-
PDF
- Split View
-
Views
-
Cite
Cite
Ahmed Refaat, Saqer Alenezi, Rawan AlShammari, Joud Abdulraheem, Latifa AlKandari, Abdulaziz Alrubaiaan, Ruptured jejunal gastrointestinal stromal tumor (GIST) presenting with acute hemoperitoneum in a young adult with autosomal dominant polycystic kidney disease (ADPKD), Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag148, https://doi.org/10.1093/jscr/rjag148
Close - Share Icon Share
Abstract
Gastrointestinal stromal tumors (GISTs) are rare mesenchymal neoplasms of the gastrointestinal tract, most commonly arising in the stomach and less frequently in the small intestine. Jejunal GISTs often present with vague symptoms, leading to delayed diagnosis, while acute presentations with hemodynamic instability are uncommon and usually result from tumor necrosis or rupture. We report a 37-year-old man with autosomal dominant polycystic kidney disease who presented with acute abdominal pain and shock requiring immediate resuscitation and intensive care admission. Ultrasonography revealed free intraperitoneal fluid and a vascular suprapubic mass. On contrast enhanced computed tomography a heterogeneous necrotic tumor with mesenteric fat stranding and free fluid was identified. Emergency laparotomy identified a hemorrhagic jejunal tumor with hemoperitoneum, which was resected en bloc with primary anastomosis. Histopathology confirmed a moderate-risk GIST. Early radiologic localization and prompt surgical intervention were lifesaving.
Introduction
Gastrointestinal stromal tumors are uncommon mesenchymal neoplasms of the gastrointestinal tract, accounting for 0.1%–3% of all gastrointestinal tumors and representing the most common mesenchymal tumors of the gut [1]. Most gastrointestinal stromal tumors (GISTs) arise in the stomach (54%–70%), followed by the small intestine (20%–30%) [2], with an estimated annual incidence of 10–15 cases per million population and a median age at diagnosis of 60–65 years; occurrence in patients younger than 40 years is rare [3].
Approximately 5%–10% of cases are associated with hereditary syndromes, including Carney triad, Carney–Stratakis syndrome, neurofibromatosis type 1, and familial KIT- or PDGFRA-related GISTs [4]. Small bowel GISTs often grow exophytically and remain asymptomatic until complications such as hemorrhage, rupture, or septic shock occur, which are uncommon but potentially fatal [5, 6]. Diagnosis is challenging due to nonspecific symptoms and limited sensitivity of early imaging. Historically, GISTs were misclassified as smooth muscle or neural tumors until the discovery of KIT (CD117) expression in 1998 established GIST as a distinct pathological entity [7]. According to the WHO classification, GISTs are defined by characteristic morphology and immunophenotype, most commonly expressing KIT (CD117) and DOG1 [8] (Table 1).
| Category . | Criteria . |
|---|---|
| Essential | Intramural or subserosal mass |
| Spindle, epithelioid, or mixed morphology | |
| KIT (CD117) and/or DOG1 positivity | |
| SDH deficiency in specific subtypes | |
| Desirable | KIT or PDGFRA mutations (~85%) |
| Category | Criteria |
|---|---|
| Essential | Intramural or subserosal mass |
| Spindle, epithelioid, or mixed morphology | |
| KIT (CD117) and/or DOG1 positivity | |
| SDH deficiency in specific subtypes | |
| Desirable | KIT or PDGFRA mutations (~85%) |
We report a rare case of a large necrotic jejunal GIST presenting with acute abdomen and shock in a young adult with ADPKD. This case has important educational value by highlighting the aggressive potential of small bowel GISTs and emphasizing the critical role of timely radiologic localization and appropriate surgical decision-making. To our knowledge, this is the first reported case of an acutely ruptured jejunal GIST in a patient with ADPKD.
Case report
A 37-year-old man with hypertension secondary to autosomal dominant polycystic kidney disease presented to the emergency department with sudden-onset severe generalized abdominal pain and dizziness. He denied trauma, fever, hematemesis, or melena, and had no prior surgical history. On arrival, he was critically ill with hypotension (80/50 mmHg), tachycardia (120 beats/min), pallor, and diaphoresis. Abdominal examination revealed generalized tenderness with guarding and rigidity. Bedside ultrasonography demonstrated free intraperitoneal fluid and a vascular suprapubic mass separate from the urinary bladder (Fig. 1). Laboratory investigations showed leukocytosis (15 × 109/L), markedly elevated C-reactive protein (400 mg/L), elevated serum lactate (4 mmol/L) hemoglobin level (Hb 9.5 g/dL), and acute kidney injury with serum creatinine of 300 μmol/L (baseline 160 μmol/L). He was admitted to the intensive care unit with an acute abdomen and shock. Immediate resuscitation was initiated with aggressive crystalloid intravenous fluid, broad-spectrum intravenous antibiotics (piperacillin–tazobactam) and close hemodynamic monitoring.
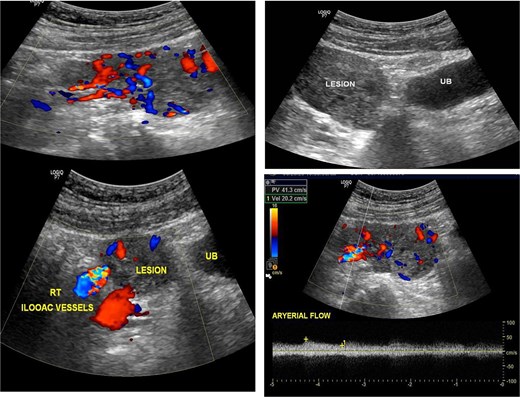
Showed ultrasonic images revealed a large vascular heterogenous supra-pubic mass, separable from the urinary bladder (UB), the peak systolic velocity within the mass = 41 cm/s.
Following hemodynamic stabilization, and improvement in lactate levels, further imaging was pursued to identify the source of hemoperitoneum and guide surgical management. Despite underlying renal dysfunction, a contrast-enhanced computed tomography (CT) was performed after multidisciplinary discussion with the nephrology team, as the potential diagnostic benefit was considered to outweigh the risk of contrast exposure. Renal protective measures were implemented, including optimization of intravascular volume and avoidance of nephrotoxic agents. Contrast-enhanced CT demonstrated a 7.0 × 8.6 × 7.7 cm exophytic jejunal mass with central necrosis, peripheral enhancement, mesenteric fat stranding, and minimal free intraperitoneal fluid, without pneumoperitoneum (Fig. 2). Radiologic features suggestive of malignant GIST included tumor size >5 cm, exophytic growth, and central necrosis [9].
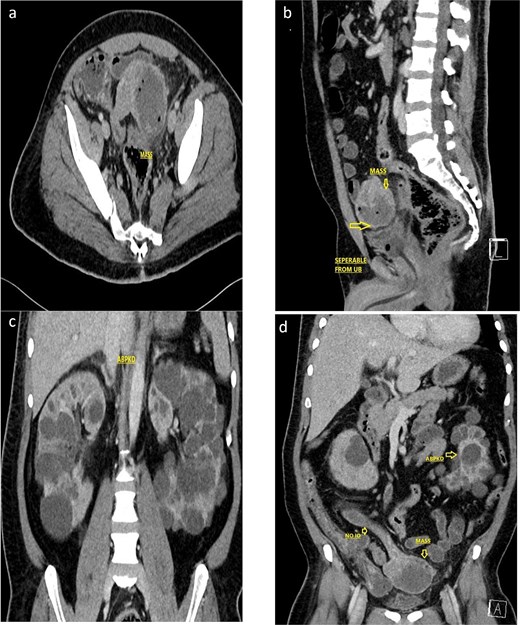
Contrast enhanced CT scan of the abdomen and pelvis (a, axial; b, sagittal) showed a 7.0 × 8.6 × 7.7 cm, exophytic mass having cystic and soft tissue components, a central hypodense area with air foci within, arising from the Jejunal loop, with mesenteric fat stranding and suspected intramural invasion. Associated findings included pericholecystic fluid, sub-centimetric mesenteric lymphadenopathy, subtle mesenteric fatty strandings, and bilateral renal cysts. Not associated with any intestinal obstruction or any obvious mesenteric tethering. The lesion is identified separable from the urinary bladder. (c, d) Coronal CT imaging showing the polycystic kidney disease.
The patient proceeded without delay to emergency exploratory laparotomy. Intraoperatively, ~700 mL of hemorrhagic intraperitoneal fluid was identified along with a necrotic jejunal mass located 80 cm distal to the ligament of Treitz. En bloc resection with primary jejuno-jejunal anastomosis was performed (Fig. 3). The patient received two units of packed red blood cell transfusion intraoperatively. A surgical vacuum drain was placed intra-operatively, and a nasogastric tube was inserted for postoperative decompression.
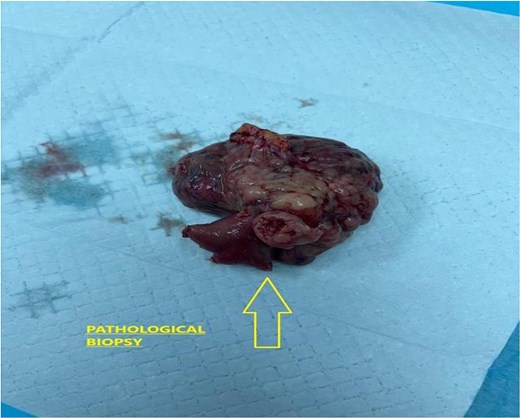
Showed gross specimen of the resected jejunal segment with extensive hemorrhage and central necrosis.
The patient was transferred to the intensive care unit postoperatively for observation. Postoperative hemoglobin level was 9 g/dl, with no further deterioration in renal function, and vital signs remained stable. He did not require vasopressor support, sedation, or supplemental oxygen. He was transferred to the surgical ward on postoperative day 2.The nasogastric tube and surgical drain were removed, and oral intake was resumed on day 4, and the patient was discharged home on day 7 without complications. Follow up at 6 months showed complete symptom resolution and stable hemoglobin levels, with no recurrence on surveillance imaging.
Histopathology confirmed a mixed-type gastrointestinal stromal tumor measuring 9.0 × 6.0 × 4.0 cm with extensive necrosis (~70%) and a low mitotic rate (2/5 mm2). Resection margins were negative, and immunohistochemistry was positive for CD117 and SMA, with a low Ki-67 index (Fig. 4, Table 2). Based on tumor size, location, mitotic rate, and necrosis, the tumor was classified as moderate risk for progressive disease [10].
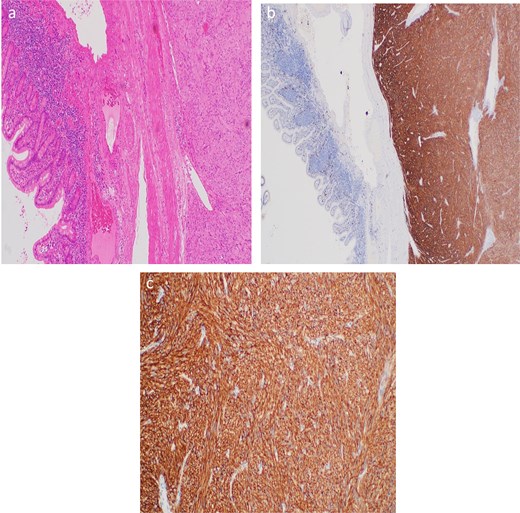
(a) Short intersecting fascicles of uniform spindle-shaped cells with pale eosinophilic cytoplasm and elongated nuclei. (b) Diffuse CD117 positivity. (c) X400 diffuse CD117 positivity.
| Feature . | Result . |
|---|---|
| Histologic type | Mixed-type GIST |
| Mitotic rate | 2/5 mm2 |
| Necrosis | ~70% |
| Grade | G1 (low-grade) |
| Margins | Negative (tumor <1 mm from inked margin) |
| Lymph nodes | None identified |
| CD117 (KIT) | Positive |
| SMA | Positive |
| CD34 | Negative |
| S100 | Negative |
| Desmin | Negative |
| Ki-67 | Low |
| Feature | Result |
|---|---|
| Histologic type | Mixed-type GIST |
| Mitotic rate | 2/5 mm2 |
| Necrosis | ~70% |
| Grade | G1 (low-grade) |
| Margins | Negative (tumor <1 mm from inked margin) |
| Lymph nodes | None identified |
| CD117 (KIT) | Positive |
| SMA | Positive |
| CD34 | Negative |
| S100 | Negative |
| Desmin | Negative |
| Ki-67 | Low |
Discussion
Small bowel GISTs represent a distinct and challenging subgroup due to their rarity, aggressive behavior, and delayed diagnosis. Compared with gastric GISTs, small bowel tumors are more frequently malignant, present at larger sizes, and have higher rates of complications such as hemorrhage, necrosis, and rupture [5, 9]. Jejunal GISTs commonly exhibit exophytic growth, allowing tumors to enlarge before causing luminal symptoms, which are often nonspecific until catastrophic events occur.
An acute presentation with hemodynamic instability and septic shock is uncommon but recognized in the setting of tumor necrosis or rupture. Ruptured jejunal GISTs may present with hemoperitoneum, peritonitis, or systemic inflammatory response, requiring urgent surgical intervention [4, 11]. In this case, extensive tumor necrosis (~70%) likely contributed to intraperitoneal hemorrhage and secondary sepsis. Central necrosis and rupture are adverse prognostic features independent of mitotic activity [10, 12].
Radiologic evaluation played a critical role in the management. Contrast-enhanced CT is the preferred modality, typically demonstrating an exophytic mass with heterogeneous enhancement, central necrosis, and mesenteric fat stranding [9]. This case highlights the value of contrast enhanced CT, performing after hemodynamic stabilization, in identifying the source of hemoperitoneum and guiding timely surgical intervention, even in patients with underlying renal dysfunction.
The coexistence of GIST and autosomal dominant polycystic kidney disease appears coincidental, with no established causal association. Chronic kidney disease complicates imaging and perioperative care, emphasizing the need for multidisciplinary management. Although sporadic GISTs usually present in older adults, young age should not reduce diagnostic suspicion.
Complete surgical resection with negative margins remains the cornerstone of treatment [13]. Lymphadenectomy is unnecessary due to rare nodal involvement. Risk stratification based on tumor size, location, mitotic rate, and necrosis is essential [10]. Despite a low mitotic index, the tumor’s size, jejunal location, and extensive necrosis placed this patient at moderate risk, warranting close surveillance and consideration of adjuvant imatinib therapy [14].
Conclusion
Early radiologic localization and prompt surgical resection were critical for survival, emphasizing the aggressive potential of small bowel GISTs and the need for timely intervention. Clinicians should consider GIST in unexplained intra-abdominal hemorrhage regardless of patient age, with appropriate risk stratification and close postoperative surveillance to optimize outcomes.
Acknowledgements
Special thanks to the nurses, staff, and doctors who were directly or indirectly involved in the care of the patient.
Conflicts of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data availability
Not applicable.



