-
PDF
- Split View
-
Views
-
Cite
Cite
Antonella Nicotera, Gualtiero Canova, Dario Bono, Luca Gattoni, Marcello Zago, Luca Domenico Bonomo, Case report of mesenteric solitary fibrous tumour and review of the literature: ‘once in a blue moon’, Journal of Surgical Case Reports, Volume 2022, Issue 3, March 2022, rjac097, https://doi.org/10.1093/jscr/rjac097
Close - Share Icon Share
Abstract
Solitary fibrous tumour (SFT) is a rare mesenchymal tumour, usually originating from the serous surfaces, typically of the pleura and pericardium. However, it can also have localizations in soft tissues and visceral organs. We report the case of a 79-year-old woman affected by mesenteric SFT, localized in the sigmoid colon. We performed open excision of the abovementioned mass en-bloc with the sigmoid colon and left adnexal tissues. Pathological examination of surgical specimen revealed a diagnosis of SFT CD34+, high-risk of metastases. Mesenteric SFTs are even rarer forms of SFT and may be asymptomatic or cause intestinal occlusion. There is no consensus on the management of this tumour. Radical surgical excision is the only curative treatment, while adjuvant therapies are indicated in case of advanced disease. Due to the high metastatic risk of malignant forms, a long follow-up is mandatory in these cases.
INTRODUCTION
Solitary fibrous tumour (SFT) is a rare mesenchymal tumour, which usually originates from the serous surfaces, typically of the pleura and pericardium. However, it can also have localizations in soft tissues and extra-pleural visceral organs [1]. Diagnosis still represents a challenge from both a clinical and radiological point of view. Moreover, differential diagnosis with other lesions is harder since no standardized guidelines are currently available. Surgical excision is the only treatment with curative intent. Adjuvant treatments, such as radiotherapy and chemotherapy, have no clear indications and are reserved for the more advanced neoplasms [2].
CASE REPORT
We present the case of a 79-year-old woman, whose clinical history reports hypertension, dyslipidemia, type 2 diabetes, obesity, tonsillectomy, open appendectomy, open hysterectomy and subtotal thyroidectomy. After an occasional finding of low haemoglobin level, the patient underwent a gastro-intestinal endoscopy with normal findings and subsequently complete abdomen contrast-enhanced-computed-tomography (CT) scan, which documented a solid mass in the left pelvic quadrant (sizes 84 × 92 × 110 mm) of uncertain origin. The mass was partially non-dissociable from the left round ligament and caused partial compression of the small bowel, without apparent invasion (Fig. 1). A diagnostic/curative laparoscopy was initially indicated. The patient entered the Emergency Room for sub-occlusive condition and was admitted to our Surgical Department. Sub-occlusion was treated conservatively and then, during the same hospitalization, the patient underwent open excision of the mass en-bloc with the colic segment and left adnexal tissues. Nasogastric tube was removed on post-operative day (POD) 1. Bowel opening to gas and stool occurred, respectively, on POD 3 and 4. Oral feeding was resumed on POD 3. Surgical drainage was removed on POD 7. The patient was discharged on POD 8.
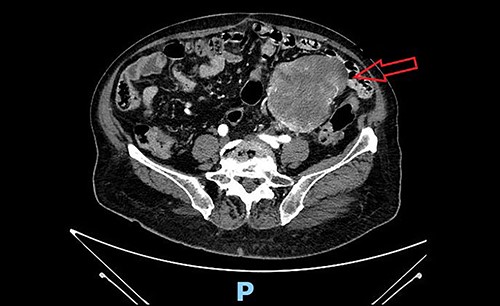
Pre-operative abdominal contrast-enhanced CT scan. Solid mass in the left pelvic quadrant originating from sigmoid mesentery, partially non-dissociable from the left round ligament, is appreciable.
On the histological examination, mass macroscopically appeared grossly oval with partly lobulated margins. Sizes were 150 × 100 mm. Mass was adhered to a 160-mm-long segment of the sigmoid colon. Solid section surface appeared grey-pink coloured and with partial aspects of confluent nodes. Microscopic evaluation revealed a histological-immunophenotypic diagnosis of SFT with the following characteristics: mild–moderate cytological atypia, minimal necrosis (Fig. 2). The immuno-phenotypic study highlighted the following characteristics: vimentin+; CD34+ (intense diffuse); caldesmone+/−; desmin−; CD99−; CD117−; DOG1−, actinMS−; myogenin−; S100−; melanA−; HMB45−; pankeratin− and ALK 1− (Fig. 3). Sigmoid wall and the resection margins were free from neoplasia. Metastasis risk score was 6 (high risk).
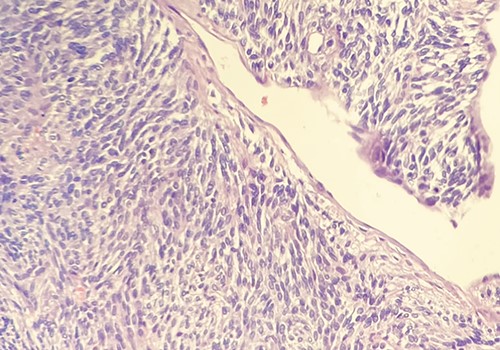
Hematossilin–eosin section showing mild–moderate cytological atypia and minimal necrosis. Courtesy of Dr Ottinetti.
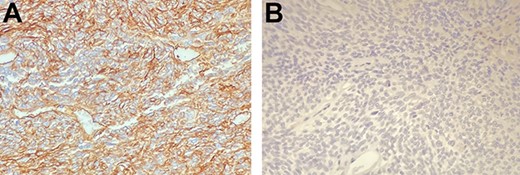
(A and B) The neoplastic cells demonstrate intense diffuse positivity for immunohistochemical staining with CD34 (A), negativity for c-Kit (B). Courtesy of Dr Ottinetti.
Taking into account available guidelines for soft tissues tumours and patient-related and disease-related factors, such as age, co-morbidities and histopathological characteristics, after a careful assessment of risk–benefit, clinical and radiological follow-up with abdominal-chest CT scan every four months was established by a multidisciplinary group. At the first follow-up visit and CT scan (4 months after surgery), there was no recurrent disease (Fig. 4).
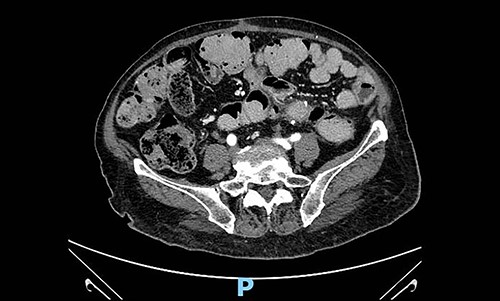
Abdominal contrast-enhanced CT scan 4 months after surgery: no evidence of local recurrence.
DISCUSSION
SFT represents a very rare fibroblastic mesenchymal neoplasm. It was initially included in the group of hemangiopericytomas (HPCs), first described by Stout and Murray in 1942 [3]. However, World Health Organization has recently revised the nomenclature, keeping the term ‘HPC’ only for neoplasms of the central nervous system [4, 5]. Initially, SFTs were described originating from the pleura. However, SFT could be localized not only in the serous surfaces but also in the retro-peritoneum, in the soft tissues of the extremities and of the trunk, and in meninges and orbits [6, 7]. The term SFT, although still used, does not clearly identify the vascular origin of this sarcoma. Indeed, histologically SFTs consist of spindle cells and a high proliferation of blood vessels, supported by a more or less thick texture of connective tissue, with highly cellularity and diffuse positivity for CD34 [8]. The presence of necrosis, haemorrhagic areas, pleomorphism and high mitotic count represent the histological criteria for malignancy. The tumour may vary from benign to very aggressive (poorly differentiated) neoplasm, similar to high-grade sarcoma. A pathognomonic nuclear marker is represented by strong, diffuse nuclear staining of STAT6. This marker is used both with diagnostic and prognostic value [9]. According to Demicco’s classification, SFT can be stratified in 3 degrees of malignancy (low–intermediate–high) considering age, number of mitoses, size of the neoplasm and percentage of necrosis [10].
SFTs can sometimes present in giant forms, causing compressive symptoms or paraneoplastic syndromes (e.g. hypoglycaemia) due to the production of insulin-growth factor 2 (IGF2). The natural history of the disease shows a risk of metastasis ranging from 10 to 15% [11].
Mesenteric SFTs are even rarer forms of SFT and may be asymptomatic or determine intestinal occlusion. In the recent (2020) review by Liu et al., it was estimated a total number of mesenteric SFTs of 21 cases, of which 3 located in the rectum, 11 in the small intestine, 4 in the sigma, 1 in the ascending colon, 1 in the transverse colon and 1 ileocecal. In almost all cases, the neoplasm was benign, while in 3 cases, it was malignant (one localized in the transverse colon, one in the ileum and one in the sigmoid colon). The median age was 54 years (range 26–82) with a male gender prevalence. In all cases, the initial treatment was surgical excision. Data regarding the follow-up were heterogeneous. However, all the authors agreed to assess a long follow-up period, given the high risk of local/distant recurrence in malignant forms. Liu added to the abovementioned list a case of mesenteric SFT localized in the small intestine also treated with surgical excision [12].
The main treatment of localized disease remains radical surgical en-bloc excision. Radiotherapy is considered in advanced disease or with adjuvant intent in selected cases, while chemotherapy and targeted therapies are currently used in clinical trials and show promising results [13, 14]. Currently, the rarity of this neoplasm does not allow to formulate specific guidelines. What is agreed is that radical surgical excision remains the only curative treatment and that a long follow-up is required due to the high metastatic risk in malignant forms.
ACKNOWLEDGEMENTS
We would like to thank Dr Alessandro Comandone for Oncological consultancy and Dr Antonio Ottinetti for histopathological diagnosis.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.
PERMISSION TO REPRODUCE FIGURES
Authors give to Oxford Academic the permission to reproduce all images included for printing and online publication purpose.



