-
PDF
- Split View
-
Views
-
Cite
Cite
Wolfgang J Schnedl, Simon Michaelis, Rudolf Schrittwieser, Dietmar Enko, Sandra J Holasek, Elevated fecal calprotectin values appear more common following esophagogastric than colon surgery, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag188, https://doi.org/10.1093/jscr/rjag188
Close - Share Icon Share
Abstract
Fecal calprotectin (FCAL) is commonly used to exclude inflammatory bowel disease in patients with symptoms of the irritable bowel syndrome spectrum. This retrospective case–control study evaluated FCAL levels in 81 postoperative patients to assess the impact of different gastrointestinal surgeries. Thirty-one patients had undergone esophago-gastric surgery (fundoplication or Roux-en-Y gastric bypass), and 50 had undergone colon surgery with hemicolectomy. Elevated FCAL levels (>50 μg/g) were observed in 56.7% of patients after esophago-gastric surgery compared with 24% of patients after hemicolectomy. Median FCAL levels were 96 μg/g and 99 μg/g, respectively. Statistically significant differences in FCAL levels were found between patients who underwent left-sided hemicolectomy and those who underwent Roux-en-Y bypass (P < .001), as well as right-sided hemicolectomy (P = .042). Overall, elevated FCAL levels were more than twice as common following esophago-gastric surgery, suggesting surgery-specific patterns of postoperative intestinal inflammation.
Introduction
Fecal calprotectin (FCAL) is released into the intestinal lumen from the gastrointestinal (GI) mucosa during intestinal inflammation. FCAL concentrations reflect the activity of inflammatory cells, primarily neutrophils, within the intestinal mucosa [1]. Current clinical guidelines recommend FCAL testing in patients presenting with symptoms within the irritable bowel syndrome (IBS) spectrum to exclude inflammatory bowel disease (IBD) [2]. In addition, FCAL is widely used as a non-invasive marker for quantifying intestinal inflammation. Its stability in stool samples, assay reproducibility, and low cost make it a valuable tool for guiding diagnostic and therapeutic decisions in clinical practice [3].
GI surgery is commonly performed and encompasses procedures involving the esophagus, stomach, and colon. Surgical treatment options for severe obesity include bariatric procedures such as Roux-en-Y gastric bypass (RYGB) and vertical sleeve gastrectomy [4]. In patients with severe gastroesophageal reflux disease (GERD) refractory to medical therapy, surgical interventions such as Nissen or Toupet fundoplication are established treatment options [5]. In colonic disease, right- and left-sided hemicolectomy are routinely performed, most commonly for oncologic indications [6].
During the implementation of an innovative laboratory method for FCAL determination, an unexpected observation was made: elevated FCAL levels appeared more frequently in patients with a history of esophageal or gastric surgery than in those who had undergone colonic surgery. Furthermore, previous studies have shown that FCAL elevations may persist for several years following RYGB surgery [7]. These postoperative FCAL elevations, which may reflect ongoing intestinal inflammation, require further investigation to determine whether they are associated with specific surgical procedures and whether they represent distinct patterns or intensities of intestinal inflammatory activity.
Case series
Study population
This retrospective case–control study used archived data collected between October and December 2023. Stool samples were obtained from patients attending a surgical outpatient clinic during routine follow-up visits as part of a collaboration aimed at validating a novel FCAL assay. In total, 81 consecutive white patients who had previously undergone GI surgery were included. Of these, 31 patients had undergone esophageal or gastric surgery, and 50 had undergone colonic surgery. All surgical procedures were reported as uncomplicated.
At the time of stool sampling, all patients were asymptomatic and reported no GI disturbances, including diarrhea. Among the 31 patients with upper GI surgery, 10 had undergone Nissen fundoplication and 4 Toupet fundoplication for severe GERD, while 17 had undergone RYGB for obesity. Among the 50 patients with colonic surgery for cancer, 25 had undergone right-sided and 25 left-sided hemicolectomy.
All patients were under active postoperative outpatient surveillance. Routine endoscopic evaluations revealed no abnormalities. Laboratory tests, including erythrocyte sedimentation rate, complete blood count, and liver and kidney parameters, were within normal limits. Patients were advised to avoid anti-inflammatory medications during stool sampling. Careful review of medical records revealed no evidence of systemic inflammation or bacterial infection. Patients with IBD, those who had undergone surgery within the preceding six months, or those receiving chemotherapy were excluded. The study did not evaluate the surgical procedures in terms of their open or minimally invasive nature with respect to GI surgery. Colonoscopy was performed in all patients aged ≥50 years and in those undergoing post-hemicolectomy surveillance, with normal findings in all cases.
Laboratory evaluations
FCAL concentrations were measured using a quantitative particle-enhanced turbidimetric immunoassay (Calprest Turbo; Eurospital, Trieste, Italy). The assay detection range was 35–1500 μg/g. FCAL levels <50 μg/g were considered normal, levels between 50 and 120 μg/g were classified as mildly to moderately elevated, and levels >120 μg/g were considered abnormal. Stool samples were collected in the morning and processed according to the manufacturer’s instructions.
Statistical analyses
Statistical analyses were performed using IBM SPSS statistics version 28.0 (IBM, Armonk, NY, USA), and GraphPad Pism version 10.3.1. (GraphPad Software, San Diego, CA, USA) was used for the generation of the figure. Data distribution was assessed using the Shapiro–Wilks test. Non-parametric tests (Mann–Whitney U and Kruskal–Wallis tests) were used for group comparisons. Gender distribution was analysed using Fisher’s exact test. Age and gender were controlled for using a generalized linear model. A P-value <.05 was considered statistically significant.
Results
Patient characteristics
Eighty-one postoperative patients (median age 65.1 years, range 25–82 years) were included. The esophago-gastric surgery group consisted of 31 patients (median age 50.6 years; 8 males, 23 females), while the colon surgery group included 50 patients (median age 67.6 years; 23 males, 27 females). The median time between surgery and FCAL testing was 5 years (range 0.5–21 years). The median time since surgery for esophago-gastric surgery was 6 years, with a range of 1–15 years, while for colonic surgery the median time since surgery was 4 years, with a range of 0.5–21 years.
FCAL outcomes
As illustrated in Table 1, the surgical procedure undergone and the outcomes of FCAL determinations are presented.
| Surgery . | Number of patients . | Number of patients with elevated FCAL >50 μg/g . | FCAL μg/g median (range) . | Number of patients with normal FCAL <50 μg/g . |
|---|---|---|---|---|
| Fundoplication | 14 | 6 (42.8%) | 94.5 (64–255) | 8 (57.1%) |
| RYGB | 17 | 11 (64.7%) | 96 (60–301) | 6 (35.3%) |
| Right-sided hemicolectomy | 25 | 4 (16%) | 109 (76–215) | 21 (84%) |
| Left-sided hemicolectomy | 25 | 8 (32%) | 74 (52–349) | 17 (76%) |
| Surgery | Number of patients | Number of patients with elevated FCAL >50 μg/g | FCAL μg/g median (range) | Number of patients with normal FCAL <50 μg/g |
|---|---|---|---|---|
| Fundoplication | 14 | 6 (42.8%) | 94.5 (64–255) | 8 (57.1%) |
| RYGB | 17 | 11 (64.7%) | 96 (60–301) | 6 (35.3%) |
| Right-sided hemicolectomy | 25 | 4 (16%) | 109 (76–215) | 21 (84%) |
| Left-sided hemicolectomy | 25 | 8 (32%) | 74 (52–349) | 17 (76%) |
Table 2 presents the surgical procedures and a summary of concomitant diseases, including medical therapies, for patients with FCAL results >50 μg/g.
The surgical procedures and summarized concomitant diseases including medical therapy in patients with FCAL results >50 μg/g.
| Surgery . | Fundoplication . | RYGB . | Right-sided hemicolectomy . | Left-sided hemicolectomy . |
|---|---|---|---|---|
| Number of patients with elevated FCAL >50 μg/g | 6 | 11 | 4 | 8 |
| Arterial hypertension | 3 with ACE- inhibitor | 0 | 2 with ACE- inhibitor | 5 with ACE- inhibitor, 1 with beta-blocker, 1 with calcium channel blocker |
| Arteriosclerosis | 3 with statin | 0 | 3 with statin | 6 with statin |
| Depression | 1 with mirtazapine and 2 with sertraline | 4 with escitalopram | 1 with escitalopram | 3 with escitalopram and 1 with sertraline |
| Hypercholesterinemia | 4 with statin | 0 | 3 with statin | 6 with statin |
| Osteoporosis | 3 with denosumab s. c. and 1 with bisphosphonate i. v. | 0 | 0 | 1 with bisphosphonate i. v. |
| Diabetes mellitus type 2 | 2 with metformin and 2 with SGLT-2i | 1 with life style modification | 2 with metformin | 2 with metformin and 1 with SGLT-2i |
| Reflux disease | 6 with PPI | 4 with PPI | 0 | 3 with PPI |
| Thyroid disease | 2 with levothyroxine | 4 with levothyroxine | 2 with levothyroxine | 1 with levothyroxine |
| Surgery | Fundoplication | RYGB | Right-sided hemicolectomy | Left-sided hemicolectomy |
|---|---|---|---|---|
| Number of patients with elevated FCAL >50 μg/g | 6 | 11 | 4 | 8 |
| Arterial hypertension | 3 with ACE- inhibitor | 0 | 2 with ACE- inhibitor | 5 with ACE- inhibitor, 1 with beta-blocker, 1 with calcium channel blocker |
| Arteriosclerosis | 3 with statin | 0 | 3 with statin | 6 with statin |
| Depression | 1 with mirtazapine and 2 with sertraline | 4 with escitalopram | 1 with escitalopram | 3 with escitalopram and 1 with sertraline |
| Hypercholesterinemia | 4 with statin | 0 | 3 with statin | 6 with statin |
| Osteoporosis | 3 with denosumab s. c. and 1 with bisphosphonate i. v. | 0 | 0 | 1 with bisphosphonate i. v. |
| Diabetes mellitus type 2 | 2 with metformin and 2 with SGLT-2i | 1 with life style modification | 2 with metformin | 2 with metformin and 1 with SGLT-2i |
| Reflux disease | 6 with PPI | 4 with PPI | 0 | 3 with PPI |
| Thyroid disease | 2 with levothyroxine | 4 with levothyroxine | 2 with levothyroxine | 1 with levothyroxine |
Abbreviations: ACE, angiotensin converting enzyme; FCAL, fecal calprotectin; i. v., intravenous; PPI, proton pump inhibitor; s. c., subcutaneous; SGLT-2i, sodium-glucose cotransporter-2 inhibitor.
Statistical findings
Performing the Mann–Whitney U test, patients with esophago-gastric surgery exhibited significantly higher FCAL values than those with colonic surgery (median 61 μg/g vs. 34 μg/g; P = .014). Significant differences were also observed between specific surgical subgroups: patients with left-sided hemicolectomy had significantly lower FCAL levels (median 20 μg/g, range 15–35) compared with patients who underwent RYGB (median 87 μg/g, range 15–301; P < .001) and right-sided hemicolectomy (median 41 μg/g, range 15–301; P = .042) (Fig. 1).
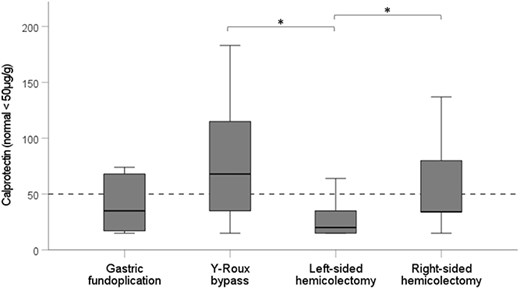
The FCAL tests include data from 31 patients who underwent esophageal and stomach surgical procedures as well as 50 patients who suffered colorectal surgery. The normal range for FCAL levels is indicated as less than 50 μg/g. Furthermore, the figure demonstrates statistically significant differences using FCAL values between patients who had left-sided hemicolectomy compared to those who underwent RYGB (P < .001) and, to those who had undergone right-sided hemicolectomy (P = .042).
Patients undergoing esophago-gastric surgery were significantly younger than those with colon surgery (median 51 vs. 67 years; P < .001). Gender distribution did not differ significantly between groups. In a generalized linear model, age and gender had no significant influence on FCAL levels (P = .501 and P = .788, respectively). After adjustment, the difference in FCAL values between surgical groups narrowly missed statistical significance (P = .065). Postoperative time intervals (P = .9263) and gender (P = .3776) were not associated with FCAL elevation.
Discussion
Chronic inflammatory diseases such as cardiovascular disease, cancer, obesity, diabetes mellitus, and chronic respiratory disease are major contributors to global mortality [8]. Neutrophil granulocytes play a central role in inflammatory processes associated with these conditions. Human neutrophils have been shown to produce a wide range of proteins and cytokines [9]. Calprotectin, a calcium-binding protein comprising approximately 60% of the cytosolic protein content of neutrophils, has antimicrobial and immune-modulatory properties [3].
FCAL is an established biomarker for intestinal inflammation and is widely used in the diagnosis and monitoring of IBD, including Crohn’s disease and ulcerative colitis as well as for distinguishing organic disease from functional disorders within the IBS spectrum [10]. During intestinal inflammation, FCAL from neutrophils is released into the intestinal lumen [11]. However, FCAL levels can be influenced by several factors, including sample handling, intra-individual variability, and interactions with the gut microbiota [12]. The ingestion of foods may interact with the microbiota, which could in turn affect the FCAL values [13]. More recently, elevated FCAL levels have been linked to adverse clinical outcomes beyond GI disease. Elevated FCAL levels have been associated with clinical outcomes in older individuals [14] and have been identified as a prognostic marker for mortality and renal outcomes in chronic kidney disease [15] and adverse outcomes and mortality in advanced chronic liver disease [16]. The observed postoperative elevation of FCAL warrants further investigation, as it may reflect underlying intestinal inflammation.
The intestinal mucosa constitutes a pivotal barrier between the host and the luminal contents. Disruption of this barrier may facilitate the translocation of bacterial products and contribute to chronic systemic inflammation [17]. This intestinal barrier, which plays a crucial role in maintaining overall health, is subject to disruption in the presence of intestinal inflammation, as evidenced by elevated FCAL values. The present study demonstrates that postoperative FCAL elevations are more common following esophago-gastric surgery than colonic surgery, even several years after the procedure. This finding raises the possibility of persistent low-grade intestinal inflammation following upper GI surgery. Owing to the small patient cohort and the potential influence of concomitant diseases or medical therapies, these variables could not be established as contributing factors in patients with FCAL concentrations exceeding 50 μg/g.
Surgical interventions may alter GI anatomy, physiology, and microbial composition. Growing evidence indicates that surgical procedures from the esophagus to the rectum can influence the gut microbiota [17]. However, technological advancements in GI surgical techniques are consistently being pursued [18]. Bariatric surgery, particularly RYGB, is known to induce profound and sustained changes in GI function and microbiota, contributing to long-term weight loss and improvement of metabolic comorbidities [19]. Despite its beneficial metabolic effects, RYGB has been associated with persistent FCAL elevation, suggesting ongoing intestinal immune activation [7]. In the present study, nearly two-thirds of patients who had undergone bariatric surgery exhibited elevated FCAL levels years after surgery.
GERD-related inflammation may also be influenced by microbial shifts and procedures of fundoplication with bariatric surgery affects esophago-gastric physiology [20]. FCAL has been proposed as a predictive marker for esophageal injury [21], and elevated FCAL levels have been reported in children following surgical repair of esophageal atresia [22]. These findings are consistent with the elevated FCAL levels observed in over half of patients with esophago-gastric surgery in this study.
Hemicolectomy is most commonly performed for colon cancer, though diverticular disease also contributes to left-sided resections [6]. Elevated FCAL levels have been reported prior to colorectal cancer diagnosis [23]. In this study, 24% of patients exhibited elevated FCAL levels after hemicolectomy, although left-sided hemicolectomy was associated with particularly low FCAL values. The reasons for this observation remain unclear and warrant further investigation.
Age-related differences in FCAL have been reported, with higher levels observed in children and younger individuals [24]. Although patients with esophago-gastric surgery were younger in this cohort, age did not independently influence FCAL levels in adjusted analyses. Limitations of this study include its retrospective design, small sample size, ethnically homogeneous population, and lack of stratification by surgical technique.
Overall, this pilot study highlights the need for further clinical and experimental research to clarify the mechanisms underlying postoperative FCAL elevation, its relationship to intestinal inflammation, and its potential long-term systemic consequences [25].
Conclusions
This retrospective analysis indicates that postoperative FCAL elevations are more than twice as common following esophago-gastric surgery compared with colonic surgery. The particularly low FCAL levels observed after left-sided hemicolectomy suggest differing patterns of intestinal inflammatory activity depending on surgical intervention. Persistent FCAL elevation following upper GI surgery underscores the need for targeted follow-up studies to elucidate underlying mechanisms and assess potential clinical implications.
Author contributions
Wolfgang J. Schnedl: Corresponding author, Study design, Data collection, Manuscript writing and Final approval. Simon Michaelis: Participation at data collection, Manuscript writing and Manuscript final approval. Rudolf Schrittwieser: Data collection and interpretation, Manuscript writing and Final approval. Dietmar Enko: Data collection and analysis, Manuscript final approval. Sandra J. Holasek: Data analysis and interpretation, Manuscript final approval. All authors read and approve the final version of the manuscript.
Conflicts of interest
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Funding
None declared.
Data availability
The data that support the findings are available from the corresponding author upon request.
Ethical approval and/or institutional review board approval
The study is of an observational and retrospective nature, and is based on existing data. The study adheres to the ethical guidelines outlined in the Declaration of Helsinki.



