-
PDF
- Split View
-
Views
-
Cite
Cite
Xiaoxia Li, Likaqi Wu, Pin Gao, Lianpeng Gao, Chaoxian Xiong, Qi hang Pei, Xuefeng Cha, Hongtao Zhang, Case report: diagnosis and surgical management of a rare malignant isolated fibrous tumor of the pelvis, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag140, https://doi.org/10.1093/jscr/rjag140
Close - Share Icon Share
Abstract
Solitary fibrous tumor (SFT) is an uncommon fibroblastic mesenchymal neoplasm. Malignant SFT in the abdominopelvic compartment,particularly originating from the anterior abdominal wall and peritoneum is exceptional. A 72-year-old woman presented with recurrent right lower-quadrant pain. Imaging demonstrated a ~13 cm heterogeneous, hypervascular mixed cystic solid pelvic mass supplied by the external iliac arteries and abutting the bowel and bladder. At operation, an encapsulated mass was found densely adherent to the peritoneum with rich peritumoral vascular connections; feeding vessels were individually clamped and secured, and the tumor was removed completely with en bloc excision of the adherent peritoneum. Pathology confirmed malignant SFT (necrosis and increased mitotic activity) with supportive immunophenotype (focal CD34/partial STAT6). The patient recovered uneventfully and remained recurrence-free on computed tomography at 12 months. This case highlights the value of vascular mapping for safe, complete resection.
Introduction
A 72-year-old woman was admitted on 27 March 2024, 12 days prior to admission, she developed persistent right lower quadrant abdominal pain of unclear etiology, without radiation, tenderness, rebound tenderness, fever, nausea, or vomiting. She was evaluated at another institution, where ultrasonography revealed a solid pelvic mass with echogenic features suggestive of uterine fibroids. A solid hypoechoic mass measuring ~13.8 × 10.2 cm was identified in the right pelvic region. Symptomatic treatment alleviated the abdominal pain. Two days before admission, the pain recurred, prompting presentation to our hospital.
At presentation, contrast-enhanced computed tomography (CT) demonstrated a multiloculated mixed cystic–solid mass in the lower abdomen/pelvis with internal septations, measuring ~13.2 × 9.5 × 12.4 cm (Fig. 1). The lesion was well outlined overall but showed indistinct planes with the adjacent bowel and the anterior bladder wall. Enhancement was heterogeneous, with multiple non-enhancing low-density foci consistent with necrosis. Arterial feeders arose from both external iliac arteries. Magnetic resonance imaging (MRI) confirmed a predominantly solid, lobulated mass (12.3 × 9.6 × 13.7 cm; Fig. 2) with heterogeneous signal (T1 isointense; variable T2 signal) and mildly increased diffusion signal; the minimum ADC was ~0.6 × 10−3 mm2/s. Small cystic/necrotic areas were present, and the tumor appeared locally adherent to the bowel and bladder, favoring a mesenchymal neoplasm.
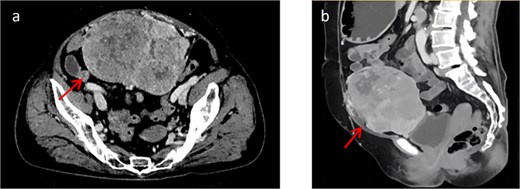
(a) Shows a cross-sectional view of the pelvic contrast-enhanced CT scan. (b) Shows a sagittal view of the pelvic contrast-enhanced CT scan.
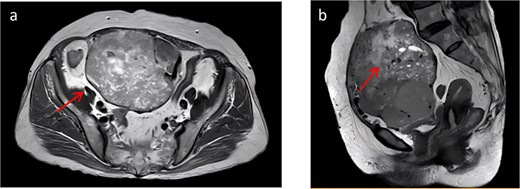
(a) Shows a cross-sectional view of pelvic contrast-enhanced MRI. (b) Shows a sagittal view of pelvic contrast-enhanced MRI.
After surgical contraindications were excluded, tumor resection was performed on 8 April 2024. Intraoperatively, the anterior abdominal wall sheath was incised, revealing a ~10 × 10 cm encapsulated mass with an intact capsule. The tumor base was densely adherent to the peritoneum, with prominent peritumoral vascular connections. Following peritoneal incision and entry into the abdominal cavity, re-exploration demonstrated a clean peritoneal cavity without ascites or additional mass lesions. The tumor, together with the adherent peritoneum, was mobilized using a combination of sharp and blunt dissection. Feeding vessels were identified; several were clamped and secured with sutures. The lesion was completely excised and submitted for pathological evaluation.
Postoperative pathology: Grossly, a well-circumscribed, encapsulated gray–white to gray–red mass was identified in the lower anterior abdominal wall, measuring ~15 × 9.6 × 8 cm. The cut surface was solid and slightly soft, and the capsule was thin (~0.1 cm). Microscopically (Fig. 3), a spindle-cell neoplasm with necrosis and a mitotic rate >4/10 high-power fields (HPFs) was observed; Van Gieson (VG) and silver stains were positive. Immunohistochemistry (Fig. 4) showed Vim (+), CK (−), Ki-67 (~20%), SMA (−), Actin (−), Desmin (−), Caldesmon (−), S100 (−), CD34 (focal +), CD117 (−), DOG1 (−), TLE1 (−), CD99 (+), MyoD1 (−), Myogenin (−), SOX10 (−), CD56 (−), FLI1 (−), Syn (−), CgA (−), STAT6 (partial +), HMB45 (−), Melan-A (−), CD10 (−), Cyclin D1 (−), CD31 (vascular +), Bcl-2 (+), TFE3 (±), and INI1 (+). Overall, the findings supported malignant solitary fibrous tumor arising from the lower anterior abdominal wall.
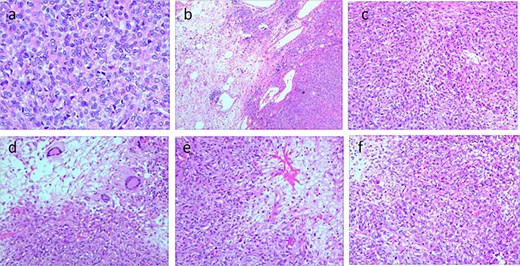
(a–d) Histological features of the tumor tissue. Positive for VG and silver stains; hematoxylin and eosin (H&E) staining reveals diffuse proliferation of spindle-shaped tumor cells arranged in bundles or wheel-like patterns, abundant collagen fibers in the stroma, areas of necrosis, and mitotic figures >4 per 10 high-power fields.
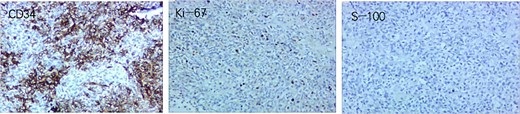
Tumor immunohistochemistry results. CD34 focal positive; Ki-67 positive rate ~20%; S-100 negative.
A 1-year postoperative follow-up CT scan demonstrated no evidence of recurrence.
Discussion
SFT is a fibroblastic mesenchymal tumor now recognized across a broad anatomic spectrum. Thoracic lesions remain the most common, whereas primary abdominopelvic disease is far less frequent. In the current World Health Organization (WHO) framework, SFT is regarded as a tumor of intermediate malignant potential, with clinical behavior ranging from indolent to overtly aggressive [1–3].
Deep abdominal or pelvic SFTs may enlarge for a prolonged period without specific symptoms and are often detected only after they compress adjacent organs [1]. A distinctive but uncommon presentation is paraneoplastic hypoglycemia [4, 5]. In our patient, pain was the presenting complaint, and imaging suggested a hypervascular mesenchymal mass closely related to the bladder and bowel, underscoring the diagnostic difficulty in this location.
Imaging appearances vary because cellularity, collagen content, hemorrhage, and necrosis differ between tumors [6–9]. On CT, a well-encapsulated lesion with brisk enhancement and non-enhancing necrotic components is frequently encountered. MRI typically demonstrates intermediate T1 signal with variable T2 signal; lower T2 signal often reflects collagen-rich stroma. In this case, the mixed cystic–solid architecture with septations and the rich arterial supply from the external iliac system were particularly helpful for surgical planning and for anticipating intraoperative bleeding risk.
Because no pathognomonic clinical or radiologic features exist, definitive diagnosis rests on histomorphology and immunophenotyping of the resected specimen [10, 11]. Classic microscopy shows a spindle-cell proliferation with alternating hypocellular and hypercellular areas and branching vessels. CD34 and STAT6 are useful markers, although CD34 staining may diminish in malignant tumors [12, 13].
Prognostication is largely histology-based. Tumor size, necrosis, and mitotic activity correlate with recurrence and metastasis; many series consider ≥4 mitoses per 10 HPFs as a threshold associated with higher risk [14]. The presence of necrosis and > 4/10 HPFs mitoses in our case, together with Ki-67 ~ 20%, supported malignant potential.
Complete resection with negative margins remains the cornerstone of care for localized malignant SFT [15]. This case highlights the importance of preoperative vascular assessment and en bloc resection in achieving complete removal of hypervascular pelvic SFTs. For unresectable or metastatic disease, radiotherapy and systemic therapy may be considered, although conventional chemotherapy responses are limited; targeted therapy (e.g. pazopanib) and combinations such as temozolomide plus bevacizumab have shown activity in selected reports [16, 17]. Given the possibility of late relapse, prolonged surveillance is advisable, particularly for pelvic tumors [18].
From a surgical perspective, careful delineation of tumor extent and arterial supply on cross-sectional imaging is crucial for operative planning and bleeding risk mitigation, and complete resection remains the cornerstone for local control.
Conclusion
A hypervascular pelvic mass with heterogeneous enhancement should prompt consideration of SFT. Meticulous surgical excision can achieve favorable short-term outcomes, but long-term follow-up remains essential.
Conflicts of interest
The authors declare no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval and consent
The study was conducted in accordance with institutional ethical standards. Written informed consent was obtained from the patient for publication of this case report and accompanying images.
References
Demicco EG, Fritchie KJ, Han A. Solitary fibrous tumour. In: WHO Classification of Tumours Editorial Board, ed. Soft Tissue and Bone Tumours. 5th ed. Lyon, France: International Agency for Research on Cancer, 2020, 104–108.
- computed tomography
- cancer
- heterogeneity
- cd34 antigens
- cysts
- immunophenotyping
- intestines
- necrosis
- neoplasms, fibrous tissue
- pain
- surgical procedures, operative
- urinary bladder
- diagnosis
- diagnostic imaging
- neoplasms
- pathology
- pelvis
- peritoneum
- pelvic mass
- abdominal wall, anterior
- solitary fibrous tumor
- excision
- iliac artery, external



