-
PDF
- Split View
-
Views
-
Cite
Cite
Shinichi Ishida, Masato Mutsuga, Takashi Fujita, Kei Yagami, Late-phase left ventricular pseudoaneurysm after three-time mitral valve surgery, Journal of Surgical Case Reports, Volume 2022, Issue 1, January 2022, rjab602, https://doi.org/10.1093/jscr/rjab602
Close - Share Icon Share
Abstract
Rupture of the left ventricular posterior wall is a fatal complication during mitral valve replacement (MVR), and rupture mainly occurs intraoperatively and up to several days after MVR, but it rarely occurs in the late phase, such as several years postoperatively. Late rupture occasionally presents as left ventricular pseudoaneurysms (LVPAs). A 40-year-old man who had a three-time history of mitral valve surgery for infectious endocarditis was incidentally found to have a left ventricular aneurysm with no symptoms. He underwent closure of a LVPA with MVR. Although there are various surgical techniques to close LVPAs after mitral valve repair, it is still a challenging problem. Here, we report the case of a patient who successfully underwent LVPA repair with MVR and describe the technique.
INTRODUCTION
Rupture of the left ventricular posterior wall is a fatal complication during mitral valve replacement (MVR). In the literature, the incidence of this condition is 0.5–2%, and the mortality rate is 65–75% [1]. Rupture mainly occurs intraoperatively and up to several days after MVR, but it rarely occurs in the late phase, such as several years postoperatively. Late rupture occasionally presents as left ventricular pseudoaneurysms (LVPAs). Here, we report a successful case of repair of an LVPA that was detected at the left ventricular posterior wall 4 years after MVR. The patient consent was obtained for publication of this report and the institutional review board approval was excepted for this retrospective report.
CASE REPORT
A 40-year-old man was referred to our hospital with suspected LVPA without any symptoms. He had a three-time history of mitral valve surgery, all of which due to infectious endocarditis (IE). Initial mitral valve repair was performed 8 years ago, and MVR were performed 7 years and 4 years ago. In addition, he had a history of severe atopic dermatitis. He had experienced another episode of IE 1 year ago, which was treated with antibiotic therapy. Enhanced computed tomography (eCT) at that time revealed a small nodular contrast region immediately below the mitral valve annulus in the left ventricular muscle. However, the evaluation could not clearly indicate an aneurysm at that point. Transthoracic echography (TTE) at our hospital revealed an LVPA that gradually expanded at the posterior wall immediately below the mitral valve annulus, and the mechanical prosthetic valve had normal function. eCT revealed a massive LVPA that was >50 mm in size (Fig. 1). We decided to close the LVPA due to a risk of future rupture. Thus, the patient underwent his fourth median sternotomy, through which a cardiopulmonary bypass was established with ascending aortic cannulation and bicaval drainage, and the heart was arrested with antegrade cardioplegia. The mitral prosthetic valve was visualized through a right-sided left atriotomy. After removing the valve, the orifice of the aneurysm was confirmed (Fig. 2). Unlike that in the case of active IE, the tissue around the aneurysm was not vulnerable; thus, the orifice was closed using a bovine pericardial patch (Fig. 3A). In addition, BioGlue (CryoLife Inc., Kennesaw, GA, USA) was injected into the aneurysm (Fig. 3B). The aneurysm was filled with BioGlue to prevent blood flowing into it. Thereafter, MVR was performed using a 25-mm On-X heart valve (CryoLife Inc., Kennesaw, GA, USA). Postoperative eCT revealed complete closure of the aneurysm (Fig. 4). The patient’s cardiac function was good, and no paravalvular leakage was observed on TTE. He was discharged home without complications. At the 10-month follow-up, the size of the aneurysm found to be reduced, as revealed using TTE.
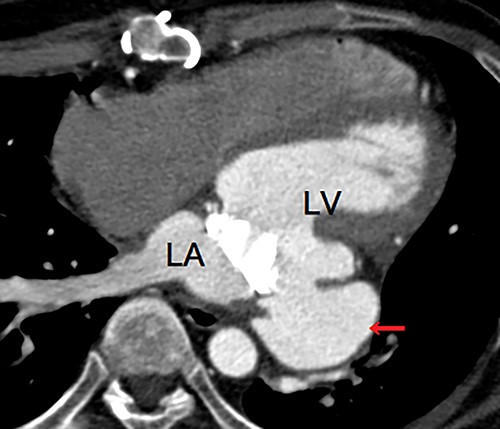
Enhanced computed tomography showing a left ventricular pseudoaneurysm (arrow) just below the mitral valve annulus. LA, left atrium; LV, left ventricle.
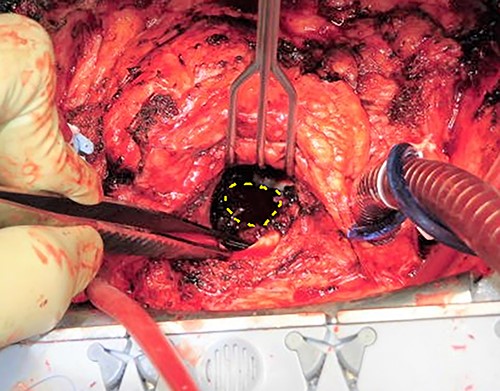
Intraoperative images obtained from the surgeon’s perspective. The orifice of the pseudoaneurysm (surrounded by a dashed circle) was found through the mitral annulus after removing the prosthetic valve.
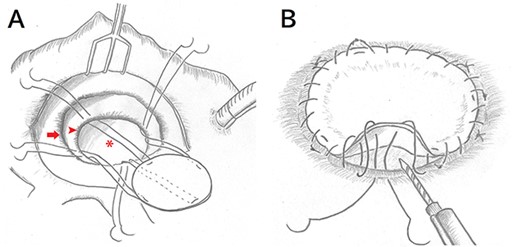
Intraoperative schema after a right-sided left atriotomy. (A) After removing the prosthetic mitral valve and visualizing the aneurysm orifice, four everting mattress stitches with 4–0 polypropylene are placed to fix the orifice of the aneurysm with a bovine pericardial patch at 90° intervals. (arrow, mitral valve annulus; arrow head, aneurysm orifice; asterisk, aneurysm) (B) Running sutures are performed to bind the edge of the orifice and the patch. Before tying the last sutures, BioGlue is injected and filled into the aneurysm.
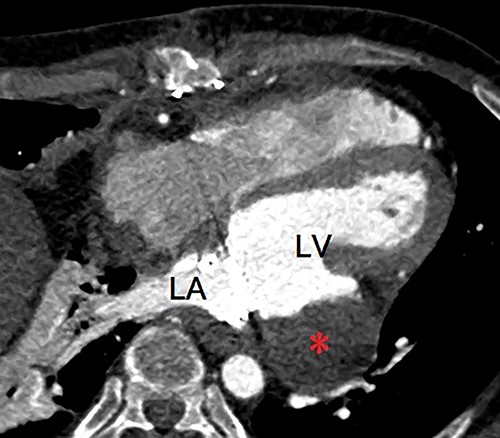
Postoperative enhanced computed tomography showing absent flow into the pseudoaneurysm (asterisk). LA, left atrium; LV, left ventricle.
DISCUSSION
Left ventricular aneurysms can be induced by myocardial infarction, IE, trauma and cardiac surgery. Rupture of the left ventricular wall is one of the devastating complications that occur during MVR. LVPA can sometimes occur in the late phase, but it more frequent in the early phase [2]. In the present case, LVPA was detected in the late phase after MVR. Frequent reoperation could be a risk factor of posterior ventricular rupture [1, 3]. Thus, in the present case, repeated mitral valve surgery could have been the trigger for LVPA. In addition, the most recent episode of IE that was treated only with antibiotic therapy could be a trigger, given the rarity of late-phase LVPA [1]. In a study of 290 cases of LVPA, Frances et al. [4] found that native mitral valve IE can cause LVPA with an occurrence rate of 1%. Furthermore, mitral prosthetic valve endocarditis as the etiology of LVPA has only been reported in a small number of cases [5].
The surgical procedure for LVPA has two approaches: the internal approach, which is initiated from inside of the left atrium, and the external approach, which is initiated from the epicardial surface. In the literature, the internal approach through a median sternotomy is the standard approach [6]. The advantage of this approach is that the operation can be performed by standard extracorporeal circulation, aortic clamp, and cardiac arrest. Furthermore, it is easy to replace the prosthetic valve if structural valve deterioration or other cardiac anomalies are identified. Furthermore, detachment of the severe adhesion is not necessary with this approach. The left thoracotomy approach (external approach) enables easy access to the LVPA in some cases, but it is difficult to completely close the aneurysm orifice from the outside layer with the normal left ventricular wall, which may cause recurrence. In the present case, we did not choose the external approach because detachment of the adhesions of the outside layer was considered difficult, considering it was the fourth reoperation. Furthermore, as preoperative eCT revealed that the orifice of the pseudoaneurysm was immediately below the mitral valve, it was easy to detect the orifice by removing the mitral prosthetic valve. In the literature, the external approach is useful in cases in which the LVPA orifice is located more distally from the mitral valve [2, 7]. Therefore, detecting the location of the LVPA orifice before surgery is important to decide the best approach. Furthermore, the injection and filling of BioGlue into the aneurysm may help prevent suture hole leakage. Consequently, the blood flow into the aneurysm was disappeared and the size of the aneurysm decreased.
In conclusion, closure of late-phase LVPA in a patient with a three-time history of mitral valve surgery was successfully performed. In this case, the internal approach using a pericardial patch and BioGlue for the pseudoaneurysm was feasible and successful.
CONFLICT OF INTEREST STATEMENT
The authors declare that there are no conflicts of interest.



