-
PDF
- Split View
-
Views
-
Cite
Cite
Yahya Alwatari, Lindsey Randall, Phillip G Rowse, Richard C Daly, Arman Arghami, Navigating missing link: innovative cannulation for robotic mitral valve repair in the absence of inferior vena cava, Journal of Surgical Case Reports, Volume 2024, Issue 3, March 2024, rjae172, https://doi.org/10.1093/jscr/rjae172
Close - Share Icon Share
Abstract
Cardiac surgeries often require the use of cardiopulmonary bypass to allow visualization and manipulation of tissues. Vascular anomalies may impose challenges with access configuration. A patient was evaluated for robot-assisted mitral valve repair and found to have an atretic inferior vena cava secondary due to chronic occlusion. The patient was cannulated arterially through the left common femoral artery, and two cannulation sites were applied for venous drainage: the right intrajugular vein and a second percutaneous access site directly into the right atrium through the chest wall. The procedure was completed without immediate complications, and the patient’s perioperative course was unremarkable.
Introduction
The mitral valve (MV) is the most commonly repaired cardiac valve, and the techniques used to perform such modifications have advanced significantly in recent years [1]. The use of annuloplasty ring implantations, leaflet resection/remodeling, neochordae implantation, and edge-to-edge repairs have improved intervention longevity and minimally invasive and robot approaches have been reported to have shorter hospital stays and lower short-term morbidity than open techniques [2].
Minimally invasive approaches rely on peripheral vascular cannulation for cardiopulmonary bypass; as a result, patients with atypical vasculature may necessitate less standard cannulation configurations [3]. Here we present a case of a patient with an untenable inferior vena cava (IVC) necessitating direct right atrial cannulation during robotic MV repair.
Case report
A 30-year-old male presented to medical attention with left lower extremity swelling and leg pain in October 2022 following a long flight. A thorough workup was consistent with extensive deep vein thrombosis (DVT)s in the left external, internal, and common iliac veins, and the thrombus continued into a large left lumbar collateral. The IVC and the high left common iliac vein were atretic (Fig. 1) with large, tortuous collaterals. He was also found to have severe mitral regurgitation. The patient was treated with mechanical thrombectomy and started on Apixaban. The patient underwent stent-grafting of his retroperitoneal collaterals to the level of the left external iliac artery. These stents were compromised (compressed or occluded) by the time the patient was evaluated for his MV repair in October 2023 when he was symptomatic and optimized for repair. Pre-op echocardiogram (ECHO) demonstrated bi-leaflet MV prolapse with myxomatous changes and severe MV regurgitation. A robotic approach was deemed appropriate for his case.
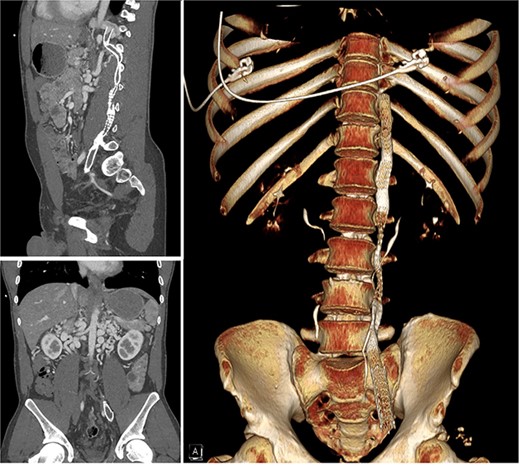
Computer tomography scan showing chronic occlusion of the infra-hepatic IVC and common iliac veins.
Standard right femoral artery cannulation and right chest robotic ports placement were performed intraoperatively. Right internal jugular vein 22 French venous cannula was placed (compared with 16 Fr we usually utilize). A larger venous cannula was employed given the fact that the lower body venous return was draining into the superior vena cava (SVC) via the azygous vein, and the right atrium was directly cannulated (Fig. 2). The patient had a very large azygous vein that was visibly draining into the SVC (Supplemental Figure 1). Partial cardiopulmonary bypass was commenced and a pursestring was then placed around the right atrial appendage. Via the third intercostal space anteriorly, a 32 French bullet tip venous cannula initially guided with a chest tube was brought into the chest and placed in the right atrium via the right atrial appendage (Fig. 3a, Supplemental Video 1).
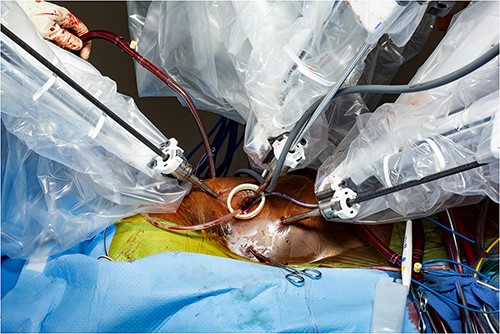
Intraoperative picture showing a 32 French bullet tip venous cannula placement in the right atrium via the right atrial appendage.
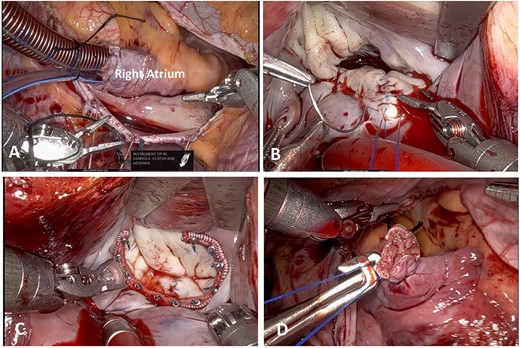
(A) Atrial Venous Cannulation. (B) Mitral Valve Repair. (C) Ring Annuloplasty. (D) Atrial Cannulation Site.
Cardiopulmonary bypass was augmented, the patient was cooled to 34° and cardioplegia was administered. The left atrium was opened parallel to the interatrial groove and the retractor was positioned. The prolapsed segment of the middle scallop of the posterior leaflet (P2) was excised in a triangular fashion. The leaflet was reconstructed with two layers of running 4–0 Prolene (Fig. 3b). A posterior mitral annuloplasty was performed with a 36 Medtronic SimuPlus Band (Fig. 3c). The valve was tested and was competent. Cardiopulmonary bypass was discontinued in a standard fashion with satisfactory hemodynamics. Transesophageal echocardiographic evaluation demonstrated trivial mitral regurgitation with a gradient of 4 mm, no systolic anterior motion (SAM), and no regional wall motion abnormalities. The right atrial cannula was removed (Fig. 3d), the snare was tied down, and the cannula was removed from the right internal jugular vein. Completion of the case was uncomplicated.
The patient’s perioperative course was uneventful, and a post-op echocardiogram revealed preserved left ventricular ejection fraction (LVEF). MV diastolic mean Doppler gradient was estimated at 1 mmHg, with trivial MV regurgitation. Post-op electrocardiography demonstrated normal sinus rhythm. The patient was discharged on post-operative Day 3 having achieved adequate pain control and demonstrating cardiopulmonary stability. He was discharged to home on metoprolol, Aspirin, and Eliquis. He completed cardiac rehabilitation following surgery and was able to resume work within 2 weeks of surgery.
Discussion
Cardiopulmonary bypass cannulation strategies in cardiac surgery are highly variable and can be modified to fit the needs of the patient and the surgical intervention to be undertaken. Peripheral femoral cannulation to the femoral vein and common femoral artery is the standard approach for robotic MV surgery [4]. The standard cannulation of the femoral vein requires a vessel with adequate patency and continuity with the IVC and right atrium. In situations where that confluence is compromised, such as in the case of IVC agenesis, femoral vein cannulation would be inappropriate. IVC agenesis is an underreported and relatively rare finding with some studies estimating occurrence in up to 1% of the population [5]. Most patients with a congenitally absent or underdeveloped IVC are asymptomatic due to robust collateral vascular support, and the diagnosis is often incidental.
While some publications have documented cannulation of alternative IVC vessels (e.g. azygous vein) in pediatric cases or dual cannulation in the face of a congenitally absent IVC [6, 7], to our knowledge, no other studies have documented the placement of direct atrial cannulation during robotic MV replacement for adequately draining venous circulation in a patient with a non-functional IVC. The successful results of this technique supports the use of such an approach in patients with vascular variations that might otherwise compromise drainage into the right heart.
The alternate cannulation arrangement required for a successful minimally invasive intervention required greater pre-operative planning and an understanding of the intraoperative hemodynamic demands of a collateral-heavy vascular circuit; however, the favorable outcomes of minimally invasive surgery compared with open are well documented, and we were glad to spare the patient a more invasive procedure and likely longer recovery. We were able to recently successfully perform atrial cannulation for another robotic-assisted MV repair in a 72-year-old patient with severe IVC stenosis with uncomplicated repair.
The decision to employ additional cannulation sites in patients with unique physiologic demands that might render the traditional dual cannulation approach insufficient requires adequate imaging, pre-operative planning, and sound clinical judgment. This case profile represents a demonstration of the utility, safety, and efficacy of novel cannulation techniques to meet the cardiovascular needs of surgery patients requiring cardiopulmonary bypass for MV repair.
Conflict of interest statement
None declared.
Funding
None declared.



