-
PDF
- Split View
-
Views
-
Cite
Cite
Brooks Melnyk, Caitlin J Cain-Trivette, Claire Gerall, Jee-Young Nina Ham, William Middlesworth, Clinical management of bilateral functional adrenal adenomas, Journal of Surgical Case Reports, Volume 2026, Issue 1, January 2026, rjaf1051, https://doi.org/10.1093/jscr/rjaf1051
Close - Share Icon Share
Abstract
Adrenocortical tumors are uncommon in the pediatric population. We report a case of a pediatric patient with Beckwith Wiedemann Syndrome (BWS) who was found to have bilateral functional adrenal adenomas: one mineralocorticoid-secreting and one androgen-secreting. The patient ultimately underwent staged surgical resection of both masses, resulting in marked reduction in hormone levels. This case highlights the pathophysiology of adrenal adenomas, association between BWS and adrenal masses, and treatment challenges of bilateral adrenal masses.
Introduction
Adrenocortical tumors are exceedingly rare in children, with about 25 cases diagnosed each year in the United States, 90% of which are functional tumors [1]. The incidence of bilateral adrenal lesions in the pediatric population is not well documented, but is reported to range from 17% to 22% in adults with incidentalomas [2].
BWS is caused by an alteration in 11p15 and is associated with cancer predisposition. Adrenal tumors are the fourth most common following nephroblastoma, neuroblastoma, and hepatoblastoma [3].
Case report
This is a case of an 8-year-old male with BWS who presented to his primary physician with asymptomatic hypertension. Initial hospitalization and workup with renal ultrasound yielded no underlying etiology, but he was started on labetalol, spironolactone, and enalapril. He returned to the emergency department 2 weeks later with symptomatic hypertension with chest pain and headaches.
Imaging workup included a renal ultrasound which revealed bilaterally elevated renal artery velocities and medullary calcinosis consistent with medullary sponge kidney. A subsequent computed tomography (CT) angiogram which showed bilateral adrenal masses, no evidence of renal artery stenosis, and multiple enhancing lesions within the liver. Magnetic resonance imaging (MRI) of the abdomen for further characterization noted the right adrenal mass to be solid, homogenous, with restricted diffusion in the cortex measuring 2.4 × 2.4 × 2.3 cm; the left mass more heterogeneous, with small cystic components, restricted diffusion in the cortex but non-restrictive in the medulla measuring 2.0 × 2.3 × 2.0 cm. The liver nodules appeared to represent multiple areas of focal nodular hyperplasia (Fig. 1). The patient had a positron emission tomography (PET) scan which revealed moderate fluorodeoxyglucose (FDG) avidity in the left adrenal mass (SUV 3.2) and low FDG avidity in the right mass (SUV 2.1).
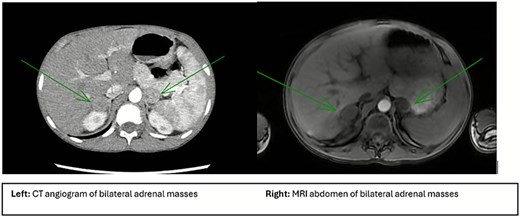
Imaging characteristics of adrenal pathology. Left image shows a CT abdomen and pelvis with arrows pointing to the bilateral adrenal masses measuring 2.5 × 2.0 cm on the right and 2.3 × 2.3 cm on the left. Right image shows an MRI of the abdomen and with arrows pointing to bilateral adrenal masses. Right mass is measured at 2.4 × 2.4 × 2.3 cm and is solid, homogeneous with a more dense capsule and moderate restricted diffusion. The left mass is measured at 2.0 × 2.3 × 2.0 cm and has a more heterogeneous appearance with small cystic components, cortex of the lesion with restricted diffusion.
The patient had laboratory testing for hyperaldosteronism, pheochromocytoma, and adrenocortical carcinoma. He was found to have elevated aldosterone levels at 47.5 ng/dL, aldosterone/renin ratio of 475, and electrolytes within normal limits. Plasma metanephrines, urine metanephrines, cortisol and vanillylmandelic acid were within normal limits. Laboratory workup further identified elevated dehydroepiandrosterone sulfate (DHEAS), testosterone, and androstenedione levels which were thought to be related to puberty given patient was nearly 9 years old.
Following completion of this workup, the patient was thought to have an aldosterone secreting adenoma or less likely adrenocortical carcinoma given low PET avidity. A multidisciplinary meeting was held with plans ultimately for resection of the left adrenal mass due to the higher FDG avidity on PET.
He underwent a laparoscopic left adrenalectomy. A smooth round mass was present at the medial aspect of the left adrenal gland. Adrenalectomy was completed and pathology demonstrated adrenal cortical tumor with favorable histology.
Postoperatively, he was weaned off all antihypertensive medications by 1 month after surgery (Fig. 2).
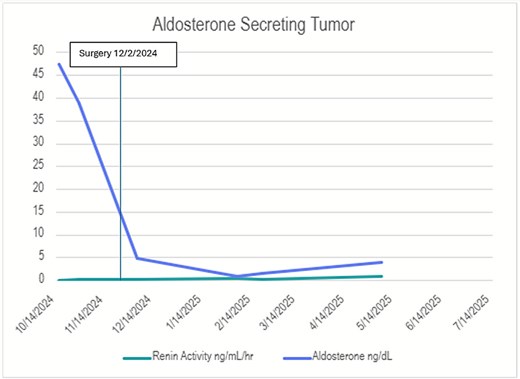
Renin/aldosterone hormone levels before and after resection. This graph shows serial measurements of renin activity (ng/mL/h) and aldosterone (ng/dL) throughout the patient’s clinical course. At presentation, the aldosterone level was markedly elevated at 48 ng/dL (normal value 4.0–44) with concurrently suppressed renin activity levels at 0.1 (normal value 0.5–5.9). Following resection of the left adrenal mass, aldosterone levels declined significantly and renin activity normalized.
The right-sided adrenal mass was re-imaged 3 months post op and remained stable in size. The patient’s DHEAS, testosterone and androstenedione remained elevated, so the patient was taken for resection of the right adrenal mass.
Laparoscopic cortical-sparing resection of his right adrenal mass was performed 6 months after the initial surgery. The mass was found to be round and encapsulated, extending from the anterior surface of the gland. Given the low suspicion for malignancy, the mass was resected along with a small margin of the gland, preserving most of the right adrenal gland to remain in place. The final pathology again revealed adrenocortical tumor with favorable histology. At one month postoperatively, his DHEAS, testosterone and androstenedione levels had normalized. (Fig. 3).
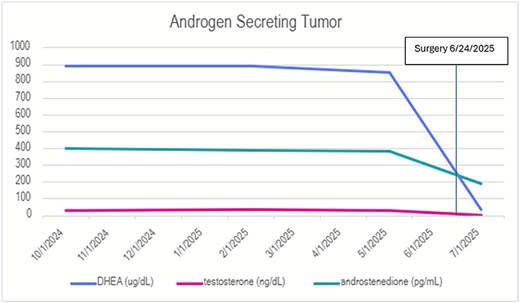
DHEA, testosterone, and androstenedione levels before and after resection. This graph shows serial measurements of DHEA (ug/dL), testosterone (ng/dL), and androstenedione (pg/mL) throughout the patient’s clinical course. At time of presentation, DHEA was elevated to 892 ug/dL (normal value 5–115), testosterone elevated to 28.7 (normal value <2.4), and androstenedione elevated to 403 pg/mL (normal value 30–300). After resection of the right adrenal gland, all of these values normalized.
Discussion
We present an unusual case of synchronous functional adrenal adenomas in an 8-year-old patient with BWS, highlighting the clinical dilemma of surgical decision-making with bilateral adrenal masses.
Functional adrenal adenomas overproduce hormones synthesized in the adrenal cortex. These tumors commonly present with virilization due to excessive androgen production. Less often, patients present with Cushing syndrome due to overproduction of glucocorticoids or hypertension due to an excess of mineralocorticoids [4].
Beckwith- Wiedemann syndrome is associated with a 5%–10% risk of developing an intra-abdominal tumor [3]. Current screening guidelines recommend abdominal ultrasounds 3–4 times per year until the age of 4 [5].
This case presented multiple reasonable operative approaches including bilateral adrenalectomy versus a staged resection. Synchronous bilateral adrenalectomy is an uncommon operation but has been shown to have favorable outcomes [6]. Synchronous bilateral adrenalectomy limits anesthetic exposure as well as the potential for developing adhesions. Benefits of staged resection include lower risk of postoperative adrenal insufficiency and may provide opportunity for adrenal sparing. In our case, we opted to proceed with staged resection due to low concern for rapidly growing malignancy given PET findings, elevations in two different hormonal pathways, and the familiarity of the surgical and postoperative care teams.
After deciding to proceed with a staged resection, we decided to proceed with left side adrenalectomy first due to the higher FDG avidity on PET scan. Following this procedure, his hyperaldosteronism and hypertension resolved. We proceeded with resection of the contralateral mass in conjunction with current guidelines [3]. We approached second surgery with goal of preservation of adrenal function, given low FDG avidity and low suspicion for malignancy of the right adrenal gland. This allowed us to avoid the morbidity of postoperative adrenal insufficiency.
Bilateral adrenal tumors are rare in children. Both synchronous adrenalectomy and staged laparoscopic adrenalectomy have been shown to be safe. We have discussed the benefits of each surgical approach, as well as factors to consider when determining the most appropriate operative approach for future patients.
Conflict of interest statement
None declared.
Funding
None declared.



