-
PDF
- Split View
-
Views
-
Cite
Cite
Siu Y Kok, Faheez Mohamed, Brendan J Moran, Sanjeev Dayal, Alexios Tzivanakis, Richard Hindley, Amr Emara, Thomas Cecil, Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy can be safely combined with nephrectomy in patients with appendiceal malignancy and synchronous renal cell carcinoma, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag170, https://doi.org/10.1093/jscr/rjag170
Close - Share Icon Share
Abstract
The combination of an appendix tumour requiring cytoreductive surgery (CRS) and hyperthermic intra-peritoneal chemotherapy (HIPEC) with the management of a renal cell cancer is rare but problematic. Acute kidney injury (AKI) is one of the major complications after CRS and HIPEC with concern that combining CRS and HIPEC with nephrectomy may further increase the risk of AKI. There is little published on this combined approach of CRS and HIPEC with nephrectomy for patients with disseminated appendiceal tumours. We report on eight patients who had CRS and HIPEC for an appendiceal tumour with synchronous nephrectomy. We performed a retrospective review of patients undergoing CRS and HIPEC for appendiceal malignancy and synchronous nephrectomy for renal cell carcinoma between 2014 and 2025. Patient demographics, operative details, pathology, postoperative morbidity and mortality were evaluated. A total of eight patients (M: F 4:4) with a median age of 65 were included, of which 5/8 (62.5%) presented with abdominal pain. Concerning the primary appendiceal pathology, CRS included right hemicolectomy or extended right hemicolectomy in 5/8 (67%). Regarding the renal pathology, 7/8 (87.5%) had either right or left nephrectomy and 1 (12.5%) had right partial nephrectomy. The mean operation time was 453.75 minutes (365–588). The mean blood loss was 1053 ml (420–2000). Mean Peritoneal Cancer Index was 13 (0–39). Complete cytoreduction was achieved in 5/8 (62.5%). Appendix tumour pathology was low grade appendiceal mucinous neoplasm in 5/8, goblet cell adenocarcinoma in 2/8 and high grade appendiceal mucinous neoplasm in 1. Pathology of the renal tumour was clear cell carcinoma in 4/8, papillary renal cell carcinoma in 3 and 1 patient with transitional cell carcinoma. Postoperative intensive care unit stay was a mean of 1 day (1–3). Median postoperative stay was 15 days (8–47). The mean drop in glomerular filtration rate was 10 ml/min/1.73 m2.There was no postoperative acute renal failure nor Clavien-Dindo Grade III-V complication with no post-operative mortality. Patients with synchronous appendiceal tumours and renal cell carcinoma have good immediate outcomes after combined CRS and HIPEC with nephrectomy with no measurable increase in risk of acute renal failure in this series.
Introduction
Appendiceal tumours with peritoneal dissemination are uncommon and are often found incidentally at cross-sectional imaging or at laparoscopy or laparotomy [1]. Optimal treatment of disseminated appendiceal tumours involves major surgical intervention, entitled cytoreductive surgery (CRS), combined with hyperthermic intra-peritoneal chemotherapy (HIPEC).
Similarly, many patients with renal cell carcinoma are initially asymptomatic and are detected at cross-sectional imaging. The combination of an appendix tumour requiring CRS and HIPEC with thevv management of a renal cell cancer is rare but problematic.
Acute kidney injury (AKI) is one of the major complications after CRS and HIPEC causing prolonged hospital stay, increase in mortality, and delay in adjuvant chemotherapy [2]. There are a number of contributing factors relating to HIPEC including the nephrotoxic nature of the intraperitoneal chemotherapy agent or hyperthermia-related changes in renal perfusion [3–7]. Nephrectomy is a major surgical procedure that can cause AKI and other morbidity. There is concern that combining CRS and HIPEC with nephrectomy may further increase the risk of AKI and postoperative morbidity and mortality. There is little published on this combined approach, especially in patients requiring extensive CRS and HIPEC for an appendiceal tumour and synchronous nephrectomy. Previous publications have reported on patients having CRS and HIPEC requiring general urological procedures but little on CRS and HIPEC with nephrectomy [8, 9]. One paper reported an experience of CRS and HIPEC with nephrectomy for various tumour pathologies [10]. Therefore, the aim of this study was to report on the experience of CRS and HIPEC and synchronous nephrectomy in patients with both an appendiceal tumour and a renal tumour. The main focus is on safety of these combinations, particularly post-operative complications related to AKI.
Method
Study design and patient data source
This was a retrospective review of a prospectively collected database of patients undergoing CRS and HIPEC (CRS + HIPEC) for appendiceal malignancy and synchronous nephrectomy for renal cell carcinoma between January 2014 and February 2025 in a high volume Institute. Demographic data, oncology details, operative findings and procedures, pathology, postoperative morbidity and mortality were evaluated.
Preoperative assessment
Patients referred with peritoneal malignancy were evaluated in a speciality multidisciplinary team (MDT) meeting with scans reviewed by specialist peritoneal malignancy radiologists (Figs 1 and 2) and experienced peritoneal malignancy surgeons. The standard imaging was dual contrast computed tomography (CT) of the chest, abdomen and pelvis. Tumour marker assessments include CEA, CA19.9, and CA 125. Patients with a renal mass were discussed in a renal cancer MDT.
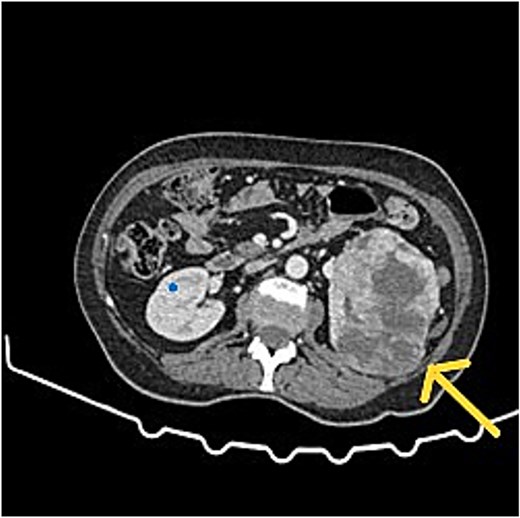
CT scan showed left RCC, appendix tumour with surrounding mucin.
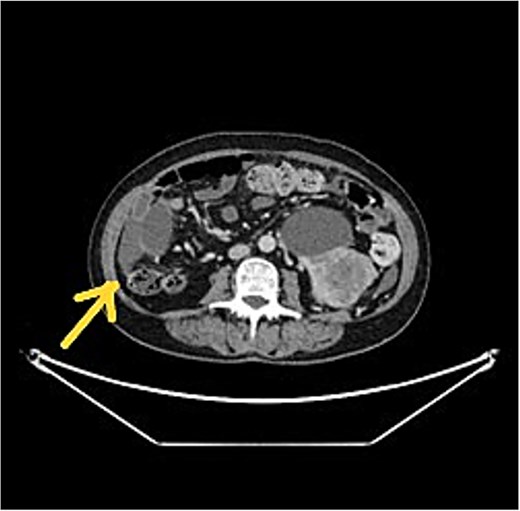
CT scan showed left RCC, appendix tumour with surrounding mucin.
Renal function monitoring
Renal function was monitored by sequential serology, urinary output, and fluid balance.
Surgical procedures
The surgical approach was CRS by a full midline laparotomy by the peritoneal malignancy team with urology specialists performing the urological intervention. Intraoperative peritoneal cancer index (PCI) [11] and completeness of cytoreductive score (CC score) were assessed [12]. After completion of surgical resection, HIPEC in the form of mitomycin C (MMC), 10 mg/metre squared was perfused over 60 minutes at 41°C. Organs removed, peritonectomy procedures, operation time, intraoperative blood loss and blood products and colloid transfusion were recorded.
Postoperative care and follow up
All patients were admitted to an Intensive Care Unit (ITU) postoperatively. Patients were transferred to a dedicated surgical ward when vital signs were stable. Length of ITU and total hospital stay were recorded.
Morbidity and mortality
Postoperative 30-day morbidity and mortality were recorded. Postoperative complications were classified based on the Clavien-Dindo classification system [13].
Statistics
SPSS version 26.0 was used for statistical analysis.
Results
Patient demographics, clinical and pathological characteristics
Overall eight patients (M: F 4:4) with a median age of 65 were included. In total 5/8 (62.5%) presented with abdominal pain and the other three patients presented with abdominal distension, abdominal wall mass and right flank swelling. In 6/8 (62.5%) the renal tumour was detected incidentally at the staging CT for the appendiceal tumour. In one patient, pseudomyxoma peritonei was found at planned nephrectomy and the nephrectomy was abandoned for further work up. One patient had a renal tumour detected incidentally on histology after nephrectomy was performed as a result of extensive ureteric involvement from recurrent pseudomyxoma peritonei (Table 1).
| M:F | 4:4 | |
| Age (Median) | 65 | |
| Performance (ECOG) | Relevant disease | 4 (50%) |
| Restrictive disease | 3 (37.5%) | |
| Fit | 1 (12.5%) | |
| Preoperative diagnosis (Renal tumour) | Incidental findings in CT | 6 (62.5%) |
| Intraoperative findings of pseudomyxoma peritonei intraoperation for nephrectomy | 1 (12.5%) | |
| Incidental at histology of resected kidney | 1 (12.5%) | |
| Preoperative renal function | Cr preop (mean) | 78 (55–103) |
| eGFR preop (mean) | 79 (62–90) |
| M:F | 4:4 | |
| Age (Median) | 65 | |
| Performance (ECOG) | Relevant disease | 4 (50%) |
| Restrictive disease | 3 (37.5%) | |
| Fit | 1 (12.5%) | |
| Preoperative diagnosis (Renal tumour) | Incidental findings in CT | 6 (62.5%) |
| Intraoperative findings of pseudomyxoma peritonei intraoperation for nephrectomy | 1 (12.5%) | |
| Incidental at histology of resected kidney | 1 (12.5%) | |
| Preoperative renal function | Cr preop (mean) | 78 (55–103) |
| eGFR preop (mean) | 79 (62–90) |
Operative details
Concerning the primary appendiceal pathology, 5/8 (67.5%) had right hemicolectomy or extended right hemicolectomy, 1 (12.5%) had subtotal colectomy and 1 (12.5%) had appendicectomy and 1 (12.5%) had no bowel resection. The mean peritoneal cancer Index was 13 (0–39). With regards to renal pathology, 4/8 (50%) had left nephrectomy, 3 (37.5%) had right nephrectomy and 1 (12.5%) had right partial nephrectomy. The mean total operation time was 453.75 minutes (range 365–588). The mean blood loss was 1053 ml (420–2000). Complete cytoreduction was achieved in 5/8 (62.5%). The mean red blood cell and cryoprecipitate transfusion was 1 unit. The mean colloid transfusion volume was 1700 ml. There was no reduction in MMC dosage or HIPEC time (Table 2).
| Approach | Open | 8 (100%) |
| Operation: Appendix tumour | CRS + HIPEC | 8 (100%) |
| Operation time (Mean) | 454 mins (365–588) | |
| Blood loss (Mean) | 1053 ml (420–2000) | |
| Completeness of Cytoreduction (CC0–3) | 0 | 5 (62.5%) |
| 1 | 2 (25%) | |
| 2 | 0 | |
| 3 | 1 (12.5%) | |
| PCI (Mean) | 13(0–39) | |
| Appendix | Low grade appendiceal mucinous neoplasm | 5 (62.5%) |
| GCA | Tang A: 1 (12.5%) Tang B: 1 (12.5%) | |
| High grade appendiceal mucinous neoplasm | 1 (12.5%) | |
| RCC | Clear cell carcinoma | 4 (50%) |
| Papillary renal cell carcinoma | 3 (37.5%) | |
| Transitional cell carcinoma | 1 (12.5%) | |
| Resection margin | Clear | 8 (100%) |
| Clavien Dindo Classification | 1 | 7 (87.5%) |
| 2 | 1 (12.5%) | |
| Postoperative complication | No complication | 5 (62.5%) |
| Ileus | 1 (12.5%) | |
| Wound infection | 1 (12.5%) | |
| High stoma output | 1 (12.5%) | |
| Acute renal failure | 0 | |
| Reoperation | 0 | |
| Readmission | 0 | |
| 30-day mortality | 0 | |
| Postoperative ITU stay (mean) | 1 day (1–3) | |
| Postoperative total length of stay (median) | 15 (8–47) | |
| Postoperative renal function | Cr postop D1 (mean) | 101 (58–145) |
| Cr before discharge (mean) | 97 (67–162) | |
| eGFR postop D1 (mean) | 72 (44–90) | |
| eGFR before discharge (mean) | 69 (48–90) |
| Approach | Open | 8 (100%) |
| Operation: Appendix tumour | CRS + HIPEC | 8 (100%) |
| Operation time (Mean) | 454 mins (365–588) | |
| Blood loss (Mean) | 1053 ml (420–2000) | |
| Completeness of Cytoreduction (CC0–3) | 0 | 5 (62.5%) |
| 1 | 2 (25%) | |
| 2 | 0 | |
| 3 | 1 (12.5%) | |
| PCI (Mean) | 13(0–39) | |
| Appendix | Low grade appendiceal mucinous neoplasm | 5 (62.5%) |
| GCA | Tang A: 1 (12.5%) | |
| High grade appendiceal mucinous neoplasm | 1 (12.5%) | |
| RCC | Clear cell carcinoma | 4 (50%) |
| Papillary renal cell carcinoma | 3 (37.5%) | |
| Transitional cell carcinoma | 1 (12.5%) | |
| Resection margin | Clear | 8 (100%) |
| Clavien Dindo Classification | 1 | 7 (87.5%) |
| 2 | 1 (12.5%) | |
| Postoperative complication | No complication | 5 (62.5%) |
| Ileus | 1 (12.5%) | |
| Wound infection | 1 (12.5%) | |
| High stoma output | 1 (12.5%) | |
| Acute renal failure | 0 | |
| Reoperation | 0 | |
| Readmission | 0 | |
| 30-day mortality | 0 | |
| Postoperative ITU stay (mean) | 1 day (1–3) | |
| Postoperative total length of stay (median) | 15 (8–47) | |
| Postoperative renal function | Cr postop D1 (mean) | 101 (58–145) |
| Cr before discharge (mean) | 97 (67–162) | |
| eGFR postop D1 (mean) | 72 (44–90) | |
| eGFR before discharge (mean) | 69 (48–90) |
Cr = Creatinine (μmol/L)
eGFR = Glomerular filtration rate (ml/min/1.73 m2)
Pathology
The appendix tumour pathology was low grade appendiceal mucinous neoplasm in 5/8 (62.5%), goblet cell adenocarcinoma in two with one patient having a high grade appendiceal mucinous neoplasm. Renal pathology was clear cell carcinoma in 4/8(50%), papillary renal cell carcinoma in 3, and transitional cell carcinoma in 1 (Table 2).
Postoperative morbidity and mortality
Postoperative intensive care unit stay was a mean of 1 day [1–3]. Median postoperative stay was 15 days (8–47). There was no postoperative acute renal failure, urinary tract infection or Clavien-Dindo Grade III-V complication with no post-operative mortality. None of the patients required reoperation or readmission (Table 2). The patient who stayed 47 days was for management of a high output ileostomy.
Renal function assessment
Renal function was assessed by urine output and blood parameters including creatinine level and glomerule filteration rate (eGFR). Kidney Disease Improving Global Outcome criteria was used to assess acute renal failure. Creatine and eGFR preoperation, Day 1 post-operation and before discharge was evaluated (Tables 1 and 2).
Discussion
CRS and HIPEC is the mainstay of treatment for patients with appendiceal tumours metastatic to the peritoneum. CRS involves extensive major surgery with reported procedure-related morbidity ranging from 12% to 67.6% and mortality from 0%–9% [14]. The incidence of AKI after CRS HIPEC reported in the literature ranges between 2% and 22% [6]. Nephrectomy, either radical or partial is the mainstay of surgical treatment for renal cell carcinoma. Nephrectomy itself can cause acute renal failure in up to 53.9% patients and can lead to subsequent chronic renal failure [15, 16] due to the underlying pathological medical conditions or acute loss of nephrons [17]. There is concern that adding nephrectomy to an already high-risk procedure may result in a higher incidence of AKI with an increased operation time leading to potentially higher morbidity and mortality rates. Previous published studies included all types of urological procedures, not only nephrectomy, and reported longer hospital stay and higher major morbidity in patients treated by CRS and HIPEC with a concomitant urological procedure [18, 19]. To our knowledge, there is no publication focusing on synchronous nephrectomy with CRS and HIPEC for appendiceal tumours. This report from a large series of patients undergoing CRS and HIPEC procedures for appendiceal tumour outlines details on the largest series of patients with this unusual dual pathology. The results did not show an increase in AKI, or unexpected morbidity or mortality.
CRS and HIPEC may result in AKI due to the magnitude of surgery with fluid shifts and the potential for dehydration and hypotension but also the addition of HIPEC with perfusion of a potentially nephrotoxic intrabdominal chemotherapy agent. Studies showed HIPEC agents have a low systemic absorption and high concentration in the peritoneum [8, 9, 20]. Cisplatin as a HIPEC agent has been shown to cause renal toxicity and electrolyte metabolic disturbance due to hyperthermia and cytokine release, resulting in musculocutaneous vasodilatation and subsequently causing intravascular volume depletion and organ hypoperfusion [6, 21, 22]. There is also a mechanical effect of application of a large volume of fluid into peritoneal cavity potentially causing systemic inflammatory response [23]. Our unit used low dose of 10 mg/m2 MMC perfused over 60 minutes. Studies on the nephrotoxicity of MMC have reported MMC to be safe with less nephrotoxic effect [7]. No HIPEC dose or application time reduction was used in this series. The use of low dose MMC is postulated to be one of the reasons, but a larger sample size study is required for verification.
Our unit policy involves close monitoring of patients’ renal function and fluid status pre, intra and postoperatively. Dedicated peritoneal malignancy anaesthetists use goal-directed therapy with invasive monitoring of central venous pressure, arterial blood pressure and cardiac output. The cardiac output was monitored with titration of intravenous fluid boluses to maintain stroke volume variation below 10% during the HIPEC phase to maintain hemodynamic stability. Patients have a mean volume of 100 millilitres per kilogram bodyweight with balanced use of gelatin-based colloids, human albumin solution, crystalloids and blood products according to individual patient need [23]. All these measures can help to reduce the risk of postoperative AKI.
Synchronous tumours have been reported in 3.7% of patients diagnosed with RCC [24]. Synchronous kidney cancer and colorectal cancer has a variable incidence ranging in the literature from 0.03%–4.85% [25]. There is no reported incidence specifically on synchronous RCC and appendiceal malignancy with the only previous publication a single case report [26–28]. This paper with a total of 8 patients (out of 2268 appendiceal tumours) (0.35%) is the largest case series so far and as expected the incidence of synchronous malignancies is rare.
Our results suggest little increase in risk from synchronous surgery and we advocate a multidisciplinary approach with imaging reviewed by radiologists and joint discussion with urologists and peritoneal malignancy surgeons for definitive plan and logistic arrangements. This joint understanding is important as peritonectomies and sometimes splenectomy can make later laparoscopic nephrectomy hazardous with risks of duodenal injury (right) or colonic or gastric injury (left) higher due to loss of landmarks and the protective barrier of the peritoneum.
Limitation
This is a retrospective review of a case series from a national referral centre. The results apply to MMC as the HIPEC agent and cannot assume similar findings in patients having other HIPEC agents. However, in view of the rare nature of the disease this series adds to the knowledge base in this field.
Conclusion
Synchronous appendiceal tumours and renal cell carcinoma are rare. Nephrectomy for synchronous renal tumours can be safely combined with CRS and HIPEC.
Conflicts of interest
There is no conflict of interest.
Funding
None declared.



