-
PDF
- Split View
-
Views
-
Cite
Cite
Koyo Fujitsuka, Keisuke Ida, Kosuke Isomura, Machi Shibata, Hiroaki Seo, Satoshi Koizumi, Shinya Mikami, Primary diaphragmatic inflammatory pseudotumour-like follicular/fibroblastic dendritic cell sarcoma mimicking a hepatic tumour: a case report, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag167, https://doi.org/10.1093/jscr/rjag167
Close - Share Icon Share
Abstract
An inflammatory pseudotumour-like follicular/fibroblastic dendritic cell sarcoma (IPT-like FDCS) is a rare Epstein–Barr virus–associated neoplasm that typically arises in the liver or spleen. We present a case of primary diaphragmatic IPT-like FDCS in a woman in her 70s who was incidentally found to have a subdiaphragmatic mass during routine follow-up. Preoperative imaging suggested a hepatic tumor. However, laparoscopic exploration revealed that the lesion originated from the diaphragm. The tumour was completely resected with partial diaphragmatic excision, followed by primary closure. Histopathological examination demonstrated characteristic inflammatory features, and in situ hybridization revealed Epstein–Barr virus–encoded RNA, confirming the diagnosis of IPT-like FDCS. The patient had an uncomplicated postoperative recovery and remained free of disease recurrence at 30 months after surgery. This case underscores the need to include diaphragmatic tumors when evaluating atypical hepatic dome lesions that lack the characteristic imaging features of hepatic malignancy.
Introduction
Primary tumors of the diaphragm are extremely rare, accounting for <0.3% of thoracoabdominal tumours, and are often detected incidentally [1]. Lesions located on the abdominal side of the diaphragm, particularly beneath the hepatic dome, are difficult to differentiate from hepatic tumors on imaging studies, making preoperative diagnosis of a diaphragmatic origin challenging [2–4].
Inflammatory pseudotumor-like follicular/fibroblastic dendritic cell sarcomas (IPT-like FDCS) represent uncommon neoplastic entities that have been reported to be associated with Epstein–Barr virus (EBV) infection. Reported cases are sporadic, and imaging findings are variable and nonspecific, further complicating preoperative diagnosis. Herein, we present a rare case of IPT-like FDCS, arising from the diaphragm that was preoperatively misdiagnosed as a hepatic tumour.
Case report
A woman in her 70s was undergoing an annual abdominal ultrasonography for follow-up of a 7-mm gallbladder polyp. During routine surveillance, a mass was detected beneath the hepatic dome in segment 8, and she was referred to our department for further evaluation. Her medical history included hypertension and dyslipidemia, but was negative for viral hepatitis and excessive alcohol consumption.
Laboratory investigations revealed no remarkable abnormalities. Liver function was preserved, with Child–Pugh score A (6 points). Tumor markers were within normal limits: carcinoembryonic antigen, 1.2 ng/ml; carbohydrate antigen 19–9, 8.9 U/ml; alpha-fetoprotein, 6.9 ng/ml; and protein induced by vitamin K absence or antagonist-II, 19 mAU/ml.
Abdominal ultrasonography demonstrated a well-defined, smooth-surfaced hypoechoic mass measuring ~23 × 19 mm protruding from segment 8 beneath the hepatic dome (Fig. 1A). The lesion contained an anechoic area and a solid component with pulsatile flow (Fig. 1B).
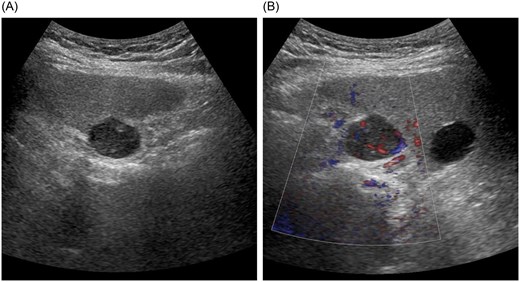
Abdominal ultrasonography findings. (A) A well-defined, smooth-surfaced hypoechoic mass measuring ~23 × 19 mm protruding beneath the hepatic dome in segment 8. (B) The lesion contains an anechoic area and a solid component with pulsatile flow.
Contrast-enhanced computed tomography (CT) showed a mass located beneath the hepatic dome in segment 8, protruding extrahepatically (Fig. 2A). The hepatic margin appeared mildly blunted, suggesting chronic liver changes. The lesion exhibited early enhancement in the arterial phase with prolonged enhancement into the equilibrium phase and only weak washout, findings atypical for hepatocellular carcinoma (Fig. 2B and C).
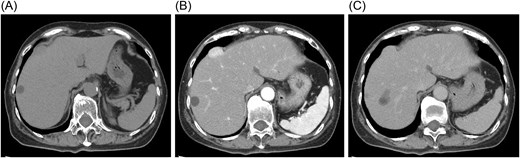
Contrast-enhanced computed tomography findings. (A) The mass is located beneath the hepatic dome in segment 8 and protrudes extrahepatically. (B) The lesion shows early enhancement in the arterial phase. (C) Prolonged enhancement persists into the equilibrium phase without obvious washout.
Based on these findings, hepatocellular adenoma was considered the most likely preoperative diagnosis. However, hepatocellular carcinoma could not be completely excluded, and laparoscopic liver resection was planned for diagnostic and therapeutic purposes.
During surgery, the falciform ligament was divided, and the liver was mobilized to expose the lesion. No tumor was observed on the liver surface. Instead, a localized white nodular lesion was identified on the diaphragm beneath the hepatic dome (Fig. 3A). An intraoperative diagnosis of a diaphragmatic tumor was made. The tumor was completely resected with partial diaphragmatic excision, and the diaphragmatic defect was closed by primary suturing (Fig. 3B and C).
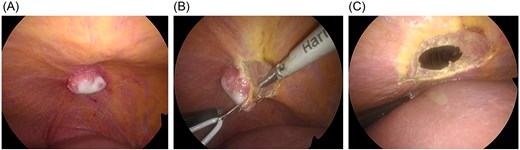
Intraoperative findings. (A) After division of the falciform ligament and mobilization of the liver, no tumor is observed on the hepatic surface; a localized white nodular lesion is identified on the diaphragm beneath the hepatic dome. (B) The tumor shows no continuity with the liver parenchyma and is diagnosed intraoperatively as a diaphragmatic tumor. Partial diaphragmatic resection is performed. (C) The diaphragmatic defect is closed by primary suturing after tumor resection.
Histopathological examination revealed a lesion composed of a diffuse infiltration of lymphocytes and plasma cells with scattered follicle-like structures containing large, atypical cells (Fig. 4A). Spindle-shaped cells were positive for smooth muscle actin and CD23. Hybridization analysis demonstrated Epstein–Barr virus–encoded RNA (EBER) signals, supporting the presence of EBV infection (Fig. 4B–D). Taken together, these findings led to a diagnosis of IPT-like FDCS.
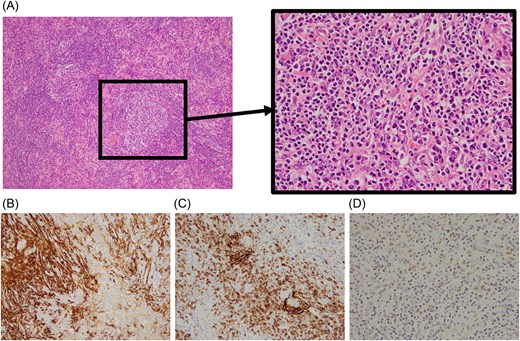
Histopathological findings. (A) Diffuse infiltration of lymphocytes and plasma cells with scattered follicle-like structures containing large, atypical cells (hematoxylin and eosin staining). (B) Tumor cells show positivity for smooth muscle actin. (C) Proliferation of CD23-positive cells is observed. (D) In situ hybridization demonstrates positivity for Epstein–Barr virus–encoded RNA (EBER), confirming EBV infection.
The patient experienced an uncomplicated postoperative recovery and was discharged on postoperative day 5. At 30 months follow-up, there was no evidence of disease recurrence.
Discussion
Primary diaphragmatic tumors are exceedingly rare, with the first case reported at autopsy in 1868 [5]. IPT-like FDCS is a rare tumor that most commonly arises in the liver or spleen, with fewer than 150 cases reported worldwide to date [6]. To our knowledge, no cases of diaphragmatic origin have been previously reported, making this an exceptionally rare presentation.
FDCS is regarded as a malignant neoplasm, while IPT-like FDCS represents a separate entity with histopathological features similar to those of an inflammatory pseudotumor. Unlike conventional FDCS, IPT-like FDCS does not consistently express follicular dendritic cell markers such as CD23. Additionally, expression of smooth muscle actin, CD68, and desmin have been reported, suggesting differentiation toward fibroblastic reticular cells. Given this immunophenotypic diversity, detection of EBV infection by EBER in situ hybridization is regarded as a critical diagnostic feature [7].
IPT-like FDCS predominantly affects the liver and spleen and has been reported more frequently in young to middle-aged women. Due to marked infiltration of inflammatory cells such as lymphocytes and plasma cells, it may be misdiagnosed as a benign inflammatory pseudotumor. Although the clinical course is generally indolent and the tumor is often classified as low-grade, recurrence has been reported, and long-term follow-up is recommended, even after complete resection [8].
Imaging findings typically demonstrate a relatively large solid mass with contrast enhancement on CT or magnetic resonance imaging, sometimes accompanied by cystic changes or necrosis. However, no specific imaging features are diagnostic, and histopathological examination with EBV testing is essential for a definitive diagnosis [9].
Because of their anatomical proximity to the lung and liver, diaphragmatic tumors may be misdiagnosed as hepatic tumors, leading to inappropriate surgical planning [10]. Conversely, cases of exophytic hepatic tumors, initially suspected to be diaphragmatic in origin, have also been reported [11]. Approximately 40% of diaphragmatic tumors are malignant, including sarcomas, underscoring the importance of an accurate preoperative assessment [12].
In the present case, the tumor was located directly beneath the hepatic dome and lacked typical imaging features of hepatic malignancy. Retrospective review of contrast-enhanced CT revealed that the inferior phrenic artery served as the feeding vessel, a finding suggestive of diaphragmatic origin. When evaluating atypical subhepatic dome lesions, diaphragmatic tumors should therefore be included in the differential diagnosis.
Conflicts of interest
The authors declare no conflict of interest.
Funding
This study received no funding.
Data availability
All data generated or analyzed during this study are included in this published article.
Consent
Written informed consent was obtained from the patient for publication of this case report.
Guarantor
Keisuke Ida.
References
- herpesvirus 4, human
- in situ hybridization
- liver neoplasms
- dendritic cells
- follow-up
- preoperative care
- sarcoma
- surgical procedures, operative
- diagnosis
- diagnostic imaging
- respiratory diaphragm
- liver
- neoplasms
- spleen
- liver cancer
- rna
- interpersonal psychotherapy
- pseudotumor
- excision
- diaphragmic neoplasms
- histopathology tests



