-
PDF
- Split View
-
Views
-
Cite
Cite
Kevin C Brown, Gabriela Esnaola, Dan Boffa, John M Morton, Gastroesophageal junction adenocarcinoma 1-year after sleeve gastrectomy, Journal of Surgical Case Reports, Volume 2023, Issue 9, September 2023, rjad518, https://doi.org/10.1093/jscr/rjad518
Close - Share Icon Share
Abstract
Gastroesophageal malignancy after sleeve gastrectomy is rare. A 70-year-old male with a BMI of 46 underwent laparoscopic sleeve gastrectomy with normal endoscopy. By 10 months postop, the patient had reduced BMI to 30.5. Eleven months postop, he presented with emesis and endoscopy showed severe stenosis at the gastroesophageal junction with EUS showing a circumferential mass. Patient had adenocarcinoma of the distal esophagus HER 3+ and MMR proficient, clinical T2N1. He underwent esophageal stent placement followed by FOLFOX switched to carboplatin-Taxol with radiation therapy complicated by a localized perforation requiring antibiotics. After PET scan of esophageal mass indicated response, he underwent an open distal esophagectomy, total gastrectomy with Roux-en-Y esophagojejunostomy, and placement of feeding tube. Pathology revealed poorly differentiated invasive adenocarcinoma with negative margins. In the USA, this represents only the second adenocarcinoma following a sleeve gastrectomy and the first in a non-immune compromised patient.
Introduction
Obesity remains one of the foremost health problems globally, with worldwide prevalence nearly tripling over the past four decades [1]. Obesity has been associated with a significantly increased risk for gastroesophageal reflux disease (GERD), erosive esophagitis, and gastric and esophageal adenocarcinoma [2, 3]. It is estimated that 14% of all cancer deaths in men and 20% of all cancer deaths in women can be attributed to overweight and obesity [3]. Weight loss can alleviate several obesity-related symptoms, and evidence shows that significant weight reduction reduces the risk of cancer-related death [4–6]. Bariatric interventions have proven to be a highly effective treatment option for long-term weight loss in obese patients, with laparoscopic Roux-en-Y gastric bypass and laparoscopic sleeve gastrectomy (LSG) being the most performed bariatric surgical procedures worldwide. Few studies have reported esophagogastric cancers following bariatric surgery [7–9]. Furthermore, there have only been five reported cases of GE cancer within a year of gastric bypass surgery, two of which were diagnosed via postoperative histological examination. We present only the second case of GE cancer within a year in the USA in a non-immune compromised patient after sleeve gastrectomy.
Case report
A 70-year-old Caucasian male with a body mass index (BMI) of 46 presented for evaluation of bariatric surgery. Medical history included atrial fibrillation, hypertension, diabetes mellitus, and obstructive sleep apnea. After metabolic center evaluation and preparation, he underwent an uncomplicated LSG with a normal intra-operative endoscopy without stricture, lesion, and patent sleeve. He demonstrated expected postoperative weight loss with a reduction in BMI to 30.5 at 10 months, remission of his diabetes, and improved hypertension control.
At 11 months postoperative, he presented to clinic with intermittent emesis and nausea to solids. A CT scan demonstrated distal esophageal reflux with some dilation. He underwent unsuccessful medical treatment for suspected GERD and subsequently underwent upper GI fluoroscopy demonstrating a tight GE junction stricture, which prompted endoscopic evaluation (Figs 1 and 2). Endoscopic findings were notable for a severe narrowing at the GE junction with inability to pass with a standard scope (10 mm diameter). Endoscopic ultrasound noted a circumferential mass and an enlarged lymph node, which were biopsied and found positive for adenocarcinoma with invasion into the muscularis propria. Final staging revealed distal esophagogastric adenocarcinoma human epidermal growth factor receptor (HER) 3+ and mismatch repair (MMR) proficient, clinical T2N1.
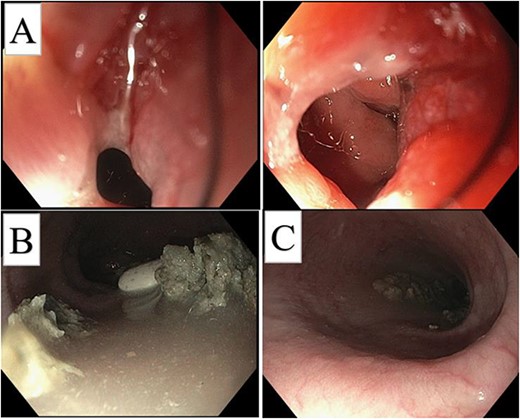
Initial post-op endoscopy, GE circumferential mass (A), esophageal dilation (C), with retained material (B).
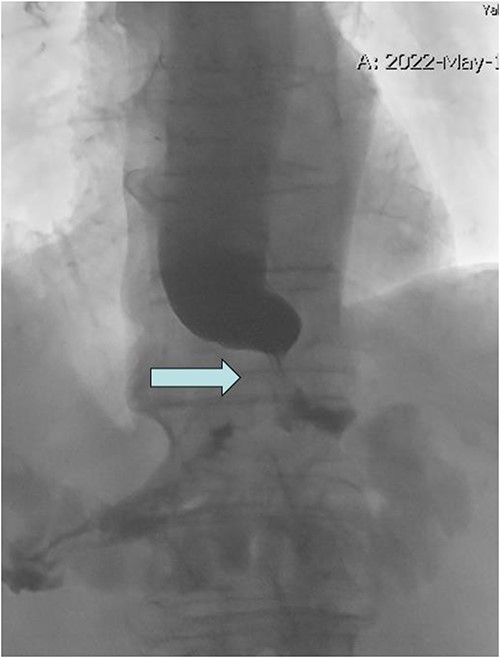
Upper GI fluoroscopy with tight GE junction stricture (arrow) and holdup of contrast.
After a multidisciplinary discussion, the decision was made to proceed with neoadjuvant chemotherapy with folinic acid (leucovorin), fluorouracil (5-FU), and oxaliplatin (Eloxatin) (FOLFOX) and radiation therapy. A positron emission tomography (PET)-CT scan was obtained prior to therapy to monitor treatment response. Due to his symptomatic stenosis, an endoscopic stent was placed to assist with nutritional intake throughout the preoperative phase. Over the first month, FOLFOX was poorly tolerated, and he was switched to carboplatin-Taxol along with radiation therapy. Although tolerated, treatment was complicated by a contained perforation at the site of the mass 3 months into therapy. This was managed conservatively with antibiotics and parenteral nutrition. Through this management, a repeat PET-CT was obtained, which demonstrated good response to his initial therapy with decrease avidity at the GE junction (Fig. 3). Without any extra-regional spread and improvement in primary mass, the decision was made to proceed with operative resection.
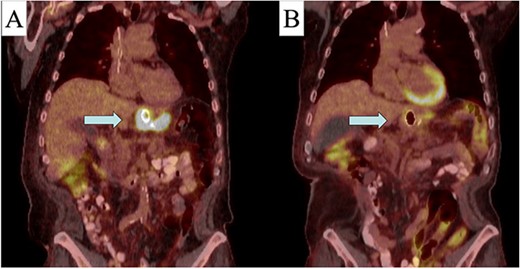
Pre (A) and post (B) initial treatment PET-CT with response to neo-adjuvant therapy (area indicated by arrow).
He underwent an open distal esophagectomy, total gastrectomy with Roux-en-Y esophagojejunostomy, and placement of feeding tube. Pathology revealed poorly differentiated invasive adenocarcinoma of the GE junction and cardia with negative margins. His nadir weight during treatment was 24.3.
At the time of this report, the patient is 13 months after initial diagnosis and 24 months after initial sleeve gastrectomy. BMI is currently 30.5 with good appetite, and there is no evidence of recurrent disease on cross-sectional imaging. Maintenance adjuvant therapy with Nivolumab is planned for a year after resection.
Discussion
In the USA, this represents only the second adenocarcinoma following a sleeve gastrectomy and the first in a non-immune compromised patient. There are limited data on the risks of cancer development in patients that have undergone recent bariatric surgery. Similar cases of GE cancer have been detected at more advanced stages of disease, often because patients present with nonspecific upper GI symptoms related to the procedure, such as epigastric pain, dysphagia, weight loss, and nausea and vomiting. This delayed detection often results in poorer prognosis and increased mortality.
A recent systematic review documented 324 global cases of esophageal and gastric cancers following bariatric surgery. Fifty-two studies were compiled, and results demonstrated that average time to diagnosis post-bariatric surgery was 62.95 months. A malignancy presenting within a year following metabolic surgery brings into question proper surveillance time postoperatively.
The value of upper GI endoscopy before bariatric surgery remains clear [10, 11]; given the high rates of abnormal findings, the International Federation for the Surgery of Obesity and Metabolic Disorders has recommended that esophagogastroduodenoscopies (EGD) be considered for all patients planning to undergo bariatric surgery. They also recommend that EGD be performed routinely for all patients that have undergone LSG or one-anastomosis gastric bypass at 1 year with follow-up every 2–3 years to facilitate detection of Barrett’s esophagus or upper GI malignancies until additional data are available. In brief, malignancies following bariatric surgeries are a rare occurrence, but prompt an educated discussion on the use of pre- and postoperative surveillance endoscopy, as well as the utility of diagnostic workup for any abnormal upper GI symptoms after these operations.
Conflict of interest statement
None declared.
Funding
None declared.



