adherence were confirmed, and the thoracic aortic diameter was <30 mm, with no dilation for at least 3 years before presentation. Briefly, in a patient with a relatively low risk for aortic events, a thoracic aortic rupture occurred during FQ administration.
Previous studies using large population databases [2, 3] and systematic reviews and meta-analyses [4] have revealed that the use of FQs is associated with an increased risk of AA/AD. Son et al. [3], in a study using a national health insurance database in Korea, reported that the risk of AA/AD was higher in patients presently receiving FQs, and the risk increased with the duration of exposure (>14 days) and increasing cumulative dose. In the present case, the patient experienced aortic rupture during FQ administration, and the duration of exposure was 29 days in the previous 2 months (Fig. 4). In this context, the patient could be considered at high risk for aortic events, thus requiring careful observation, particularly during current use of FQs and in the early period after exposure, despite stable blood pressure and no significant dilation of the thoracoabdominal aorta.
However, recent studies [6, 7] have indicated that there was no significant difference in the risk of AA/AD between patients administered FQs and those administered other antibiotics. They noted that previous studies did not distinguish the effect of infections treated with FQs from that of the FQs themselves and that systemic inflammation associated with infection may have a role in AA/AD onset [7]. Thus, they concluded that when clinically indicated, FQs use should not be avoided because the priority is to control infections with adequate antibiotics rather than vascular adverse events. FQs have certain advantages as first-line drugs for complicated UTIs; therefore, based on the patient’s pathology and background in this case, the use of FQs was reasonable.
Although the mechanism underlying FQ-induced AA/AD remains unknown, several hypotheses have been proposed, including FQs reducing collagen synthesis by potentiating the activity of matrix metalloproteinases [1, 2]. In experiments with Marfan mice, FQ exposure accelerated aortic root enlargement and increased the incidence of AA/AD [8]. No evidence of human congenital aortopathies has been reported, and previous epidemiological studies have not addressed differences in patient-specific backgrounds; however, there might be patient-related factors leading to susceptibility to AA/AD with exposure to FQs. For example, in the present case, the aortic wall with significant arteriosclerotic changes in addition to the advanced age may have made the patients susceptible to the effect of FQs.
In summary, the association between FQ exposure and AA/AD is still debated. The possibility of aortic events alone should not preclude the use of FQs when the appropriate indications are met. However, because AA/AD is potentially lethal, careful follow-up should be considered in patients with known or suspected aortic fragility, particularly in older patients who require repeated FQ therapy.
Acknowledgements
The authors would like to thank Dr. Kotaro Sato, Motowanishi Family Clinic, for sharing the patient’s clinical information.
Conflict of interest statement
The authors declare no conflicts of interest associated with this manuscript.
Funding
None declared.
Consent for publication
Written informed consent was obtained from the families of the patient for scientific activity including publication of this case report.
Ethics committee approval
Date: 22 April 2024. Number: 24–0826.



