-
PDF
- Split View
-
Views
-
Cite
Cite
Brendan M Flynn, Amy M Wang, Nicholas L Michael, Ariel N Kleydman, Matthew D Nealeigh, A case of metaplastic breast cancer presenting as a fungating breast wound, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag152, https://doi.org/10.1093/jscr/rjag152
Close - Share Icon Share
Abstract
Metaplastic breast cancer (MBC) is a rare and aggressive breast neoplasm, accounting for less than 1% of all breast cancer diagnoses. We report the case of a 74-year-old female who presented with a rapidly enlarging breast mass causing a worsening, non-healing wound. A biopsy was consistent with metaplastic breast cancer, and the patient underwent upfront surgery and adjuvant chemotherapy. Unfortunately, after three rounds of chemotherapy, she passed away at home without a clear etiology. Histopathologic diagnosis can be challenging due to the variety of cell types and associated metaplasia involved. This case demonstrates a dramatic presentation of metaplastic breast cancer and the challenges associated with diagnosing and treating this rare disease.
Introduction
Metaplastic breast cancer (MBC) is a rare and aggressive subtype of breast carcinoma, accounting for less than 1% of all breast malignancies and characterized by histologic heterogeneity and poor clinical outcomes. Its variable pathologic features often complicate diagnosis, and optimal management strategies remain ill-defined. This case report underscores the diagnostic challenges, aggressive clinical behavior, and therapeutic considerations associated with this rare neoplasm in an elderly patient that presented with a rapidly progressive, fungating breast mass.
Case report
A 74-year-old female with a past medical history of chronic lymphocytic leukemia (CLL) and hypertension presented to our emergency department with complaints of right breast changes for the past 4–6 months. She had reported increased swelling, tenderness, and new-onset drainage from a non-healing wound over her right breast. Her physical exam was notable for a right breast with a large (~ 11 cm), firm, erythematous mass with central ulceration, and skin thickening but minimal associated tenderness (Fig. 1). She had no clinical evidence of lymphadenopathy nor nipple drainage. Laboratory values were only significant for a leukocytosis of 30 × 103/mcL. Her last mammogram was in 2015 where a BI-RADS 3 lesion was detected in the left breast only, but the patient did not obtain the recommended repeat imaging.
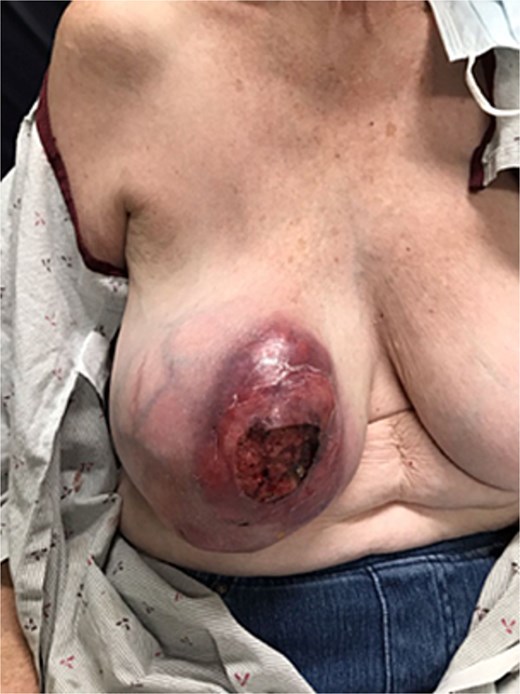
Patient's right breast on initial presentation demonstrates a large, firm, erythematous mass (~11 cm) with central ulceration and overlying skin thickening.
Her workup was expedited given the expected diagnosis of malignancy, with the differential diagnosis including a neoplasm arising from breast tissue that had ulcerated, or an advanced squamous cell carcinoma. Ultrasound of her right breast and a diagnostic mammogram were performed that noted a mixed solid/cystic BI-RADS 5 lesion within the right central breast with extension to the nipple (Fig. 2). There were no abnormal lymph nodes appreciated on breast imaging. A CT of the chest, abdomen, and pelvis was obtained for staging which showed multiple stable sub-6 mm nodules in the left lung, <1 cm mediastinal and hilar lymph nodes, and multiple additional suspicious nodes in the right axillary, supraclavicular, and neck chains. A bone scan was obtained to complete the staging workup, which did not show any evidence of osteoblastic metastatic disease. A core needle biopsy of the solid component demonstrated ‘invasive malignant squamous cells surrounded by cellular, spindled, and proliferative stroma in an inflammatory background’. The lesional cells were highlighted with p63, CK5/6, and GATA3, and were negative for desmin, SMA, SOX10, and CD34. Mucicarmine showed focal intracellular mucin. AE1/AE3 was used to evaluate the presence of carcinoma in sentinel lymph nodes obtained during upfront surgery. Cytology was obtained from the cystic component that was concordant with pathologic findings from the core needle biopsy (Fig. 3).
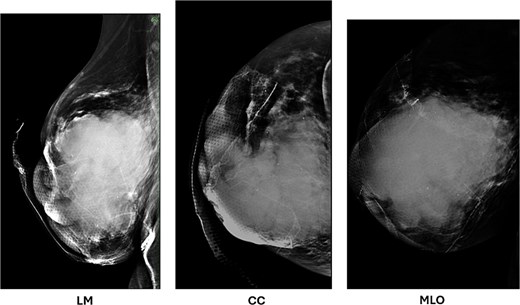
Diagnostic mammogram of the right breast demonstrates a mixed solid and cystic lesion in the central breast (BI-RADS 5), with extension to the nipple.
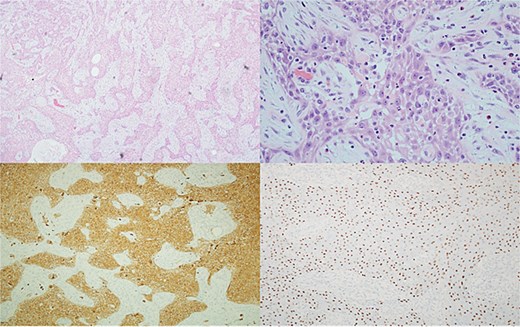
Top left: Tumor cells arranged in cords and trabeculae infiltrate through the background breast stroma. Top right: Tumor cells have nuclear atypia and abundant eosinophilic cytoplasm consistent with squamous differentiation with surrounding desmoplastic stromal response and inflammation. Bottom left, bottom right: Diffuse positive cytoplasmic staining with CK5/6 and nuclear staining with p63, respectively.
Upfront curative-intent surgery was advised by our multidisciplinary tumor board given the open, ulcerated wound associated with the tumor. Two weeks following initial presentation, the patient underwent a right simple mastectomy with sentinel lymph node biopsy, with flat closure (Figs 4 and 5). With an open, infected wound and urgency of proceeding to systemic treatment, reconstruction was deferred. The large fungating mass was excised and two axillary sentinel lymph nodes were obtained. A palpable node in her inframammary fold was also identified and excised. The procedure was uncomplicated, and the patient was discharged from the hospital on post-operative day one.
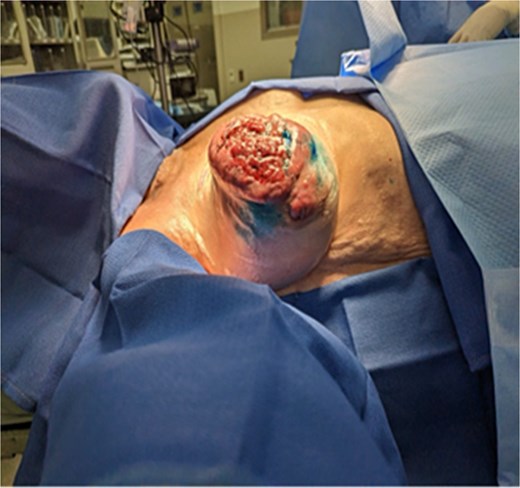
Preoperative image obtained before simple mastectomy with sentinel lymph node biopsy demonstrating rapid interval progression over a short two-week period, with marked increase in size, worsening skin involvement, and progression of ulceration.
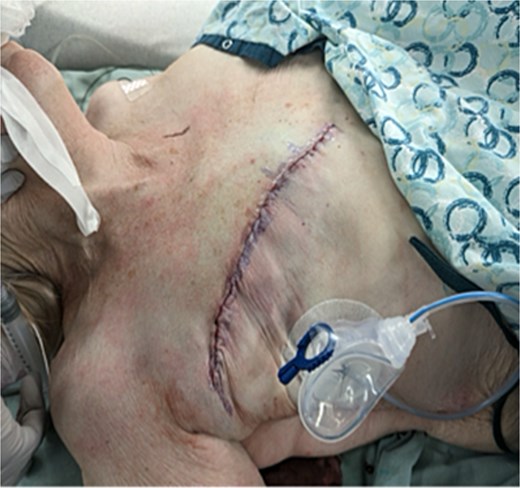
Post-operative flat closure incision after right simple mastectomy and sentinel lymph node biopsy.
Surgical pathology results revealed stage IIIb disease (pT4bN1mi(sn)M0) triple-negative metaplastic breast carcinoma. Her case was re-presented at our multidisciplinary tumor board where she was deemed surgically complete with recommendations for ongoing treatment with adjuvant chemotherapy utilizing the ddAC-T regimen (Adriamycin, Cytoxan, Taxol) in addition to chest wall and regional nodal radiation.
The patient underwent three cycles of adjuvant chemotherapy prior to suddenly passing away at home approximately two months after surgery.
Discussion
The characteristic pathologic finding for MBC is the existence of heterogeneity in cell type metaplastic differentiation [1], and it has a propensity for locoregional recurrence and metastasis despite treatment [2]. Histopathologic diagnosis can thus be challenging, due in part to the variety of cell types and associated metaplasia involved; MBC may be initially misdiagnosed as a lower-grade breast neoplasm of either mesenchymal or epithelial origin. When lacking clear cell type heterogeneity or a component of a mammary carcinoma, immunohistochemical testing for p63, myoepithelial cell markers and keratin can be of use [2, 3]. A multidisciplinary approach to these patients, including specialty pathologists, is critical. A clear treatment algorithm for MBC remains elusive. MBC is almost universally a triple-negative breast cancer (TNBC) [4], and MBC follows the algorithm for other TNBC [5], with neoadjuvant systemic therapy as indicated based on staging, followed by surgery, and adjuvant chemoradiotherapy. In this case, the rapidly progressing fungating mass with an open wound prompted upfront surgery instead of neoadjuvant chemotherapy. Involvement of the skin, including ulceration, has been previously reported and is consistent with the high-grade and rapidly progressive nature of this neoplasm [6]. In contrast to other TNBC, MBC has a significantly worse prognosis, with a higher degree of treatment resistance and lower overall survival [7]. As with other breast malignancies, skin involvement is predictive of poor clinical outcome.
This case demonstrates a dramatic presentation of metaplastic breast cancer and the challenges associated with diagnosing and treating this rare disease.
Conflicts of interest
None declared.
Funding
None declared.



