-
PDF
- Split View
-
Views
-
Cite
Cite
Dionysios Prevezanos, Dimitrios K Vlachos, Konstantinos Chatzidimitriadis, Ioannis-Georgios Zacharias, Stratigoula Sakellariou, Evangelia Ntikoudi, Stylianos Kykalos, Dimitrios Mantas, Low-grade mucinous neoplasm arising in a completely isolated duplication cyst of the abdominal wall: a unique case report, Journal of Surgical Case Reports, Volume 2026, Issue 3, March 2026, rjag139, https://doi.org/10.1093/jscr/rjag139
Close - Share Icon Share
Abstract
Intestinal duplication cysts (IDCs) are rare congenital anomalies, most commonly arising in the small intestine. A particularly uncommon subtype, completely isolated duplication cysts (CIDCs), has an independent blood supply and lacks communication with the native bowel. Although IDCs are typically benign, malignant transformation—including low-grade mucinous neoplasms (LGMNs)—has been reported only rarely. A Caucasian male with a history of infant colostomy for ileus, later reversed, presented with an abdominal wall mass at the former stoma site. Imaging demonstrated a well-encapsulated cystic lesion originating above the posterior rectus sheath, and complete surgical excision was achieved uneventfully. Histopathology revealed a mucin-secreting epithelial lining with low-grade dysplasia, consistent with LGMN, with immunohistochemistry excluding alternative gastrointestinal neoplasms. The patient recovered without complications and showed no recurrence on follow-up. This represents the first documented CIDC with LGMN in the abdominal wall, highlighting the importance of early identification and surgical management.
Introduction
Intestinal duplication cysts (IDCs) are rare congenital anomalies that can arise anywhere along the gastrointestinal (GI) tract, most commonly on the mesenteric side of the bowel, with the small intestine being the most frequently affected site [1, 2]. Their incidence is estimated at ~1 in 4500 live births [3]. IDCs usually present during infancy or early childhood, while adult cases are uncommon [1]. Histologically, they typically share a muscular wall and vascular supply with the adjacent bowel and are lined by gastrointestinal-type epithelium [4].
A particularly rare subtype, known as completely isolated duplication cysts (CIDCs), occurs in ~1 in 10 000 births [4]. Unlike typical IDCs, CIDCs have an independent blood supply and no communication with the native bowel, making preoperative diagnosis challenging. Although most IDCs are benign, malignant transformation has been documented in rare cases. Reported malignancies include adenocarcinoma and neuroendocrine tumors. Low-grade mucinous neoplasms (LGMNs) arising within IDCs are exceedingly rare, with only a few cases reported in the literature [5–7]. We present a rare case of a CIDC of the abdominal wall containing a low-grade mucinous neoplasm.
Case report
A 52-year-old Caucasian male presented with a palpable mass in the abdominal wall at the site of a previous colostomy. His surgical history included a colostomy performed at 15 months of age due to ileus, followed by restoration of digestive continuity 1 month later, as well as a cholecystectomy in adulthood. The patient was a smoker with no other known comorbidities. Physical examination revealed a firm, well-defined, non-tender mass in the area of the previous stoma. The overlying skin was intact, without signs of inflammation. Laboratory investigations, including tumor markers, were within normal limits. Contrast-enhanced computed tomography (CT) demonstrated a well-encapsulated cystic lesion located above the posterior sheath of the rectus abdominis muscle, without communication with the gastrointestinal tract. No lymphadenopathy or other suspicious findings were observed (Figs 1 and 2).
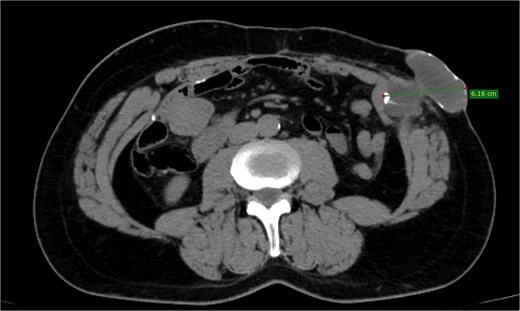
Depicting a well-encapsulated cystic lesion situated above the posterior sheath of the rectus abdominis muscle, with no communication with the bowel, measuring 6.16 cm.
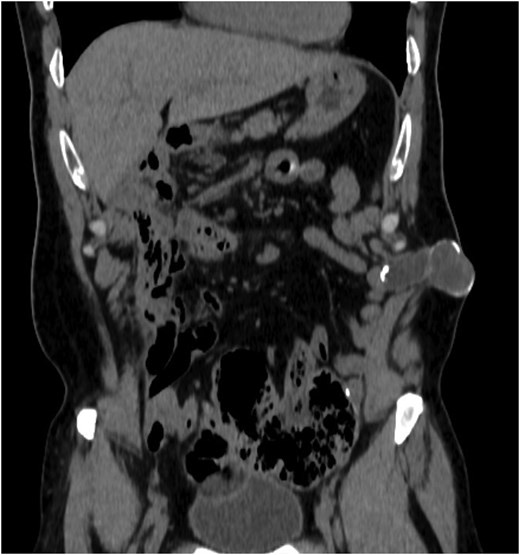
Coronal perception of CT demonstrating the lesion of the abdominal wall.
Based on these findings, surgical excision was planned. Intraoperatively, a well-circumscribed cystic mass originating from the posterior sheath of the rectus abdominis muscle was identified. The lesion had an independent vascular supply and no connection to the gastrointestinal tract. Complete excision was achieved without complications. The rectus abdominis fascia was repaired, and an onlay prophylactic mesh was placed to prevent postoperative herniation.
Results
The postoperative course was uneventful. The patient was mobilized early, tolerated oral intake, and was discharged on postoperative day two in stable condition. Histopathological examination revealed a cystic lesion with a well-formed muscular wall composed of two smooth muscle layers. Nerve fibers were identified within the wall in a distribution resembling the enteric nervous plexus. The inner lining consisted of flat to pseudopapillary epithelium composed of cuboidal to columnar pseudostratified mucin-producing cells with mild cytological atypia. The cyst lumen was filled with acellular mucin. Epithelial proliferation and mucin accumulation disrupted the muscular wall through pushing invasion, with extravasated mucin extending into the surrounding adipose tissue and inducing a granulomatous reaction.
Immunohistochemical staining demonstrated diffuse positivity for CK20, CDX2, and SATB2, with partial positivity for CK7 (Fig. 3a–d). These findings supported the diagnosis of a low-grade mucinous neoplasm arising within a CIDC. The multidisciplinary team recommended surveillance. The patient remained asymptomatic with no evidence of recurrence.
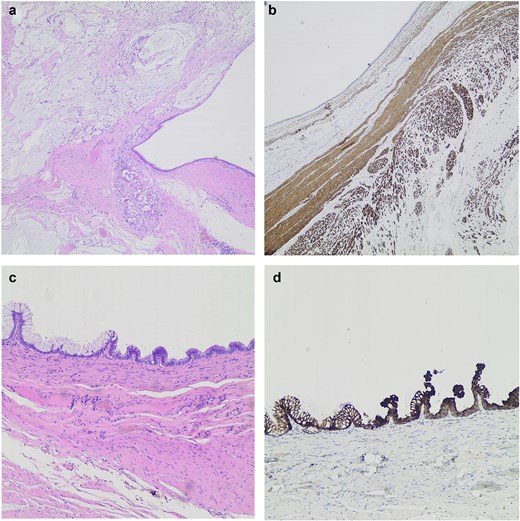
(a) Cystic mass with muscular wall disrupted by mucin (H&E ×40). (b) Two layered muscular wall highlighted by Desmin immunostain (H&E ×40). (c) Pseudopapillary epithelium with columnar pseudostratified mucus-producing cells (H&E ×100). (d) Epithelial lining diffusely positive for CK20 (H&E ×100).
Discussion
This case describes a rare combination of a low-grade mucinous neoplasm arising within a completely isolated duplication cyst located in the abdominal wall, a presentation not previously reported. The etiology of IDCs is considered multifactorial. One embryological theory suggests abnormal development of the neurenteric canal, described as split notochord syndrome by Bentley and Smith [8]. Alternative hypotheses include incomplete recanalization of the embryonic gut or persistence of embryonic diverticula [9]. In the present case, the patient’s history of colostomy in infancy suggests that residual colonic tissue at the stoma site may have contributed to the development of a CIDC within the abdominal wall.
IDCs have been reported most commonly in the ileum, ileocecal valve, esophagus, duodenum, stomach, jejunum, colon, and rectum [1]. Rare extraintestinal locations include the tongue, pleural cavity, retroperitoneum, liver, biliary tree, bronchial tree, and pancreas [2, 10]. Abdominal wall localization of a CIDC has not been previously described. Malignant transformation of IDCs is rare, with adenocarcinoma being the most frequently reported malignancy [11–13]. Other reported neoplasms include small cell neuroendocrine carcinoma [14] and neuroendocrine tumors [2, 15]. LGMNs arising within IDCs are exceedingly rare, with only three previously reported cases [5–7]. Our case represents the first reported instance of an LGMN arising within a CIDC of the abdominal wall.
Conclusion
This case highlights the unique occurrence of a low-grade mucinous neoplasm arising within a completely isolated duplication cyst of the abdominal wall. Given the rarity of CIDCs and their potential for malignant transformation, thorough clinical evaluation and complete surgical excision are essential. This report expands the existing literature and underscores the importance of considering rare congenital anomalies in the differential diagnosis of abdominal wall masses.
Conflicts of interest
The authors declare no conflict of interest, financial, or otherwise.
Funding
No funding was received for this research.
Data availability
The authors have nothing to declare.
Consent for reporting
Informed consent was obtained from the patient.



