-
PDF
- Split View
-
Views
-
Cite
Cite
Paola Solis-Pazmino, Luis Figueroa, Eduardo Andres Pilatuña, Carla Rocha, Richard Godoy, Cristhian García, Surgical approach for thyroglossal duct cyst cancer: a case report of challenging conventional thinking, Journal of Surgical Case Reports, Volume 2023, Issue 8, August 2023, rjad448, https://doi.org/10.1093/jscr/rjad448
Close - Share Icon Share
Abstract
This article reports an older woman with a 2-year history of enlarging submental neck mass. Head and neck imaging displayed a group with a solid central component. Additionally, a fine-needle aspiration suggested neoplasia. Thyroglossal duct cyst cancer is sporadic, accounting for ˂1% of all thyroid malignancies. A Sistrunk procedure was followed by a total thyroidectomy and unilateral neck dissections. From histological and immunohistochemical examinations, the incidental finding of carcinoma indicates the diagnosis of thyroid tissue thyroglossal duct cyst carcinoma, as it is the most common site for malignancy in ectopic thyroid tissue. The patient had an excellent recovery without additional treatments after surgery.
INTRODUCTION
Thyroglossal duct remnants form during the embryologic development of the thyroid gland. In rare cases, carcinoma is ~1% in these thyroglossal duct cysts, most commonly papillary thyroid carcinoma. The exact cause still needs to be fully understood. However, it is believed to arise from the transformation of the lining cells of the cyst into cancerous cells [1].
Treatment for thyroglossal duct cyst cancer usually involves surgical removal of the cyst. Sistrunk procedure (SP) is the main form of treatment, and optimal patient management includes multidisciplinary consultation to enhance survival. The extent of surgery depends on the size of the tumor, the involvement of nearby structures and the presence of metastasis (spread) to other parts of the body [2]. After surgery, radioactive iodine therapy may be administered to eliminate any remaining thyroid tissue and cancer cells. This article aims to report an older patient diagnosed and managed with thyroglossal duct cyst cancer.
CASE REPORT
We present a case of an older woman diagnosed with thyroglossal duct cyst cancer. The patient underwent a medical consultation due to a sizeable submental mass gradually enlarging for ~2 years. She had symptoms of dysphagia but denied odynophagia, hoarseness or shortness of breath. The patient had high blood pressure and a penicillin allergy but was otherwise healthy without a family history of carcinoma.
Physical examination revealed a sizeable submental mass (4.5 × 4 × 3.5 cm) that was hard, with smooth edges and did not move with deglutition. There was no extension into the oral cavity on bimanual palpation or palpable masses/nodules in the cervical region. There was a thyroid gland enlargement (right lobe 5 × 3 × 1.5, left lobe 4.5 × 2 × 1.2 cm). An ultrasound demonstrated enlarged cervical lymph nodes.
The ultrasound (Fig. 1) showed a soft tissue mass (13.7 ml) with multiple small calcific densities at the submandibular level with thick septations separating the locules and arterial vascularity of the solid component. A neck ultrasound demonstrated a bilateral thyroid enlarged (right lobe 5 × 3 × 1.5, left lobe 4.5 × 2 × 1.2 cm). There were also enlarged cervical lymph nodes in levels III (right) and IV (left). The fine-needle aspiration (FNA) was performed on the suspected thyroglossal duct. The biopsy suggested metastasis of thyroid cancer (TC).
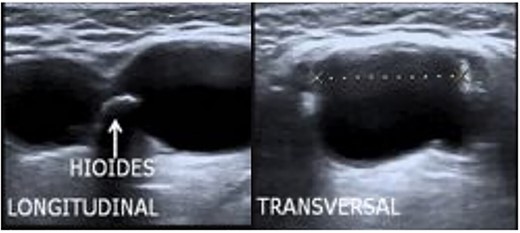
Treatment
The patient underwent an elective surgical procedure. SP and total thyroidectomy (TT) with lateral neck dissection were performed. The patient had an unremarkable recovery period and was discharged with thyroid hormone replacement and oral calcium supplementation the day after surgery. The patient did not need further treatments after the surgery.
The pathology reported a Thyroglossal duct cyst cancer.
Differential diagnosis
The first differential diagnosis was a metastasis of primary TC since the vascular invasion was detected (Fig. 2a). Surprisingly, the gross and microscopic examinations of the thyroid gland revealed no malignancy. Because of the location, and solid and trabecular morphology, a paraganglioma was considered (Fig. 2b). These tumors are neural crest-derived and stain-positive for the immunohistochemical markers: Synaptophysin and Chromogranin, which was not the case in our patient (Fig. 2c). Another neoplasia with similar morphology is the hyalinizing trabecular tumor that has a characteristic and distinctive cell membrane expression pattern with the immunohistochemical marker Ki-67 (MIB-1); this marker was present but showed nuclear expression (Fig. 2d). Finally, the histopathological diagnosis of a benign thyroglossal duct cyst requires evidence of respiratory/squamous epithelial lining and thyroid follicles; neither feature was found in our case. Nevertheless, the immunohistochemical markers thyroid transcription factor 1 (TTF-1) and CK7 were consistent with thyroid carcinoma and demonstrated nuclear and cytoplasmic staining, respectively (Fig. 2e, f).
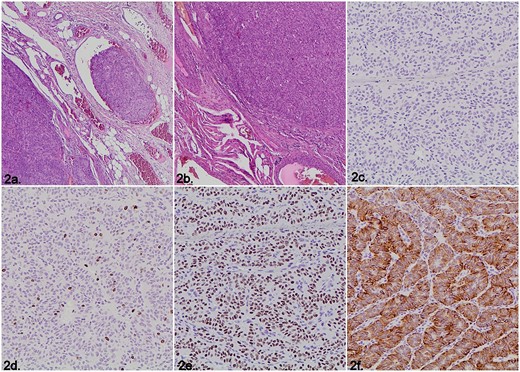
Microscopic images of the histopathological examination. (a) Vascular invasion (Hematoxylin &eosin staining). (b) Solid and trabecular tumor growth pattern (Hematoxylin & eosin staining). (c) Negative immunohistochemistry staining for Synaptophysin. (d) Nuclear instead of membrane immunohistochemistry expression for Ki-67 (MIB-1). (e) Nuclear positivity for the immunohistochemical marker TTF-1. (f) Cytoplasmic staining for the immunohistochemistry marker CK7.
DISCUSSION
Thyroglossal duct cysts carcinoma (TGDCC) is rare, occurring in 0.7–1.5% of cases [2]. Its diagnosis and management require an interdisciplinary approach, starting with clinical suspicion, imaging studies and Sistrunk surgery, followed by pathological examination, including immunohistochemical markers, to diagnose accurately.
The most common histological subtype is papillary carcinoma (75–85%). The mean age at diagnosis of TGDCC is ~40 years old, most common in women [3]. The incidence of associated symptoms is low, with 1.8% of patients endorsing a history of pain and 3.1% stating that they had dysphagia. A rapid increase in the mass size or a solid and palpable mass can suggest malignancy. Our elderly patient had an enlarging group causing dysphagia for 2 years.
Although Sistrunk surgery is accepted, performing a TT for TGDCC is controversial. Evidence suggests that it should be reserved for patients older than 45, like the patient described in this case. TT is preferably performed based on the fine needle aspiration biopsy result (high-risk malignant) or if both thyroid lobes do not consist of healthy tissue anymore. In this case, the patient underwent TT due to the FNA reporting a metastasis of TC.
Lateral neck dissection should be performed in patients if the FNA results suggest metastasis from the thyroid with positive nodes or if the location of interest displays aggressive features, such as large tumors and invasion of local structures [4]. Some studies reported an incidence ranging from 10% to 15% of patients with positive lateral neck nodes. We initially thought that the large mass of our patient was related to thyroid carcinoma. Thus, we decided to perform a TT and lateral neck dissection while accounting for her potential need for additional treatments, such as radioactive iodine (RAI) ablation. However, some studies suggest that RAI is appropriate for patients with large tumors, lymph node involvement or malignancy present in both the thyroid and TGDC [5]. Our patient had TGDCC and goiter; therefore, she did not need RAI ablation.
Since TGDCC is a rare malignancy, a broad differential diagnosis should be considered: metastasis from an unknown primary, paraganglioma, hyalinizing trabecular tumor and thyroglossal duct cyst, among others. Clinicopathological correlation and the use of FNA and immunohistochemical markers improve the accuracy of diagnosis and aid in determining the best treatment for the patient. In this case, the result of the FNA was inaccurate, and the diagnosis of TGDCC was incidental after the surgery.
The overall prognosis in TGDCC is excellent, with a survival rate of 99.4% and a recurrence rate of ˂5% [6]. Long-term follow-up measures, such as annual clinical examinations, ultrasonographic imaging and monitoring serum thyroid stimulating hormone (TSH) and thyroglobulin levels, have been recommended for all appropriate patients.
CONCLUSION
Considering the presented findings in the context of the patient’s symptoms and medical history is crucial. Given the size of the submental mass, the presence of dysphagia and the biopsy suggesting metastasis of TC, further evaluation and management are necessary. Treatment options may include surgical removal of the thyroglossal duct cyst, affected thyroid tissue and involved lymph nodes. Additional therapeutic approaches, such as radioactive iodine therapy or external beam radiation, may be considered based on the extent and aggressiveness of the cancer.
CONFLICT OF INTEREST STATEMENT
The authors declare that there is no conflict of interest.
FUNDING
The authors declare that no funds, grants or other support were received during the preparation of this manuscript.
AUTHORS' CONTRIBUTIONS
Paola Solis-Pazmino and Cristhian Garcia contributed to the study’s conception and design. Paola Solis-Pazmino, Luis Figueroa, Eduardo Pilatuña, Carla Rocha and Richard Godoy performed material preparation, data collection and analysis. Paola Solis-Pazmino and Cristhian Garcia wrote the first draft of the manuscript, and all authors commented on previous versions.
All authors read and approved the final manuscript version to be published.
ETHICAL STANDARDS
All procedures followed were by the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
INFORMED CONSENT
Informed consent was obtained from the patient to be included in the study.
CONSENT TO PARTICIPATE
Informed consent was obtained from the patient.
References
Alatsakis M, Drogouti M, Tsompanidou C, Katsourakis A, Chatzis I.



