-
PDF
- Split View
-
Views
-
Cite
Cite
Derqaoui Sabrine, Elouazzani Hafsa, Mkira Omar, Ahmed Jahid, Kaoutar Znati, Bernoussi Zakia, Fouad Zouaidia, Retroperitoneal leiomyoma of gynecologic type: a case report and review of the litterature, Journal of Surgical Case Reports, Volume 2020, Issue 12, December 2020, rjaa489, https://doi.org/10.1093/jscr/rjaa489
Close - Share Icon Share
Abstract
Retroperitoneal leiomyomas are rare benign tumors with smooth muscle differentiation, with only ~1001 cases reported in the English literature to date. Because of its scarcity and non-specific presentation, the preoperative diagnosis might be challenging. On histology, these neoplasms share the same macroscopic, morphological and phenotypic features with uterine leiomyoma; thus, they are referred to as leiomyoma of gynecologic type. Herein, we describe a case of a voluminous retroperitoneal leiomyoma in a 51-year-old woman with a history of total hysterectomy to raise awareness about this condition, as its differential diagnosis might be challenging.
INTRODUCTION
Leiomyoma is benign tumors with smooth muscle differentiation, accounting for the most common gynecologic neoplasms [1]. Retroperitoneal growth pattern in leiomyoma is a rare condition, as only ~100 cases have been reported in the English literature to date [1]. Because of its scarcity and non-specific presentation, the preoperative diagnosis might be challenging [2]. On histology, these neoplasms share the same macroscopic, morphological and phenotypic features with uterine leiomyoma [3]; thus, they are referred to as leiomyoma of gynecologic type [4]. Herein, we describe a case of a voluminous retroperitoneal leiomyoma in a 51-year-old woman with a history of total hysterectomy to raise awareness about this condition, which can have a significant challenging pre-operative diagnosis.
CASE REPORT
A 51-year-old woman presented to the department of surgery, with a 6 months history of an increasing pelvic heaviness and chronic constipation without urinary or gynecological disorders. Her medical history was positive for a total hysterectomy for multiple uterus fibroids, 8 years ago with no remarkable comorbidities. Upon physical examination, she had a palpable mass located in the left flank with a posterior and iliac extension. It was firm with regular borders. The patient’s blood cell counts, serum electrolyte levels, renal and liver functions, CA-125 and carcinoembryonic antigen were all within normal limits. Abdominal magnetic resonance image revealed a 32 × 29 × 12 cm retroperitoneal tissular mass with a signal that is isointense to that in muscle on T1-weighted images, and a homogeneous enhancement by contrast material (Fig. 1). The patient underwent surgical excision of the tumor. Careful exploration of the abdominal cavity revealed the retroperitoneal growth pattern of the mass extending from the pelvis to the flanks without adherences to adjacent structures. On gross, the specimen weighed 1660 g and measured 33 × 28.5 × 12 cm. It was well-circumscribed, firm, white-gray colored at sectioning. Hematoxylin–eosin-stained sections revealed mature smooth muscle cells with blunt-ended nuclei, and abundant eosinophilic cytoplasm arranging in intersected fascicles. The stroma contained vessels with mural hyalinization and myxoid changes (Fig. 2). Necrosis and nuclear atypia were absent. Mitotic count was 1/20HPF without atypical mitotic figures. On Immunohistochemistry, tumoral cells showed strong, diffuse positivity with smooth muscle actin (SMA) desmin, estrogen and progesterone receptors. They were negative for CD117, DOG1, PS100 and CD34 (Fig. 3). After surgery, the patient has had no recurrence (1 year).
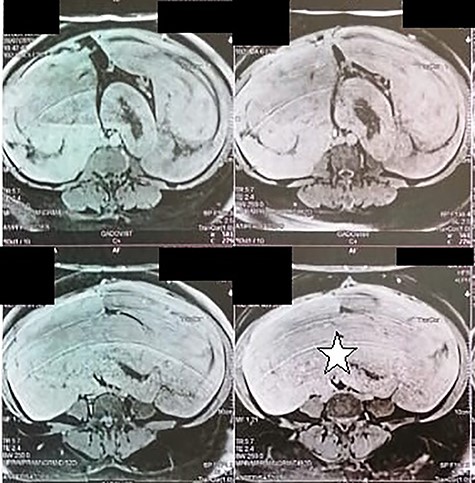
Abdominal magnetic resonance showing a 32 × 29 × 12 cm retroperitoneal tissular mass with a signal that is isointense to that in muscle on T1-weighted images (white star).
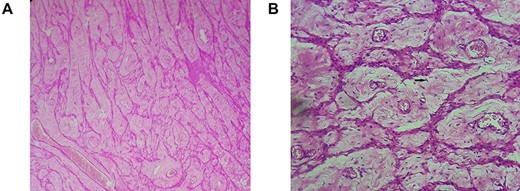
Retroperitoneal leiomyoma’ histology: mature smooth muscle cells with blunt-ended nuclei (black arrow), and abundant eosinophilic cytoplasm arranging in intersected fascicles. (A) Hematoxylin and eosin, ×100. (B) Hematoxylin and eosin, ×400.
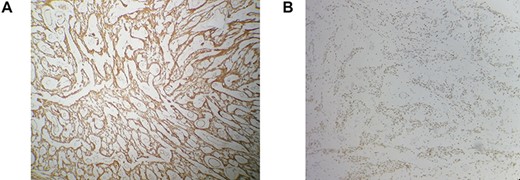
Immunohistochemistry findings: strong and diffuse positivity with desmin (A), estrogen receptors (B). Immunohistochemical stain, ×200.
DISCUSSION
Leiomyomas are smooth muscle benign tumors that rarely involve the retroperitoneum, and most of retroperitoneal tumors are believed to be malignant [5]. Billings et al. [6] have described two distinct subsets of deep soft tissue leiomyoma: (i) leiomyomas of somatic soft tissue which develop in extremities and (ii) retroperitoneal–abdominal leiomyomas. The latter occurs almost exclusively in women during the perimenopausal period [6] with (i) concurrent uterine leiomyoma or (ii) history of hysterectomy for uterine fibroids being, reported in up to 40% of the cases [7]. Clinically, presenting symptoms of retroperitoneal leiomyomas are often non-specific (including discomfort, fatigue, and back pain) or are related to compression of adjacent structures [1, 7]. as these tumors may extend to the upper retroperitoneum, being as high as the level of the renal hilum [7]. Pathogenesis of these lesions remains unclear. In fact, retroperitoneal leiomyomas might arise from the hormonally sensitive smooth muscle elements or the embryonal remnants of Müllerian and Wolffian duct might the cells of origin. They might also represent metastatic or synchronous primary lesions because only nine cases of this condition have been reported in men [1]. In our case, the patient had a history of hysterectomy for uterine fibroids; this supports the pathway of hormonally sensitive embryonal remnants or smooth muscle retroperitoneal elements. On imaging, magnetic resonance imaging (MRI) represents the most reliable technique for evaluating retroperitoneal masses. Classic leiomyomas are isointense to muscle on T1-weighted images and are hypointense to muscle on T2-weighted images, with homogenous enhancement. Although MRI is the most useful imaging modality for these tumors, histopathologic study is required to confirm the diagnosis, as in our case, where the anatomic origin of the mass could not be identified by MRI [7]. On gross, they range in size from 3.2 cm to 37 cm, and present as oval to spherical, well-circumscribed, lobulated, solid masses with a white to gray cut surface. Cystic degenerative and myxoid changes have been described in some cases [6]. Histologically, they share almost the same features with their uterine counterparts. The tumor consists of intersecting fascicles of spindled mature smooth muscle cells, with blunt-ended, or slightly tapered nuclei. The stroma contains numerous vessels with thick wall and mural hyalinization [4, 6]. Varying degrees of hydropic changes like those seen in uterine leiomyoma are present in the majority of tumors. Fatty changes, hyaline necrosis, myxoid changes, epitheliod or cord-like arrangement are commonly seen in retroperitoneal leiomyomas [4]. By definition, classic retroperitoneal leiomyoma lack of atypia, necrosis and significant mitotic activity (no >3 mitoses per 50 HPF) [5]. If mitotic activity is 5/10 HPF or more, these lesions are best referred to as ‘leiomyoma of uncertain malignant potential’; although high mitotic rates might still be compatible with benign condition. Definitive diagnosis requires immunostaining. Neoplastic cells are positive for SMA desmin and caldesmon. Positivity for CD34 has been reported in some cases, whereas tumoral cells are negative for CD117, S-100 protein and HMB45 [2]. Estrogen and progesterone receptors are typically positive [5].
The main differential diagnosis is leiomyosarcoma, which can be primary or metastatic. Useful criteria include mitotic rate, presence of atypia, tumor necrosis and presence of multiple lesions. In addition, eostrogen receptor (ER) positive primary retroperitoneal leiomyosarcoma is a rare condition. Other diagnoses to be ruled out are gastro intestinal stromal tumors (GISTs) with retroperitoneal extension or extra-GISTs; histologic appearance and positivity for CD117 are of diagnostic value. The differential diagnosis also includes PEComa (at least focal HMB45 positivity) and aggressive angiomyxoma (composed of oval cells positive for desmin and only focally for ER). Other tumors to be considered in the differential diagnosis are retroperitoneal space schwannoma, solitary fibrous tumors, retroperitoneal angiomyolipoma and undifferentiated liposarcoma. For the latter, undifferentiated spindle cells, occasional lipoblasts and strong diffuse nuclear MDM-2 positivity can rule out this diagnosis. Thus, the diagnosis is based on a careful histological and immunohistochemical analysis [5, 9].
Molecular genetics of these tumors have not been enough studied. To the best of our knowledge, the first cytogenetical analysis of retroperitoneal leiomyoma has been conducted by Panagopoulos et al. [3], it showed a fusion of the genes KAT6B (10q22) with KANSL1 (17q21) as a result of translocation t(10;17). Surgical excision of the mass is the gold standard treatment; it can be by laparotomy or laparoscopy [8]. In addition, minimally invasive gynecologic surgery and occasionally morcellation might be performed. However, when pre-operative evaluation cannot rule out malignancy, morcellation should not be employed [9]. Retroperitoneal leiomyoma of gynecologic type represents a challenging diagnosis. Awareness of this entity is crucial to distinguish it from other primary retroperitoneal tumors which are, in most of the cases, malignant.
CONFLICT OF INTEREST STATEMENT
None declared.
FUNDING
None.



