-
PDF
- Split View
-
Views
-
Cite
Cite
Lorenz Mertens, Leon Bruder, Jan Carstens, Haidar Haidar, Andreas Greiner, Surgical treatment of a giant aneurysm of the superior mesenteric artery in a patient with systemic lupus erythematosus, Journal of Surgical Case Reports, Volume 2026, Issue 1, January 2026, rjaf1036, https://doi.org/10.1093/jscr/rjaf1036
Close - Share Icon Share
Abstract
Visceral artery aneurysms (VAAs) are rare, representing ˂2% of all arterial aneurysms in adults, with superior mesenteric artery aneurysms (SMAAs) being the third most common type. Systemic lupus erythematosus (SLE) is associated with vascular complications, including vasculitis and antiphospholipid syndrome, which may contribute to aneurysm formation. However, SMAAs in SLE patients are seldom reported. A 45-year-old male patient with SLE and chronic abdominal pain was presented to our clinic. Imaging revealed a 56-mm SMAA with thrombosis and stenosis. Due to rupture risk, open surgical repair was performed using a reversed saphenous vein graft, successfully revascularizing four mesenteric branches. Recovery was uneventful. Although endovascular techniques are less invasive, open surgical repair remains the preferred approach for complex aneurysms involving multiple branches. This case underscores the role of SLE-related vasculitis in aneurysm formation and highlights the need for early diagnosis and intervention.
Introduction
Visceral artery aneurysms (VAAs) are rare, comprising ˂2% of all arterial aneurysms in adults. Superior mesenteric artery aneurysms (SMAAs) are the third most common type of VAA, accounting for only 3%–7% of cases. The most common etiologies of true SMAAs (as opposed to pseudoaneurysms) include nonspecific or degenerative causes, previous superior mesenteric artery (SMA) dissections, and mycotic or infectious causes secondary to endocarditis. However, multiple other causes have been implicated, including connective tissue disorders or rheumatologic diseases [1].
Systemic lupus erythematosus (SLE), a chronic autoimmune disease, is known for its multisystem involvement, including vascular abnormalities. Vasculitis and antiphospholipid syndrome, which are complications commonly seen in patients with SLE, have been implicated in the pathogenesis of arterial aneurysms. Vasculitis occurs in ~11%–36%, while antiphospholipid syndrome is present in 10%–15% of SLE patients, in 30%–40% of cases with detectable antiphospholipid antibodies [2, 3]. However, VAAs in SLE patients remain a rarely reported phenomenon.
Similar to abdominal aortic aneurysms, the treatment of SMAAs involves balancing the risk of rupture with the risk of surgical intervention. Unfortunately, the natural progression and treatment risks remain poorly defined due to the rarity of SMAAs, although rupture risks have been estimated to range from 20% to 50%, with associated mortality rates of 30%–100% [1].
The determinants of treatment include patient comorbidities, the aneurysm’s location, its underlying etiology, presenting symptoms, and the bowel perfusion status. The goals of treatment are to prevent rupture, embolization, and thrombosis while maintaining visceral perfusion and controlling the infectious source in cases of mycotic aneurysms. Open surgical repair with aneurysmectomy and revascularization is the traditional approach, although endovascular techniques have been described more recently [4, 5]. The literature highlights sporadic cases, underscoring both the diagnostic challenges and therapeutic strategies, which include both endovascular and open surgical approaches [6]. This report details the surgical management of a SMAA in a 45-year-old male patient with a 9-year history of SLE, emphasizing the importance of timely intervention and the role of open surgery in achieving definitive management.
Case presentation
A 45-year-old man with a 9-year history of SLE was presented to our vascular surgery department with chronic, recurrent abdominal pain for over a year. The pain was localized to the upper abdomen and was independent of food intake. His medical history was significant for lupus nephritis with nephrotic syndrome, arterial hypertension, hypercholesterolemia, hyperlipoproteinemia (a), a hepatic hemangioma, and a history of herpes zoster. Relevant medications included prednisolone since the diagnosis of SLE, and phased applications of hydroxychloroquine, mycophenolate mofetil, voclosporin, belimumab, and rituximab.
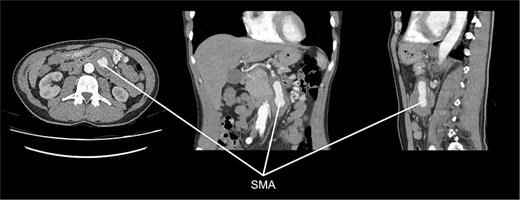
Initial workup included esophagogastroduodenoscopy, which showed no abnormalities. A computed tomography (CT) angiography (Fig. 1) revealed a 56-mm SMAA, with a perfused segment measuring 23 mm in diameter and 58 mm in length. The aneurysm’s proximal segment exhibited significant stenosis due to thrombosis, beginning 21 mm distal to its origin from the aorta. Given the risk of rupture or ischemic complications, intervention was indicated. Open surgical repair was chosen over endovascular treatment because the aneurysm involved multiple SMA branches, making stenting unsafe due to the high ischemic risk.
The patient underwent open surgical repair via median laparotomy. The aneurysm was accessed through the mesentery and resected. Reconstruction was performed using a reversed saphenous vein graft harvested from the left thigh. Four branches, including the middle colic artery, right colic artery and the ileocolic artery, were anastomosed to the graft. After completing the anastomosis, strong Doppler signals were detected, and the bowel showed no signs of ischemia. Intraoperative findings confirmed an inflammatory aneurysmal wall (Fig. 2). Pathological examination revealed arterial tissue with aneurysmal wall changes, evidence of recanalization, focal myxoid degeneration, and fresh thrombus material. No malignancy was identified. Postoperative anticoagulation with Rivaroxaban was initiated due to the use of a venous graft for reconstruction.
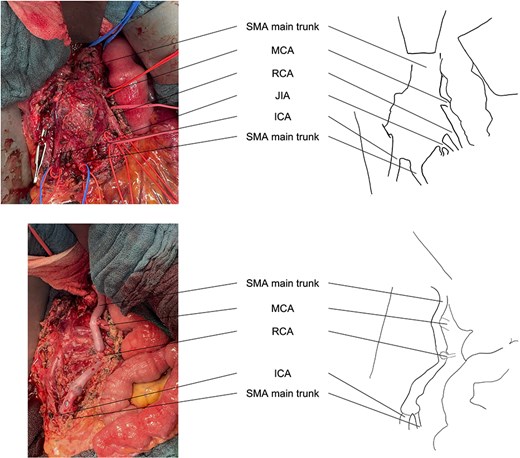
Intraoperative view before and after resection of the aneurysm and reconstruction of the SMA branches. ICA, ileocolic artery; JIA, jejunoileal artery; MCA, middle colic artery; RCA, right colic artery.
Postoperatively, the patient had an uneventful recovery. He was transferred from the intensive care unit to the regular ward on postoperative day 1 and discharged home on postoperative day 7 in stable condition. Follow-up at 5 weeks showed no evidence of complications and resolution of symptoms.
Discussion
The development of mesenteric artery aneurysms (MAAs) in SLE patients is a rare but significant clinical entity. Pathophysiological mechanisms may involve immune-mediated vasculitis, endothelial dysfunction, or thrombotic phenomena associated with antiphospholipid syndrome [3, 7]. The literature highlights few comparable cases, often associated with high morbidity due to delayed diagnosis or rupture [8].
Our case underscores the diagnostic challenges posed by nonspecific symptoms such as chronic abdominal pain, necessitating advanced imaging for definitive diagnosis. Nonspecific symptoms are common in SMAAs, further complicating diagnosis. CT angiography remains the gold standard for identifying the size, location, and thrombosis of MAAs [9].
Therapeutic strategies range from endovascular interventions to open surgical repair, with the choice largely determined by aneurysm complexity, involvement of critical branches, and the patient’s health status. While endovascular techniques are less invasive, they are often unsuitable for complex aneurysms involving multiple branches. Open surgical repair using autologous vein grafts provides durable, adaptable reconstruction, particularly in cases with inflammatory or thrombotic involvement of the aneurysmal wall [10, 11].
Our patient’s aneurysm exceeded the recommended threshold for surgery (2–2.5 cm) and was appropriately treated with open surgical repair [12–14]. However, debate remains on whether SMAAs should be treated regardless of size [1]. Using a reversed saphenous vein graft allowed successful revascularization of critical mesenteric branches, preventing bowel ischemia.
Compared to similar case reports, our patient’s rapid recovery reflects the efficacy of open surgical management. This approach remains the gold standard for complex aneurysms unsuitable for endovascular repair. The presence of an inflammatory aneurysmal wall aligns with findings linking SLE to vasculitic changes, reinforcing the need for vigilant monitoring and early intervention [1].
In conclusion, this case illustrates the rare occurrence of SMAA in a SLE patient and highlights the importance of open surgical repair. Further studies are needed to explore incidence, optimal timing for intervention, and long-term outcomes in this patient population.
Conflict of interest statement
None declared.
Funding
None declared.
References
- antiphospholipid syndrome
- thrombosis
- saphenous vein graft
- vasculitis
- aneurysm
- chronic abdominal pain
- systemic lupus erythematosus
- adult
- constriction, pathologic
- superior mesenteric artery
- mesentery
- rupture
- surgical procedures, operative
- tissue transplants
- diagnostic imaging
- aneurysm of superior mesenteric artery
- arterial aneurysm
- aneurysm of visceral artery
- giant intracranial aneurysm



