-
PDF
- Split View
-
Views
-
Cite
Cite
Gjorgji Trajkovski, Vanja Trajkovska, Slavica Kostadinova-Kunovska, Ognen Kostovski, Radomir Gelevski, Andrej Nikolovski, Low-grade malignancy glomus tumor of the abdominal wall: a case report and literature review, Journal of Surgical Case Reports, Volume 2023, Issue 12, December 2023, rjad680, https://doi.org/10.1093/jscr/rjad680
Close - Share Icon Share
Abstract
Glomus tumors are painful neoplasms arising from the glomus body (responsible for body temperature and blood pressure regulation) in the skin. Although mostly present in the interdigital areas, glomus tumors can arise elsewhere. The vast majority of them are benign. Rarely, malignant variants are reported and are associated with the potential for local infiltration and metastatic dissemination. The abdominal wall location of glomus tumors is extremely rare regardless of whether they present as benign or malignant. We present a case of a 58-year-old female patient with a low-grade malignancy infiltrative glomus tumor of the abdominal wall.
Introduction
Glomus tumors arise from the glomus body located in the dermis or subcutis, responsible for blood pressure and temperature adjustment. They account for 2% of all soft-tissue tumors in the extremities but can arise elsewhere [1]. They mostly present as benign lesions, also called glomangiomas. The rarer malignant variant accounts for <1% of all soft-tissue tumors [2]. They are described to have predominant low-grade malignancy, but reports of aggressive glomangiosarcoma behavior thus leading to fatal metastases in up to 38% of cases are reported in the literature [3]. We present a case of a female patient with a palpable large and painful mass in the abdominal wall. Written informed consent was obtained from the patient.
Case report
A 58-year-old female patient (smoker) complained of abdominal wall pain and a palpable tumor, 3 months before presenting in the outpatient ward. Previously diagnosed comorbidities were hypertension, hypothyroidism, and Type II diabetes mellitus. Preoperative serum parameters were within the normal range, except for the serum glucose level (15.04 mmol/L). Contrast-enhanced abdominal computerized tomography revealed a tumor with expansive growth toward the subcutaneous tissue and the peritoneum, with origin from the left rectus abdominis muscle. The tumor was described as multinodular, lobulated, well-demarcated, relatively homogenous mass with dimensions 11 × 8.5 cm (Fig. 1). Surgical excision of the lesion with abdominal wall reconstruction was indicated. Intraoperatively, wide excision with partial resection of the involved abdominal wall muscles was performed (Fig. 2). Primary abdominal wall reconstruction with a nonabsorbable suture was performed (without the use of prosthetic mesh). Consecutive abdominal compartment syndrome did not develop. The postoperative period was uneventful and the patient was discharged on postoperative Day 5.
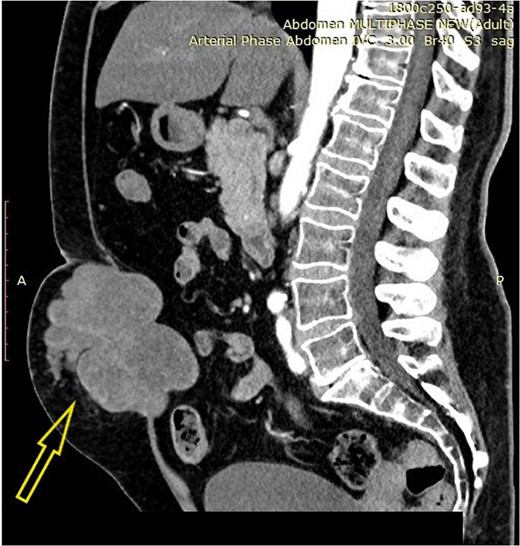
Contrast-enhanced abdominal computerized tomography with a tumor in the abdominal wall (arrow).
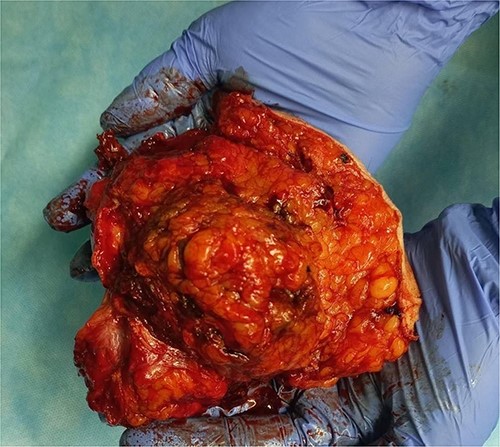
The macroscopic pathology report revealed the total dimension of the removed tissue to be 10.5 × 8.5 × 10.5 cm. However, the tumor itself had dimensions of 8 × 7.5 × 7 cm, surrounded by a pseudo capsule. The tumor was described to be multicystic, formed by loculi filled with hemorrhagic and partially with yellowish gelatinous myxoid content. In its periphery, yellowish tumor tissue was present (Fig. 3). Microscopic findings described a tumor made of myxoid stromal nodules and small capillaries concentrically surrounded by monomorphic round cells with mild nuclear pleomorphism. Large pseudo-angiomatous spaces filled with blood were seen in some of the nodules. Part of the nodules was hypocellular, made mostly by myxoid stroma and stellate cells. A thick fibrous pseudo capsule with numerous multifocal deposits of hemosiderin deposits was present peripherally. Tumor tissue infiltrates the abdominal wall muscles (Figs 4 and 5).

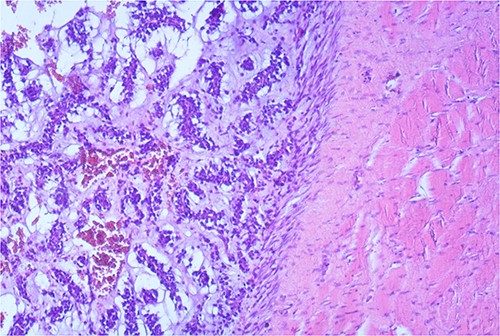
Small capillaries lined by relatively monomorphous round cells, with mild pleomorphism and sparse mitotic activity, surrounded by myxoid stroma (HeEo, ×100).
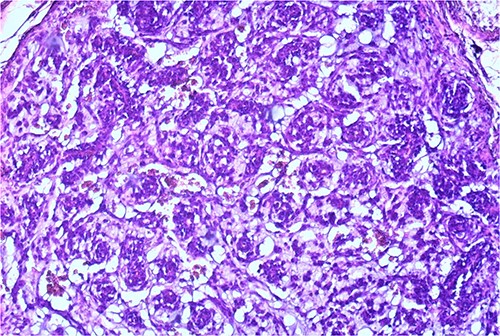
On immunohistochemical staining, tumor cells were positive on Vimentin, Smooth Muscle Actin (SMA), and focally on CD34. They stained negative on Desmin, Caldesmon, CD31, HMB 45, GFAP, CKAE1/AE3, CK7, EMA, S100, CD99, and p63. The tumor’s proliferation index after staining with Ki-67 was ~5% (Fig. 6a–c).
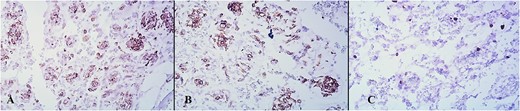
Discussion
Historically, Wood [4] first reported a glomus tumor with the description: “painful subcutaneous tubercle.” Malignant glomus tumors were initially reported by Lumley and Stansfeld [5]. The predominant localization of these tumors, no matter the malignant potential are the extremities (mostly digits), followed by the head, neck, trunk, stomach, peritoneum, and lungs [1, 6, 7]. According to gender, the occurrence is predominant in the female population [2]. In 90% of patients, pain and tenderness over the tumor are the main symptoms [1, 2].
By the use of the keywords: “glomus tumor” and “abdominal wall” for electronically advanced search, only four case reports are present, of whom only one is described as a malignant glomangiosarcoma in the present literature [6, 8–10]. To our knowledge, the current paper represents the second report of malignant glomus tumor of the abdominal wall (Table 1).
| Reference number . | Author/year . | Age/gender . | Malignancy presence . | Dimensions . | Treatment . |
|---|---|---|---|---|---|
| [8] | Kim et al./2012 | 71/male | No | 14 × 10 × 7 mm | Excision |
| [9] | Dar et al./2020 | 70/male | No | 27 × 22 × 21 mm | Excision |
| [6] | Rodríguez-Justo et al./2001 | 69/male | Yes (sarcoma) | 7.0 × 5.0 mm | Excision |
| [10] | Bezzerra et al./2019 | 45/male | No | 15 × 11 × 0.6 mm | Excision |
| This case report | Trajkovski et al./2023 | 58/female | Yes (low-grade) | 80 × 75 × 70 mm | Excision |
| Reference number | Author/year | Age/gender | Malignancy presence | Dimensions | Treatment |
|---|---|---|---|---|---|
| [ | Kim et al./2012 | 71/male | No | 14 × 10 × 7 mm | Excision |
| [ | Dar et al./2020 | 70/male | No | 27 × 22 × 21 mm | Excision |
| [ | Rodríguez-Justo et al./2001 | 69/male | Yes (sarcoma) | 7.0 × 5.0 mm | Excision |
| [ | Bezzerra et al./2019 | 45/male | No | 15 × 11 × 0.6 mm | Excision |
| This case report | Trajkovski et al./2023 | 58/female | Yes (low-grade) | 80 × 75 × 70 mm | Excision |
Three groups of malignant glomus tumors according to Gould et al. [11] are: locally infiltrative glomus tumors (as in this case report), glomangiosarcoma arising from a preexisting benign glomus tumor, and glomangiosarcoma de novo (round-cell sarcoma), with high mitotic rate.
For almost two decades, the criteria for malignancy of Folpe et al. were used. They comprehended the deep location and size of the tumor more than 2 cm, present atypical mitotic figures, or a combination of moderate to high nuclear grade and mitotic activity [2]. Recently, the World Health Organization established the current criteria for malignant glomus tumors:
Atypical mitotic figures or marked nuclear atypia regardless of mitotic activity and
A glomus tumor of uncertain malignant potential is characterized by a tumor that does not meet the criteria for malignancy but has at least one atypical feature other than nuclear pleomorphism [12].
The tumor in this case presented with a size larger than 2 cm with a deep location (criteria no longer considered for malignancy), with a low proliferative index and infiltrative growth. Therefore, it was classified as a low-malignancy glomus tumor.
The diagnosis of glomus tumor is based on the symptoms and three clinical tests (cold sensitivity test, Love pin test, and Hildreth test). The presence of painful clinical behavior within the tumor should raise suspicion of glomus tumor existence. Additionally, ultrasonography, computerized tomography, and magnetic resonance imaging can be used in diagnostic uncertainties [1, 6]. Definitive pathologic diagnosis is established by immunohistochemistry. Glomus tumors typically show positivity to α-SMA, Muscle-Specific Actin (MSA), and h-Caldesmon. Vimentin (Vim), HHF35 MSA, p53 protein, and CD34 are reported to have variable expression [6, 13]. The tumor in this case report presented with positivity to Vimentin, SMA, and focally to CD34.
Since malignant glomus tumors are symptomatic, surgery is the treatment of choice. Complete excision is required for local recurrence avoidance [8]. The present case is the second report on the large malignant (low-grade) glomus tumor in the abdominal wall with an infiltrative pattern of spread and low proliferative index treated with surgery.
Conflict of interest statement
None declared.
Funding
None declared.



