-
PDF
- Split View
-
Views
-
Cite
Cite
Hafsa El Ouazzani, Mohammed Yassaad Oudghiri, Salma Abbas, Asmaa Regragui, Abdessamad Elouahabi, Fouad Zouaidia, Nadia Cherradi, Diagnostic challenge: primary leptomeningeal melanoma with melanomatosis, illustrative case report, Journal of Surgical Case Reports, Volume 2023, Issue 6, June 2023, rjad323, https://doi.org/10.1093/jscr/rjad323
Close - Share Icon Share
Abstract
Primary leptomeningeal melanoma is an extremely rare type of intracranial melanoma. It cannot be reliably distinguished from metastatic melanoma on neuroimaging and histopathological characteristics alone; its diagnosis is established only after exclusion of secondary metastatic disease from a cutaneous, mucosal or retinal primary. Prognosis is poor, partly due to its high rate of misdiagnosis. Herein, we report a case of a primary meningeal melanoma of the skull base with melanomatosis, in a 31-year-old man, mimicking meningioma. Our aim is to highlight the diagnostic pitfalls and to discuss the histopathological differential diagnoses, especially with other pigmented lesions of central nervous system.
INTRODUCTION
The CNS can be affected by a spectrum of melanocytic lesions, ranging from diffuse neurocutaneous melanosis to a focal and benign neoplasm (melanocytoma) and to an overtly malignant tumor (melanoma). Primary leptomeningeal melanoma is an extremely rare type of primary intracranial melanoma, with a global incidence of 1 case per 20 million individuals [1, 2]. It may be classified into two types based on the behavior of the tumor; one type invades the pia mater diffusely and spreads into the subarachnoid space, while the other causes nodular tumors [1].
Because of its rarity and aggressive behavior, it is often misdiagnosed and is associated with a therapeutic failure.
CASE REPORT
We report the case of 31-year-old man, with no past medical history and no extracranial abnormality. He was admitted to the neurosurgery department for the management of an intracranial skull base tumor suggestive of a meningioma. On admission, the patient was experiencing signs of increased intracranial hypertension, and he reported a rapid deterioration of visual acuity causing bilateral blindness. He had no motor deficit.
Magnetic resonance imaging (MRI) scan showed a skull base tumor that was hypointense on T1, hyperintense on T2, with regular and homogeneous contrast enhancement. The radiologist suggested an anterior skull base meningioma, although atypical for the age of the patient (Fig. 1). A control computed tomography (CT) scan—performed before admission—demonstrated a rapid growth of the tumor, almost reaching double the initial volume over a 2-month period, with perilesional edema (Fig. 1). This was clearly in favor of an aggressive behavior and questioning the suggested diagnosis of meningioma.
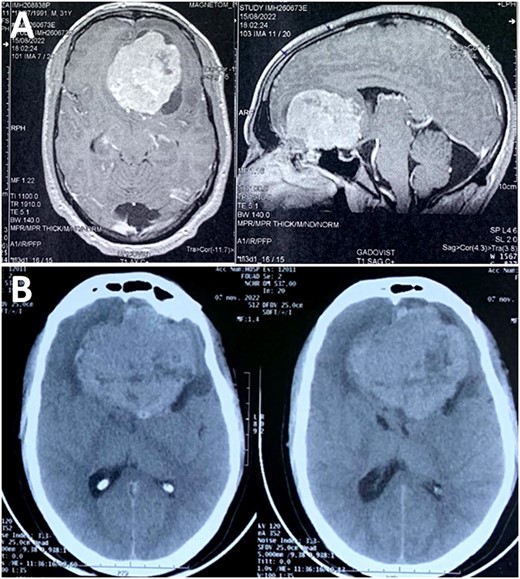
(A) MRI scan showing a skull base tumor that was hypointense on T1, and hyperintense on T2, with regular and homogeneous contrast enhancement; (B) CT scan demonstrated a rapid growth of the tumor, almost reaching double the initial volume over a 2-month period.
Surgical records reported a dark and highly vascular extra-axial tumor with a dural attachment to the left anterior skull base. The dura mater underlying the tumor surface was blackened (Fig. 2). The tumor extended throughout the subarachnoid space and invaded the cerebral parenchyma. In front of this strange aspect, the surgeon evoked a hemangioblastoma and could achieve a gross total resection with complete decompression of both optic nerves.
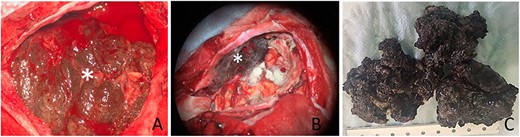
(A) Intra-operative appearance of the tumor (star); (B) aspect of dura mater after excision (star); (C) macroscopic appearance of the tumor after formalin fixation.
At pathological examination, the tumor was black, firm with irregular surface (Fig. 2C). Intra-operative smears revealed monomorphic and round tumor cells with basophilic characteristics. Some pigmented deposits were observed, but at this stage, the pathologist proposed the diagnosis of an undifferentiated round cell tumor (Fig. 3). Histologically—after formalin fixation—the tumor was composed of hypercellular sheets, nests and spindled cells associated with many melanic deposits. The tumor cells had a prominent nucleoli and a moderate amount of eosinophilic cytoplasm. We did not observe a significant nuclear pleomorphism or numerous atypical mitoses; however, we noted foci of tumor necrosis (Fig. 3).
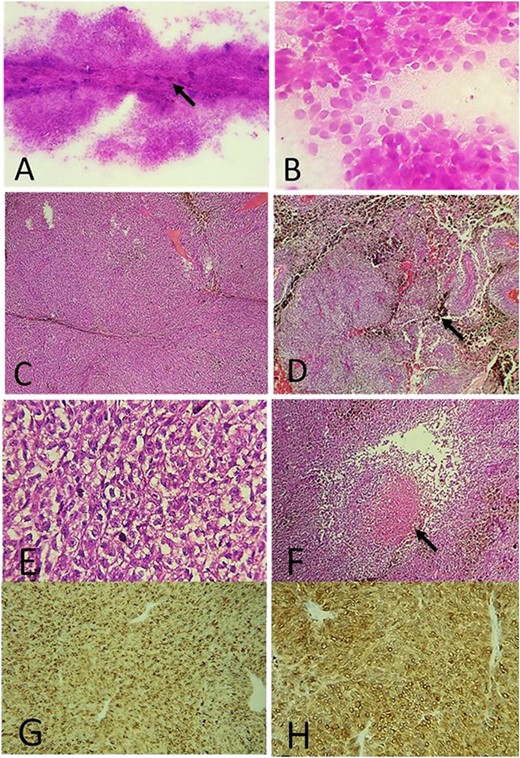
(A) Intra-operative cytological smear showing a hypercellular proliferation with pigmented deposits (arrow); (B) tumor cells were monomorphic, round and undifferentiated; (C) histologically, the tumor was composed of hypercellular sheets and spindled cells (H&E ×100); (D) many melanic deposits have been observed (arrow) (H&E ×100); (E) at high magnification, the tumor cells had an enlarged oval-shaped nuclei and prominent nucleoli (H&E ×400); (F) foci of necrosis within the tumor (arrow) (H&E ×200); on IHC: the tumor cells express HMB45 (G) and Melan A (H).
On immunohistochemistry (IHC), the neoplastic cells stained positively for melanoma antibody HMB45 and Melan A (Fig. 3), while they were negative for epithelial membrane antigen (EMA) and BRAF V600E. The Ki67 labeling index was 30% in the highest proliferating areas.
Thus, the diagnosis of meningeal melanoma with melanomatosis was made.
A diagnosis of primary meningeal melanoma was confirmed after careful dermatologic and ophthalmologic examination, which ruled out the presence of cutaneous or choroidal melanoma.
The post-operative course was serious. The patient died due to ischemic complications.
DISCUSSION
Melanocytic cells are normally found in the meninges of the central nervous system (CNS). They are numerous only on the ventral aspect of the lower part of the medulla oblongata, and a slight pigmentation is macroscopically visible in this region [2]. Diffuse or circumscribed primary leptomeningeal melanocytic neoplasms derived from meningeal melanocytic cells are rare [3].
According to the 2021 edition of the World Health Organization (WHO) classification of tumors of the CNS, diffuse lesions are characterized by the involvement of large expanses of the subarachnoid space with or without focal nodularity. Based on whether the lesion has benign or malignant histological phenotype, it is called meningeal melanocytosis or meningeal melanomatosis, respectively [4].
Circumscribed neoplasms may be designated as melanocytomas if they are benign or as melanomas if they are malignant. Intermediate-grade lesions are also designated as melanocytomas.
Usually, all meningeal melanocytic tumors strongly express PS100, Melan A, HMB45 and MITF [5].
On histology, meningeal melanoma is accompanied by marked cytological atypia, increased mitotic activity >1.5 mitoses/mm2 and often demonstrated unequivocal invasion of the CNS parenchyma or coagulative necrosis. It may be composed of pleomorphic spindled or epithelioid cells and display variable cytoplasmic melanin and numerous mitotic figures [5].
Other meningeal melanomas—such as our case—are highly cellular and less pleomorphic and usually consist of smaller, tightly packed spindle cells. In the absence of nuclear pleomorphism the distinction between melanoma and melanocytoma can be a challenge for the pathologist. Melanocytoma is a benign tumor and lack cytological atypia, necrosis and mitoses (on average <0.5 mitoses/mm2, equating to <1 mitosis/10 HPF of 0.5 mm in diameter and 0.2 mm2 in area). Melanocytoma do not show invasion of CNS parenchyma. In the cases where a histological melanocytoma shows CNS invasion or increased mitotic activity (0.5–1.5 mitoses/mm2, equating to 1–3 mitoses/10 HPF of 0.5 mm in diameter and 0.2 mm2 in area) and no necrosis, the tumor is defined as intermediate-grade melanocytoma [5]. Cases of malignant transformation of melanocytoma into melanoma are described in the literature but are rare and occur especially after recurrence [6, 7].
Through the WHO description, we deduce the value of tumor necrosis and mitotic activity to classify these tumors.
Another aspect of diagnostic difficulty encountered with meningeal melanoma is to confirm its primitive character and to distinguish it from other cerebral pigmented neoplasms.
Primary CNS melanoma is generally diagnosed following the exclusion of a primary cutaneous or mucosal/retinal melanoma, as differential histological diagnosis between primary and metastatic origins is often difficult [8, 9].
In addition of meningeal melanocytoma described above, the other differential diagnoses are represented by meningioma which can have similar neuroradiologic findings and may mimic cerebral melanoma. The differential diagnosis is more difficult in some cases of meningioma with cells containing melanin pigment; in this case, the immunohistochemical positivity of somatostatin receptor 2a, EMA and progesterone receptor is in favor of meningioma [3].
The distinction between meningeal melanoma and other melanotic tumors of the CNS such as melanotic schwannoma—which has a similar immuoprofile to that of melanoma—can be difficult [3]. Melanotic schwannoma is characterazed by the presence of psammoma bodies and adipose-like cells. It has cells with benign or mildly atypical cytology, and demonstrates evidence of pericellular basement membrane synthesis [10].
In some cases, mutation analysis (including for GNAQ, GNA11PLCB4 and CYSLTR2) and methylation profiling are useful for recognizing meningeal melanoma as primary CNS tumor and discriminating them from another pigmented CNS tumor. Because of the limited number of primary leptomeningeal melanocytic lesions analyzed, other mutations are less well characterized [11].
The survival for melanoma diagnosis with leptomeningeal involvement is poor. It is known that there are no specific guidelines for the management of both the primary CNS melanomas and leptomeningeal melanomatosis as they are rarely seen. However, radiotherapy, especially after surgical resection of a primary tumor, systemic chemotherapy, with agents such as dacarbazine, cisplatin, thalidomide or temozalamid, or intrathecal therapy, with methothraxate, cytarabine and sometimes immunmodulatory agents such as peginterferon alpha-2b, have all been used as treatments [12, 13].
The prognosis and life expectancy for patients with primary leptomeningeal melanoma associated with melanomatosis is worse despite treatment. Most authors quote a median survival of <1 year [14].
CONCLUSION
Meningeal melanomas has a poor prognosis, partly due to their high rate of misdiagnosis. This case highlights the diagnostic pitfalls encountered by the pathologist during the analysis of leptomeningeal melanocytic neoplasms. We insist on the importance of the histological interpretation to classify these tumors in the absence of clinical, radiological and immunohistochemical specificity.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
No external funding sources were obtained for this submission.
DATA AVAILABILITY
No new data were generated or analyzed in support of this research.
CONSENT FOR PUBLICATION
Written informed consent for publication of their clinical details and/or clinical images was obtained from the patient.



