-
PDF
- Split View
-
Views
-
Cite
Cite
Aroosha Safari, Saud Hamza, David Paton, A rare case: synchronous ipsilateral breast implant-associated anaplastic large cell lymphoma and invasive ductal carcinoma, Journal of Surgical Case Reports, Volume 2023, Issue 6, June 2023, rjad338, https://doi.org/10.1093/jscr/rjad338
Close - Share Icon Share
Abstract
A 78-year-old female with a history of cosmetic breast implants presented with unilateral breast enlargement and was subsequently diagnosed with stage IA breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) as well as stage IB ipsilateral synchronous invasive ductal carcinoma (IDC). Her assessment included bilateral breast ultrasounds,mammograms and MRIs with right-sided fine needle aspiration of peri-implant fluid, core biopsy of right breast mass and a whole-body positron emission tomography scan. She was surgically treated with bilateral capsulectomy, implant removal and mastectomy. No adjuvant treatment was required for the BIA-ALCL. The IDC required adjuvant chemotherapy, radiotherapy and endocrine therapy. This rare case highlights the paramount importance of thorough evaluation of suspected BIA-ALCL patients for synchronous breast pathologies. We conclude with a concise summary of the salient points on evaluation and management of BIA-ALCL for surgeons.
INTRODUCTION
Breast implant-associated anaplastic large cell lymphoma (BIA-ALCL) is a rare type of T cell lymphoma associated with textured breast implants used for breast reconstruction and augmentation. About, 1.5 million prostheses are implanted annually worldwide. BIA-ALCL’s incidence is quoted from 1 in 4000 to 1 in 30 000 women with implants per year [1].
The first published case was in 1997. In 2016, the World Health Organization classified it as a distinct clinical diagnosis, separate from other categories of ALCL [2].
We present a rare case of synchronous ipsilateral invasive ductal carcinoma and BIA-ALCL. Based on our literature search, this will be the first published case where both entities were diagnosed concurrently. The only other published case was in 2019, in a patient where ipsilateral IDC was diagnosed 6 months after BIA-ALCL [3]. Thereafter, we discuss salient BIA-ALCL facts for surgeons.
CASE REPORT
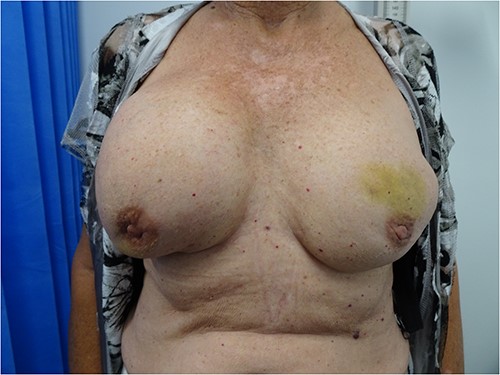
A 78-year-old female presented with an enlarged right breast. She underwent bilateral augmentation mammoplasty 14 years prior with subpectoral Allergan implants. Her clinical history was otherwise insignificant except a family history of unilateral breast cancer after the age of 50 in her maternal aunt. On examination, the right breast was larger than the left with palpable peri-implant fluid and no palpable lymphadenopathy (Fig. 1).
She underwent bilateral mammograms and ultrasounds of breasts and axillae (Figs 2, 3A and 3B). Within the right breast, the mammogram demonstrated a suspicious mass with two calcified foci that correlated to a 19 × 15 × 6-mm hypoechoic lesion at the 9/1 o’clock position on ultrasound. Core biopsy confirmed this to be a Grade 2 invasive ductal cancer (IDC), which was ER/PR positive and HER2 negative.
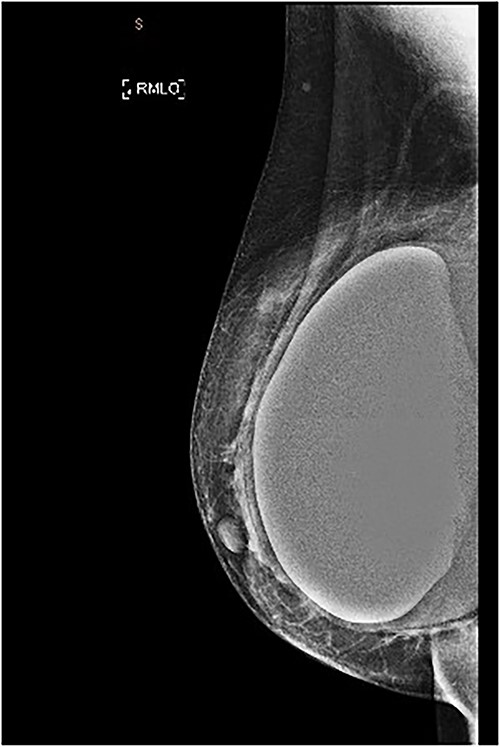
MMG of the right breast in MLO view showing a lesion in the upper breast.
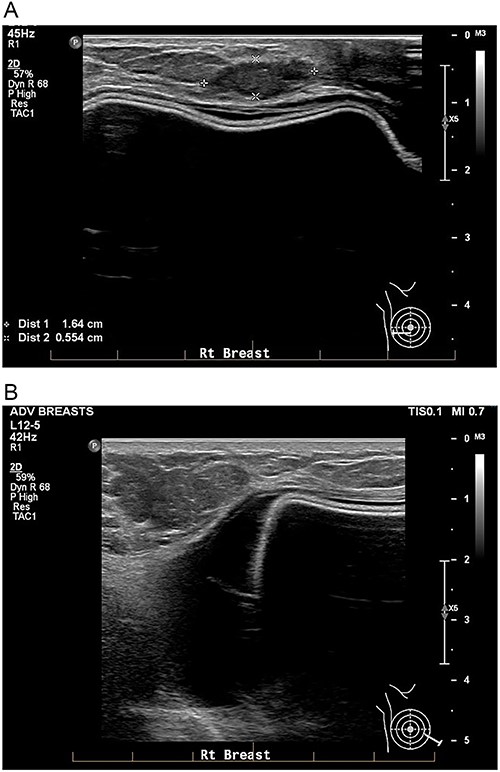
A: USS of the right breast showing a mass at the 9 o’clock position. B: USS of the right breast showing a peri-implant effusion.
Bilateral breast MRI demonstrated intact implants, the known right-sided IDC and a right-sided septated collection of peri-implant fluid (Fig. 4). Aspiration of 20 mls of turbid fluid did not show evidence of an atypical or aberrant lymphoid population by cytology or flow cytometry.
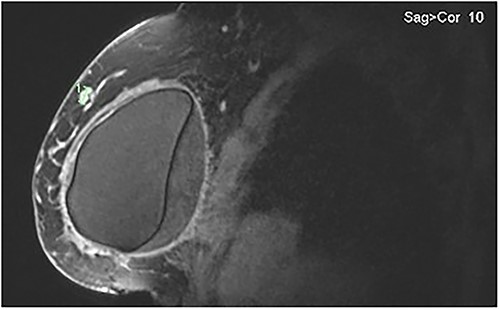
MRI of the right breast demonstrating peri-implant effusion and the confirmed IDC in the upper outer quadrant at the 9 o’clock position.
The FDG PET-CT did not show any evidence of regional or distant metastasis. After extensive counselling, the patient opted for a bilateral mastectomy, implant removal and capsulectomy, with a right sentinel lymph node biopsy (SLNB). Intraoperatively, a right-sided capsule enlarged by an effusion was noted (Fig. 5). Due to failure of the dual localization, the SLNB progressed to level I axillary dissection.
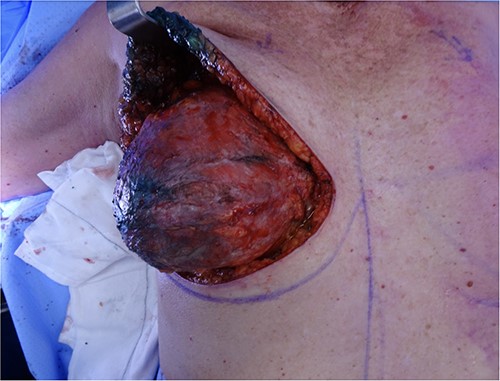
Histology of right breast tissue confirmed a grade 2, ER/PR positive and HER 2 negative 11 mm IDC with associated 20 mm of intermediate grade DCIS, with clear margins. In total, 2 of 14 axillary lymph nodes were involved by carcinoma, with extracapsular extension (T1N1M0/Stage IB).
Histology of the right breast capsule showed diffuse thickening with an eosinophil-rich inflammatory infiltrate (Figs 6A and 6B). Microscopic aggregates of atypical lymphoid cells were noted in the lining fibrin (Fig. 6C). These showed strong CD30 positivity (Fig. 6D) without ALK expression. Atypical lymphoid cells were also noted cytologically in the implant fluid (Fig. 7), with an aberrant CD30+ immunophenotype demonstrated by flow cytometry. These findings were consistent with BIA-ALCL (Stage IA). Histology of left breast and capsule was unremarkable. Her recovery was complicated by a non-infected seroma that responded well to ultrasound-guided drainage.
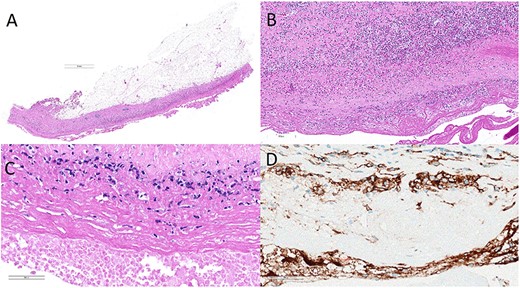
Histology of right breast capsule (A–C) showing an inflammatory infiltrate with aggregates of atypical lymphoid cells with strong CD30 positivity on immunohistochemistry (D).
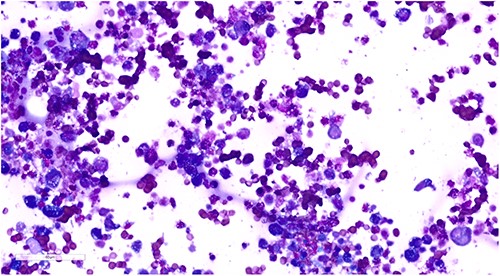
Cytology of intraoperative peri-implant fluid demonstrating atypical lymphoid cells.
Following a multidisciplinary discussion, she was reviewed by hematology and oncology teams. Her stage IA fully resected BIA-ALCL required no additional treatment. Her IDC (stage IB) is being treated with adjuvant chemotherapy, radiotherapy and endocrine therapy.
DISCUSSION
BIA-ALCL can present 2–32 years after insertion of textured implants [1, 4]. Majority present with a peri-implant effusion and one-third present with a capsular mass. Less common symptoms include erythema, pain, capsular contraction, axillary lymphadenopathy and B-type symptoms [5, 6]. There are no guidelines promoting surveillance for BIA-ALCL, although the FDA recommends MRI surveillance to screen for occult implant ruptures [1].
Diagnostically, ultrasound has an 84% sensitivity and 75% specificity for detecting an effusion and 46% sensitivity and 100% specificity for detecting a mass in BIA-ALCL. If ultrasound is indeterminate, then MRI is recommended [7]. The capsule may drain to multiple lymph node basins, and thus, sentinel lymph node biopsy is not recommended [8].
Definitive diagnosis is via cytology, flow cytometry and immunohistochemistry of the peri-implant fluid or core biopsy of any capsular masses [8]. Hallmarks are large pleomorphic CD30+/ALK-epithelioid lymphocytes [9]. This is believed to be due to chronic inflammation elicited by the implants’ chronic biofilm, combined with mutations within the JAK/STAT pathways [5, 10, 11].
Staging requires PET-CT [5]. Although a lymphoma, BIA-ALCL behaves like a solid tumor and is staged using the TNM staging criteria (Table 1) [12, 13].
| Tumor extent (T) . | Lymph nodes (N) . | Metastasis (M) . |
|---|---|---|
| T1: Effusion only | ||
| T2: Early capsule invasion | N1: One regional lymph node | M0: No distant spread |
| T3: Mass, confined to capsule | N2: Multiple regional lymph nodes | M1: Spread to other organs/sites |
| T4: Mass, invading outside of capsule | ||
| STAGE | IA: T1 N0 M0, IB: T2 N0 M0, IC: T3 N0 M0 | |
| IIA: T4 N0 M0, IIB: T(1–3)N1 M0 | ||
| III: T4 N(1–2)M0 | ||
| IV: T(any) N(any)M1 | ||
| Tumor extent (T) | Lymph nodes (N) | Metastasis (M) |
|---|---|---|
| T1: Effusion only | ||
| T2: Early capsule invasion | N1: One regional lymph node | M0: No distant spread |
| T3: Mass, confined to capsule | N2: Multiple regional lymph nodes | M1: Spread to other organs/sites |
| T4: Mass, invading outside of capsule | ||
| STAGE | IA: T1 N0 M0, IB: T2 N0 M0, IC: T3 N0 M0 | |
| IIA: T4 N0 M0, IIB: T(1–3)N1 M0 | ||
| III: T4 N(1–2)M0 | ||
| IV: T(any) N(any)M1 | ||
Management is guided by stage and the National Comprehensive Cancer Network (NCCN) guidelines [7].
Stage I disease localized to the capsule, which can be completely resected, can usually be cured by surgery alone with total capsulectomy, implant removal and excision of capsular masses [7]. Excisional biopsy of any suspicious lymph node should also be performed [8]. About, 4.6% of cases will have lymphoma in contralateral breast, which is why bilateral capsulectomy and implant removal is recommended [13]. There is currently no evidence to support mastectomy [7].
In cases of incomplete resection, positive margins or unresectable disease, radiation therapy should be considered [7]. In patients with a tumor mass or with lymph node involvement or systemic spread, chemotherapy is indicated [14, 15]. Neoadjuvant chemotherapy before capsulectomy is also an option [15].
For patients in remission, it is recommended they are examined every 3–6 months, with CT or PET-CT every 6 months for 2 years [15].
Prognosis is stage dependent and difficult to calculate given the limited data on this new disease entity. A systematic review in 2017 found overall survival to range from 1 month to 20 years depending on stage [15].
In conclusion, although a mammogram is not in the recommended work up for BIA-ALCL, this case illustrates the importance of routine triple assessment in any patient presenting with any breast pathology, to ensure timely diagnosis of synchronous disease.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.
References
- positron-emission tomography
- radiation therapy
- breast mass
- adjuvant chemotherapy
- fine needle biopsy
- immunologic adjuvants
- pharmaceutical adjuvants
- infiltrating duct carcinoma
- ki-1+ anaplastic large cell lymphoma
- mammography
- breast
- breast implants
- mastectomy
- endocrine therapy
- breast hypertrophy
- core needle biopsy
- joint capsule excision
- implant removal
- implants
- breast implant–associated anaplastic large cell lymphoma



