-
PDF
- Split View
-
Views
-
Cite
Cite
Winnie Long, Abid Qureshi, Peter Einersen, Infusion of tPA-dornase for management of intraabdominal abscesses: a case report, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag177, https://doi.org/10.1093/jscr/rjag177
Close - Share Icon Share
Abstract
Postoperative intra-abdominal abscesses are a common postsurgical complication and can lead to prolonged hospitalization and severe sepsis. The management of these abscesses is typically conservative, with intravenous antibiotics and percutaneous drainage. In those who fail conservative management, the next step is typically surgical intervention with washout. However, this may be contraindicated in a patient with multiple co-morbidities, who may not tolerate general anesthesia. Fibrinolysis via tissue plasminogen activator (tPA) and dornase infusion into the pleural cavity is well established for treatment of complicated empyemas. In comparison, infusion into the abdominal cavity has only been reported in one case series. In this case report, we describe our experience successfully treating an intraabdominal abscess with administration of tPA-dornase into the intra-abdominal cavity.
Introduction
Postoperative intraabdominal abscesses are common complications of severe abdominal infection, at rates of 1%–10% and may present with persistent leukocytosis, malaise, and postoperative ileus [1]. Treatment of these abscesses is typically guided by the size of the abscess and the window for percutaneous drainage. Patients with abscesses >3 cm with an adequate window are candidates for percutaneous drainage, whereas patients with smaller abscesses are managed with antibiotics alone. Resolution is evaluated by the quantity of drainage, resolution of clinical signs of sepsis, and imaging. For inadequately drained abscesses, patients may undergo drain upsizing, or in severe cases, they may require going to the operating room for abdominal washout. A revisit to the operating room, however, puts patients with severe comorbidities at high risk for complications.
As described in the MIST2 trial, the combination of tissue plasminogen activator (tPA) and dornase has been widely utilized in management of empyemas [2]. tPA works by degrading the fibrin strands that comprise the septations in complicated empyemas, allowing the fluid collection to drain [3]. Dornase supplements the action of tPA by breaking down extracellular DNA and thinning the viscosity of the fluid [4]. By this mechanism, a septated intraabdominal abscess hypothetically could be safely drained in a similar manner.
Prior randomized control trials have evaluated the use of tPA alone in the abdominal cavity without clear benefit. Recently however, there has been newly published data on the potential application of tPA and dornase in the intraabdominal cavity. In this case report, we report our experience with instilling tPA and dornase to treat an intraabdominal abscess refractory to drainage in a surgically complex patient.
Clinical presentation
A 41 year old female presented to the emergency department with clinical presentation of peritonitis 4 days after a robot-assisted laparoscopic cholecystectomy, hiatal hernia repair, and gastric sleeve converted to Roux-en-Y gastric bypass. The patient underwent a diagnostic laparoscopy and was found to have a necrotic Roux limb in the setting of venous congestion. The Roux limb along with the necrotic congested mesentery was resected, and the Roux-en-Y gastric bypass was recreated. Postoperatively, the patient developed fevers and tachycardia despite receiving broad-spectrum antibiotics. Labwork showed leukocytosis to 16.1 with left shift to 81.5%. Computed tomography (CT) scan showed a saddle pulmonary embolism and pelvic fluid collection measuring 14.2 × 7.5 cm (Figs 1 and 2). The patient was started on therapeutic anticoagulation, and interventional radiology (IR) was consulted. No thrombectomy was indicated as there were no signs of right heart strain, however IR placed a 8.5 French drainage catheter in the abscess cavity. Wound cultures grew Enterococcus faecalis Group D and Proteus mirabilis.
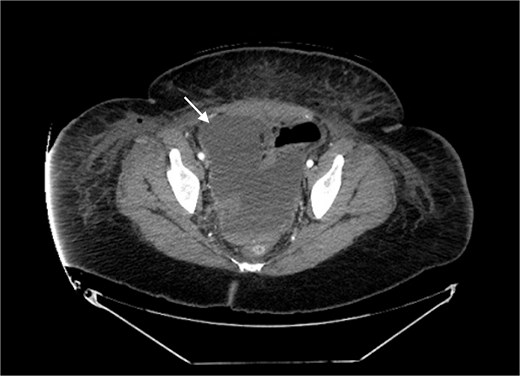
Pelvic fluid collection prior to IR drainage measuring 15.4 × 10.6 cm, in the axial view.
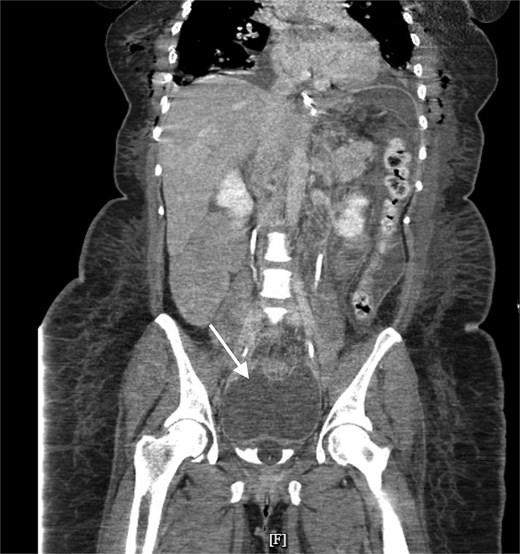
Pelvic fluid collection prior to IR drainage measuring 15.4 × 10.6 cm, in the coronal view.
Over the next week, the catheter drainage volume gradually decreased and became minimal. Interval imaging was obtained and revealed minimal decrease in the size of the pelvic fluid collection to 10.7 × 7.7 cm. The 8.5 French drain was then upsized to 12-French (Fig. 3), with withdrawal of only 3 cc seropurulent output upon exchange. The next day, the drain output remained minimal at 15 cc, and the patient remained persistently tachycardic in the 130 s. At this point, the patient’s hospital course was not only complicated by pulmonary embolism, but also bilateral pleural effusions that had been increasing in size and rhabdomyolysis secondary to daptomycin. Due to the acute risk of acute kidney injury (AKI) from rhabdomyolysis, daptomycin was discontinued, and the patient’s antibiotic regimen was adjusted to ceftriaxone and flagyl.
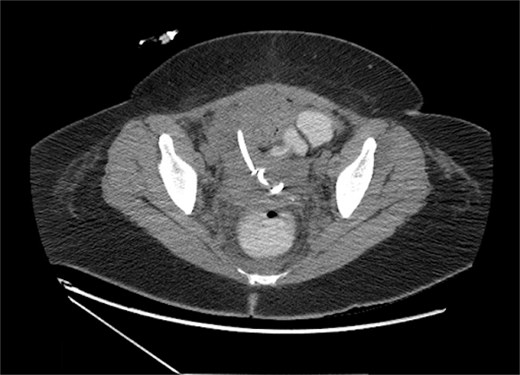
Pelvic fluid collection on upsizing IR drain from 8.5 to 12 Fr, measuring 10.7 × 7.7 cm prior to tPA-dornase instillation, in the axial view.
We discussed in detail returning to the operating room compared to conservative management with tPA-dornase, which we regularly used with the thoracic surgery service. In light of the risk of worsening rhabdomyolysis and pleural effusions, the risk of triggering severe AKI and worsening respiratory status made further exposure to general anesthesia suboptimal. After thorough discussion, the patient declined further surgery, preferring medical management.
To ameliorate the bleeding risks of tPA-dornase, anticoagulation was decreased from therapeutic to prophylactic lovenox dosing. The patient received an infusion of 10 mg alteplase in 30 ml normal saline followed by 5 mg dornase in 30 ml normal saline via the IR drain. The drain was clamped for 1 h, and the patient was encouraged to ambulate while the drain was clamped. An hour afterward, the drain was unclamped, and the drainage was monitored for character and volume. Hemoglobin was trended to ensure laterality. The patient underwent a total of five tPA-dornase infusions over 5 days and tolerated the infusions well without signs of bleeding (Table 1). A CT scan was performed after the final infusion, showing resolution of the pelvic fluid collection (Fig. 4). Antibiotics were discontinued, and the patient was discharged after 10 weeks inpatient. At follow-up, the patient had no recurrent signs and symptoms of recurrent abscess.
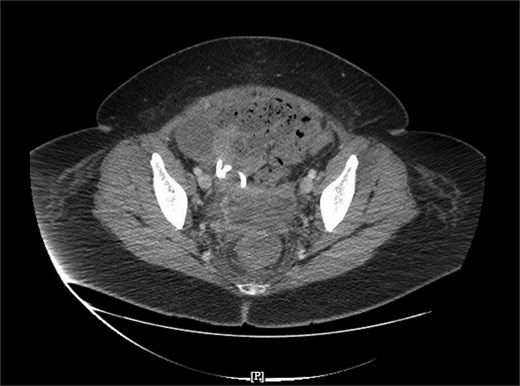
Resolution of pelvic fluid collection with 12 Fr catheter in place, in the axial view.
Table of drain outputs in context of tPA-dornase infusion after drain upsizing.
| Day . | Event . | Drain output (cc) . |
|---|---|---|
| 1 | IR drain upsized (Fig. 3) | 15 (seropurulent) |
| 2 | Infusion 1 | 131 (serosanguinous) |
| 3 | Infusion 2 | 125 (serosanguinous) |
| 4 | Infusion 3 | 160 (serosanguinous) |
| 5 | Infusion 4 | 45 (serosanguinous) |
| 6 | Infusion 5 | 110 (serosanguinous) |
| 7 | 110 (serosanguinous) | |
| 8 | CT demonstrating resolution (Fig. 4) | 15 (serosanguinous) |
| 9 | 0 (serosanguinous) | |
| 10 | IR drain removed | 6.5 (serosanguinous) |
| Day | Event | Drain output (cc) |
|---|---|---|
| 1 | IR drain upsized ( | 15 (seropurulent) |
| 2 | Infusion 1 | 131 (serosanguinous) |
| 3 | Infusion 2 | 125 (serosanguinous) |
| 4 | Infusion 3 | 160 (serosanguinous) |
| 5 | Infusion 4 | 45 (serosanguinous) |
| 6 | Infusion 5 | 110 (serosanguinous) |
| 7 | 110 (serosanguinous) | |
| 8 | CT demonstrating resolution ( | 15 (serosanguinous) |
| 9 | 0 (serosanguinous) | |
| 10 | IR drain removed | 6.5 (serosanguinous) |
Discussion
Management of an abscess that fails percutaneous drainage typically involves an abdominal washout in the operating room. However, due to the risks of further operative procedures in this complex patient, tPA-dornase was used as an alternative.
In the retrospective case series by Schultz et al., alteplase and dornase were instilled in 13 patients with intra-abdominal abscesses at 10 and 5 mg, respectively, with varying volumes [4]. We elected to formulate the medications in 30 ml volumes, in accordance with the MIST2 trial [2]. Two of the patients in the Schultz et al. series were kept on therapeutic anticoagulation during their infusions of tPA-dornase, with one patient experiencing life-threatening bleeding [4]. We elected to ameliorate this risk by reducing the therapeutic lovenox to prophylactic levels in our patient.
In the Schultz series, eight patients were discharged without drain displacement or re-operation, and successful outcomes were established by increased abdominal drain output or reduction in size of fluid collection [4]. Our patient had a final CT demonstrating resolution of the fluid collection.
Several other studies have suggested therapeutic benefits of tPA infusion into the intra-abdominal cavity [3]. However, prospective randomized control trials have not been able to demonstrate a clear advantage to tPA infusion compared to percutaneous drainage alone [5–7]. One randomized control trial of 47 patients demonstrated increased drainage in those who received tPA instillation, but ultimately both the treatment and control group had resolution of the abscesses [5]. No significant complications were noted in these trials.
Conclusion
There currently is only one case series of 13 patients reporting usage of both tPA-dornase for abdominal fluid collections refractory to surgical drainage. Our case represents the 14th reported case. In our experience, tPA-dornase is a potential treatment option for surgically complex patients, in whom a re-operation for washout may be too high risk. A greater sample size and ultimately a randomized-controlled trial will be required to further assess the safety and efficacy of intra-abdominal alteplase-dornase therapy.
Conflicts of interest
None declared.
Funding
None declared.
References
Taha M, Abouelsadat MK, Elfakharany M et al.



